Clear Sky Science · en
Impedance analysis on the structural evolution of NaCl-induced corrosion products formed on pure titanium
Why tiny grains of salt can threaten mighty jet engines
Aircraft engines often rely on titanium because it is strong, light, and normally resists rust. But when hot titanium parts meet moist, salty air—like in marine or coastal flight paths—salt can trigger a special kind of corrosion that quietly weakens the metal from within. This study explains how common table salt (NaCl) can create microscopic pores inside titanium at high temperature, and shows that an electrical testing technique can spot these hidden defects before they grow into dangerous cracks. 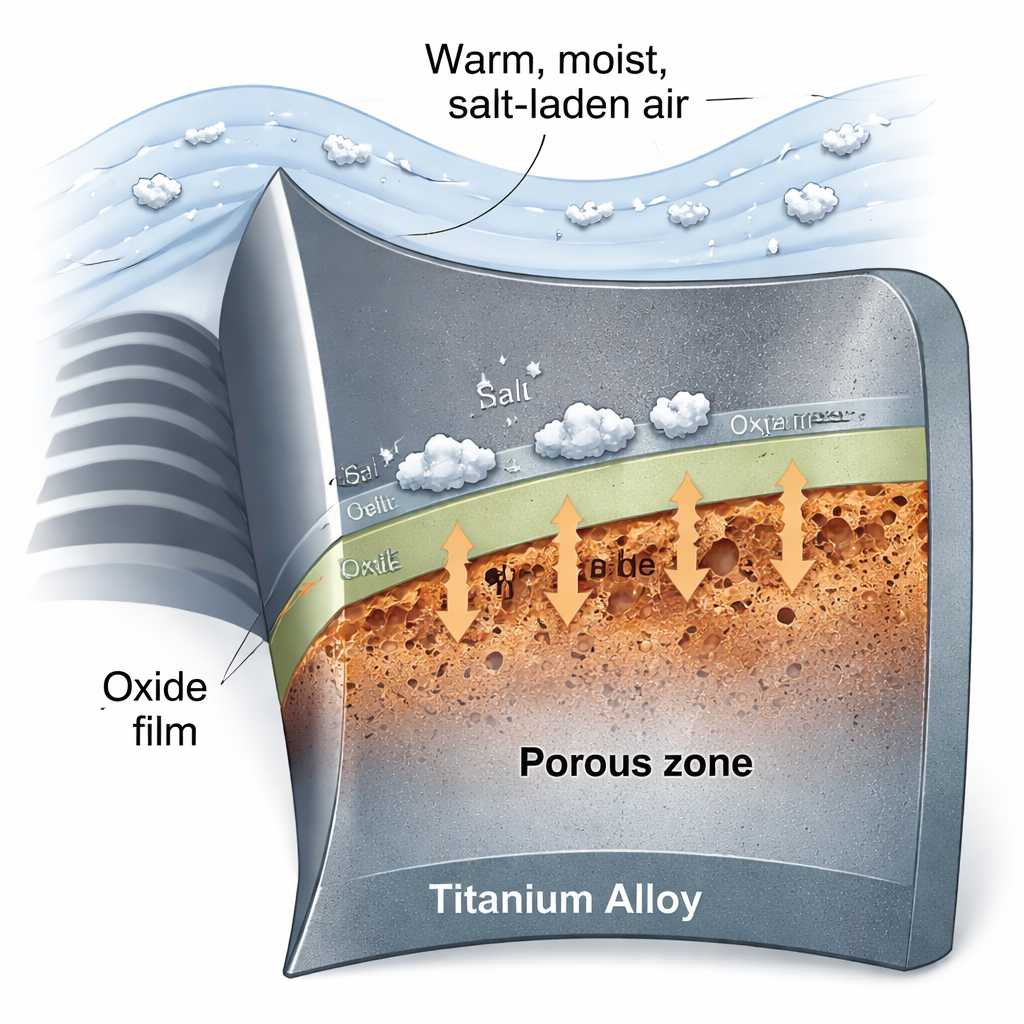
Salt, heat, and hidden damage beneath the surface
Titanium naturally protects itself with a thin, tightly packed oxide film, a kind of built‑in ceramic skin that blocks further attack. In marine conditions at around 600 °C, however, salt crystals landing on this surface begin to react with the oxide. The authors examined very small amounts of NaCl deposited on pure titanium and exposed the samples to hot, wet oxygen—the kind of environment engine parts might see in service. They found that the salt not only speeds up surface corrosion, but also reshapes the internal structure of the corrosion layer, turning it into a porous, sponge‑like region that can severely weaken the metal.
From big voids to fine pores: how the damage evolves
Microscope images revealed two distinct kinds of pores forming in the corroded zone. Larger “macropores” appeared mainly in the outer oxide layer, while finer “mesopores” developed right at the boundary between the oxide and the underlying metal. With very little salt, the oxide film stayed relatively thin and dense, and only macropores formed. As the amount of salt increased, the oxide thickened, corrosion accelerated, and many tiny mesopores appeared in organized, layered patterns inside the titanium. Over time, these mesopores could first grow and then partly disappear again as fresh oxide filled in the gaps.
Chemistry that eats and then patches the metal
The study links these pore patterns to a tug‑of‑war between attack and repair. Salt reacts with the protective oxide and water vapor to form compounds and gases containing chlorine. These hot chlorine‑rich gases can reach the metal and convert titanium into a volatile chloride that escapes, leaving behind empty spaces—mesopores—inside the matrix. At the same time, new oxides grow as oxygen diffuses inward and titanium diffuses outward. Some of these oxides are not the usual fully oxidized TiO₂, but lower‑oxygen forms that eventually transform into denser material. Because titanium oxide expands as it forms, this growth can gradually refill and heal some of the pores, especially once the supply of salt and chlorine dwindles.
Listening to the pores with electrical signals
Directly cutting open engine parts to look for such tiny pores is not practical. Instead, the researchers turned to electrochemical impedance spectroscopy, a method that applies a small alternating electrical signal and measures how the material responds over a wide range of frequencies. They treated the porous corrosion layer like a maze of tiny channels and used an established “transmission line” model to interpret the data. A key finding is that the shape of a standard plot of this data—the Nyquist plot—changes when many mesopores are present. In the high‑frequency range, the curve tilts: when only macropores exist, its angle to the horizontal axis is near 45 degrees, but when abundant mesopores form, the angle drops below about 31 degrees. 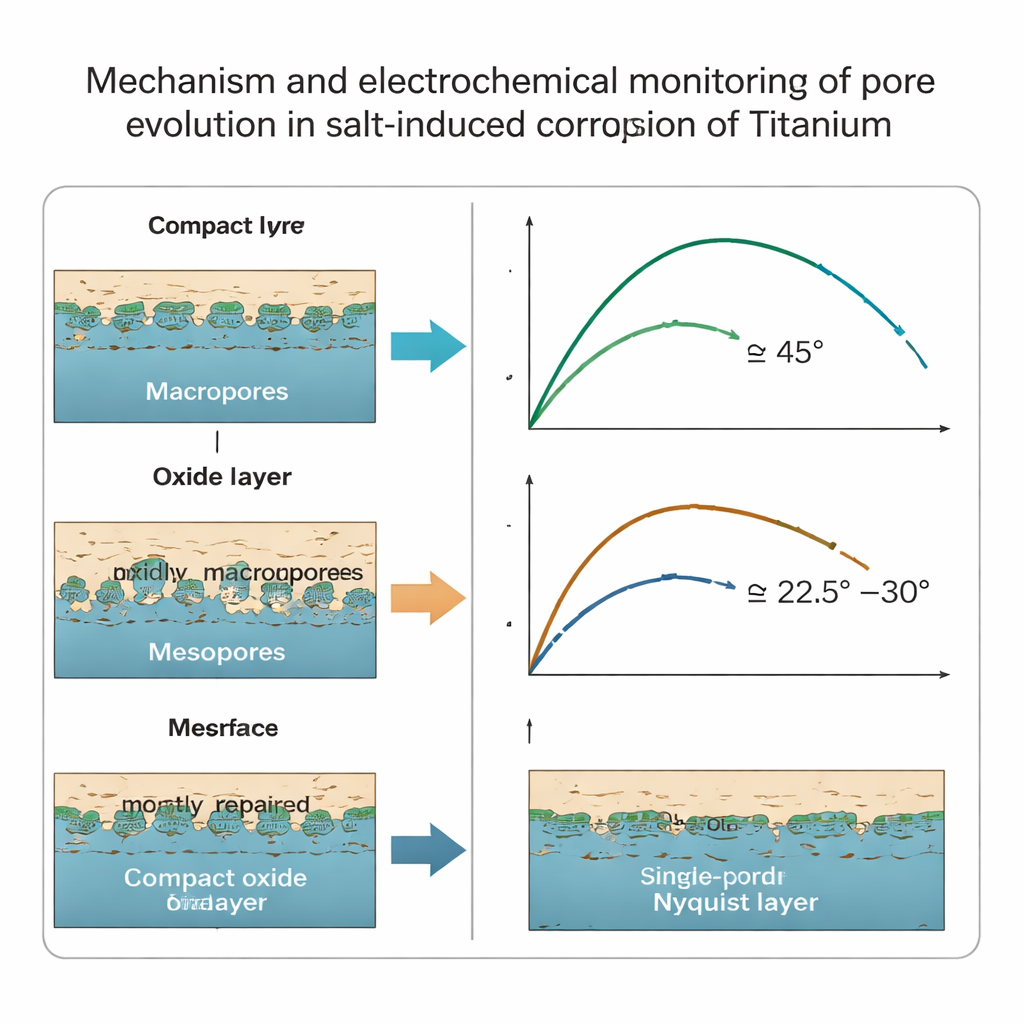
A practical warning sign for crack‑prone damage
For engineers, the most worrying pores are the mesopores at the metal/oxide boundary, because they are prime sites for the start of stress‑corrosion cracks that can lead to brittle, sudden failure. This work shows that by measuring the impedance of hot, salt‑exposed titanium and watching the slope of the Nyquist plot at high frequency, it is possible to tell when these hidden mesopores have formed and when they are being repaired. In simple terms, a high‑frequency angle below roughly 31 degrees is a red flag that aggressive salt‑driven corrosion is dominating and that the metal is developing crack‑ready internal damage—long before any fracture is visible to the naked eye.
Citation: Chen, W., Liu, L., Cui, Y. et al. Impedance analysis on the structural evolution of NaCl-induced corrosion products formed on pure titanium. npj Mater Degrad 10, 30 (2026). https://doi.org/10.1038/s41529-026-00743-6
Keywords: titanium corrosion, salt damage, aircraft engines, electrochemical monitoring, stress corrosion cracking