Clear Sky Science · en
Artificial aging induced changes in ZnO- and TiO₂-based polyacrylic surface coatings
Why long-lasting germ-killing surfaces matter
Door handles, railings, and touchscreens can quietly spread infections, especially in hospitals. One idea to keep them cleaner is to coat surfaces with materials that, when exposed to light, generate tiny reactive molecules that kill bacteria and break down grime. This study asks a practical question: do such smart coatings actually stay safe, strong, and effective over time, or do they slowly destroy themselves while destroying germs?
Light-activated coatings in everyday settings
The researchers focused on clear acrylic coatings—similar to protective lacquers used on furniture or metal—loaded with microscopic particles of either zinc oxide (ZnO) or a common form of titanium dioxide (TiO₂, known as P25). Under ultraviolet A (UVA) light, both materials act like miniature chemical reactors, creating reactive oxygen species that can damage bacteria and decompose organic residues such as fingerprints and dried cells. Because these coatings do not rely on slowly leaking metals like silver or copper, they promise long-term, low-maintenance antibacterial action with minimal release of chemicals into the environment.
Putting coatings through an accelerated "lifetime" test
To mimic years of heavy use in a bright, humid environment, coated stainless-steel plates were placed in a climate chamber for up to nine weeks under continuous UVA light, high humidity, and elevated temperature. Some samples were kept in the same chamber but shielded from light to isolate the effect of UV. Over time, the team tracked how the coatings changed in structure and chemistry using electron microscopy and infrared spectroscopy, and they measured how well the surfaces broke down a dye (a standard test of photocatalytic power) and how quickly they killed two common bacteria: Escherichia coli and Staphylococcus aureus.
Two similar materials, two very different fates
Despite starting from the same acrylic base and being exposed to the same harsh conditions, the ZnO and TiO₂ coatings aged in strikingly different ways. ZnO particles caused only small defects—tiny holes around some particles—and the acrylic layer mostly stayed intact even after nine weeks of UVA. Tests showed only modest shifts in chemical bonds and a slight, non-linear change in photocatalytic activity, likely due to a balance between surface passivation and slow “refreshing” of ZnO by photocorrosion. By contrast, TiO₂ proved far harsher on its host polymer. Under UVA, the acrylic matrix around TiO₂ gradually broke down until it was nearly gone, leaving bare TiO₂ particles sitting on a weakened, porous surface that could shed material when touched.
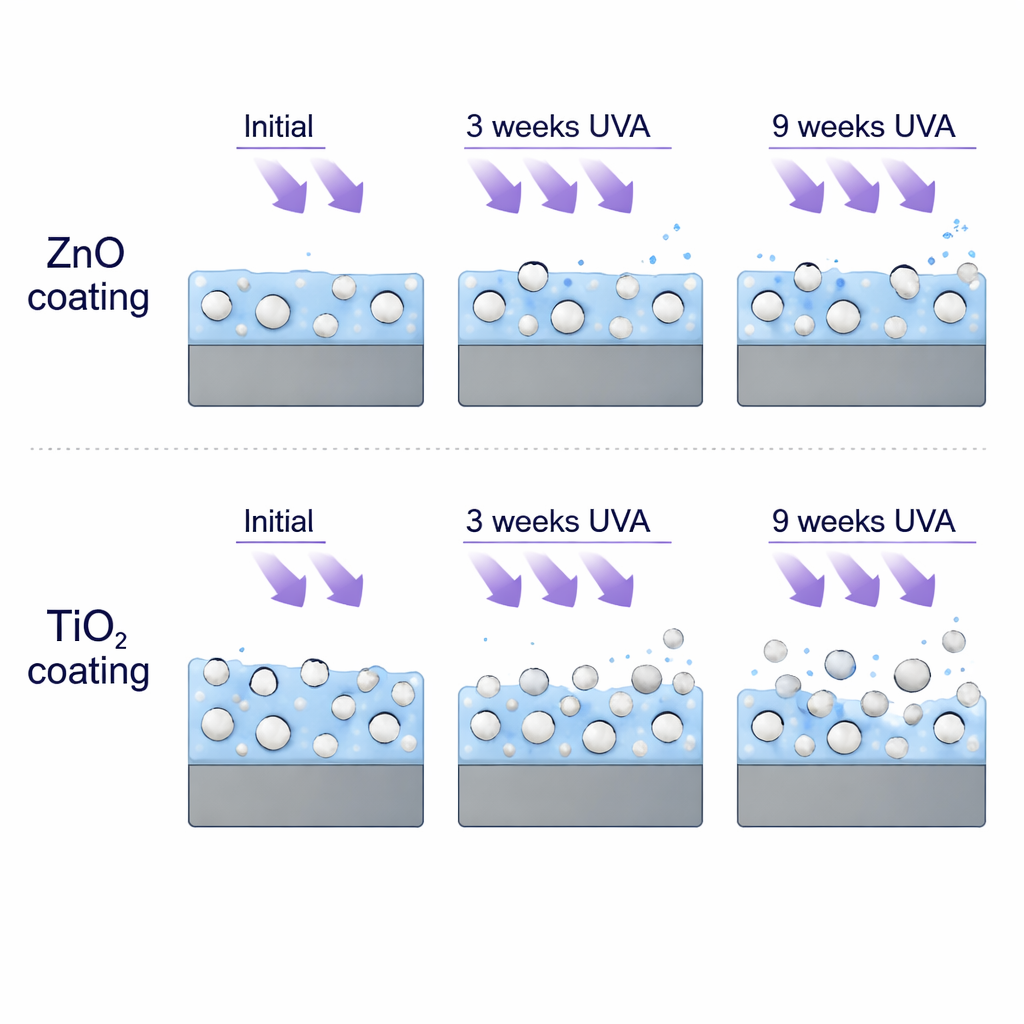
Germ killing versus durability
In the beginning, ZnO-based coatings were clearly better antibacterial performers: under UVA they wiped out around five orders of magnitude of both test bacteria within 20 minutes or less, while TiO₂-coated surfaces needed well over an hour to achieve a comparable effect. The ZnO coatings likely benefit from both light-driven reactive species and the controlled release of zinc ions that further stress and disrupt bacterial membranes. Over time, however, UVA aging reduced the antibacterial strength of ZnO surfaces, even though their light-driven dye degradation and zinc release stayed similar or slightly improved. For TiO₂, the pattern was reversed: as the acrylic binder disintegrated and more particles became exposed, antibacterial activity increased, but at the cost of the coating’s mechanical stability—much of the active material could be rubbed off, making the effect effectively single-use.
Balancing safety, strength, and self-cleaning power
Tests on human skin cells suggested that aged TiO₂ coatings were not toxic and that any potential concern with ZnO lessened with aging under realistic contact times. Overall, the study concludes that, in this acrylic system, ZnO offers a far more stable route to long-lasting self-disinfecting surfaces, even if some antibacterial power is lost with extended light exposure. TiO₂, in contrast, is too destructive to the acrylic binder: it eventually strips away its own support, turning a promising antibacterial coating into a fragile, shedding layer. For designers of future germ-resistant surfaces, the message is clear: choosing the right light-activated material is not just about how fast it kills bacteria—it is also about how gently it treats the coating that holds it in place.
Citation: Kook, M., Peterson, C., Bhat, A.S. et al. Artificial aging induced changes in ZnO- and TiO₂-based polyacrylic surface coatings. npj Mater Degrad 10, 28 (2026). https://doi.org/10.1038/s41529-026-00741-8
Keywords: antibacterial coatings, photocatalytic surfaces, zinc oxide, titanium dioxide, UVA aging