Clear Sky Science · en
A comparative study on the corrosion resistance of Ti-6Al-4V produced via material extrusion and other additive manufacturing technologies
Why 3D-Printed Titanium Implants Matter
Many modern hip replacements, bone plates, and dental screws are made from a titanium alloy called Ti-6Al-4V. This metal is strong, light, and usually very resistant to rust-like damage inside the body. New 3D-printing methods promise cheaper, more customized implants, but they also change the tiny inner structure and porosity of the metal. This study asks a simple question with big medical implications: do different 3D-printing routes make this trusted alloy more likely to corrode and release metal into the body?
Different Ways to Print the Same Metal
The researchers compared three advanced 3D-printing methods plus a traditional forged version of Ti-6Al-4V. Two methods, electron beam melting (EBM) and laser powder bed fusion (LPBF), use intense beams to melt layers of loose powder into dense parts. The newer material extrusion (MEX) route instead prints a metal-filled plastic filament into a shape, removes the plastic, and then sinters the packed metal powder into a solid. Although all four routes start from the same recipe of titanium, aluminum, and vanadium, they leave behind very different surfaces, pores, and internal patterns of crystals inside the metal. These hidden differences strongly influence how fluids and dissolved oxygen reach the metal when it is in service as an implant.
Rough Surfaces and Hidden Voids
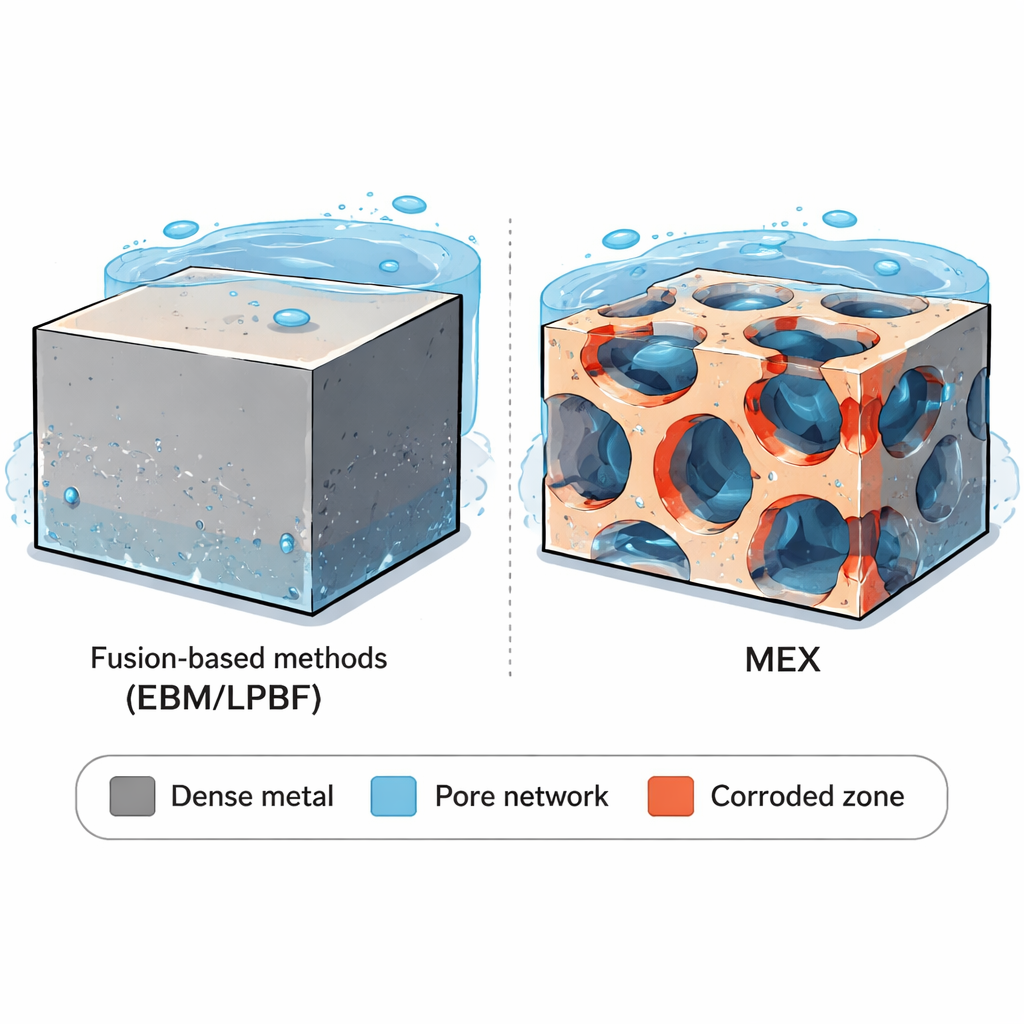
Using 3D surface scans and microscopes, the team found that all 3D-printed samples had rough, wavy outer surfaces. That roughness can be a blessing or a curse. On one hand, it helps bone grow onto implants, improving anchoring. On the other hand, it can also provide shelter for bacteria. The key contrast appeared inside the parts. EBM and LPBF created mostly dense metal with only a few tiny, rounded pores. MEX, in contrast, contained a periodic network of larger, elongated voids aligned with the printed filaments and layers. This built‑in pore network is not just a few isolated flaws: it forms paths that can potentially let liquid penetrate deep into the part. All samples shared the same basic “two-phase” crystal structure, but the shape and arrangement of those phases differed, which can subtly shift how different regions of the alloy respond to corrosive conditions.
How the Alloy Behaves in Body-Like Fluids
To mimic exposure inside the human body, the researchers immersed the samples in salt-based solutions at body temperature and monitored how much current flowed during electrochemical tests, a measure of corrosion activity. In a mildly aggressive, body-like phosphate-buffered solution, all 3D-printed alloys — including MEX — formed a stable, protective oxide film on their surfaces, similar to the traditional wrought metal. After long hours, the currents settled at very low values, indicating excellent overall resistance. Small differences emerged when the surfaces were polished smooth. In that case, MEX parts showed slightly higher currents, hinting that polishing had cut into their large pores and exposed internal surfaces to the liquid, increasing the effective area where corrosion could start. Still, in this gentle environment, even MEX behaved acceptably.
What Happens in Harsher, Acidic Conditions
The story changed in more extreme conditions, meant to simulate local environments that can arise near implants, such as inside tight crevices or inflamed tissue, where the fluid can turn acidic and oxygen may be scarce. Short tests in very acidic salt solutions showed that all versions of Ti-6Al-4V corroded faster, and that certain microscopic regions of the metal dissolved more readily than others. The researchers observed that one of the alloy’s phases (the so‑called alpha phase) tended to corrode slightly faster than the other (beta), creating fine-scale selective attack. However, the overall corrosion rates of the different manufacturing routes still looked similar over short times. In long-term tests lasting many weeks, the deeper impact of pores became clear. The wrought, EBM, and LPBF samples mainly showed gentle, overall thinning with only occasional small pits, and their corrosion rates even slowed as protective layers thickened. MEX parts, however, lost material three to five times faster. Microscopy revealed that once the outer surface was polished, the interconnected macro-pores opened directly into the testing fluid. This allowed the acidic solution to seep along the pore network, enlarging voids and driving corrosion far into the interior.
What This Means for Future Implants
For patients and designers, the key takeaway is reassuring but nuanced. When Ti-6Al-4V is made by modern beam-based 3D-printing methods (EBM and LPBF), its resistance to corrosion in body-like fluids remains comparable to traditional wrought metal. The main concern arises with sinter-based material extrusion: its built-in network of large, connected pores can seriously undermine durability in harsh, acidic environments that sometimes occur around implants. The authors conclude that while all tested routes can produce chemically robust parts under normal conditions, MEX will need better control of porosity — through optimized printing, sintering, or post-treatments — before it can safely match the long-term corrosion performance required for the most demanding biomedical and engineering applications.
Citation: Lorenzi, S., Nani, L., Persico, T. et al. A comparative study on the corrosion resistance of Ti-6Al-4V produced via material extrusion and other additive manufacturing technologies. npj Mater Degrad 10, 32 (2026). https://doi.org/10.1038/s41529-026-00745-4
Keywords: titanium implants, 3D printing, corrosion, biomaterials, additive manufacturing