Clear Sky Science · en
The carbon sinking-corrosion dilemma in concrete: insights from early-age CSA-PC mortar
Why locking carbon into concrete is not so simple
Concrete is one of the world’s biggest sources of carbon dioxide, but it can also re-absorb CO2 over time. A new idea is to deliberately force extra CO2 into fresh concrete to “lock” it away and even make the material stronger. This study asks a crucial practical question: if we aggressively push CO2 into a popular low-carbon cement mix at an early age, do we truly gain durability—or do we quietly make the reinforcing steel inside more likely to rust?
Concrete as a hidden carbon sponge
Modern society pours about 30 billion tonnes of concrete every year, and cement-based materials already soak up close to a gigaton of CO2 annually as they slowly react with the air. Engineers are now experimenting with “enforced carbonation,” where fresh or recycled concrete is exposed to concentrated CO2 under pressure. At this early stage the material is still quite porous, so gas can easily penetrate, speeding up the chemical reactions that trap CO2 as solid carbonate minerals. These reactions can also pack the pores more tightly and raise the early strength, offering an attractive route to greener, tougher buildings and infrastructure.
A low-carbon cement mix under the microscope
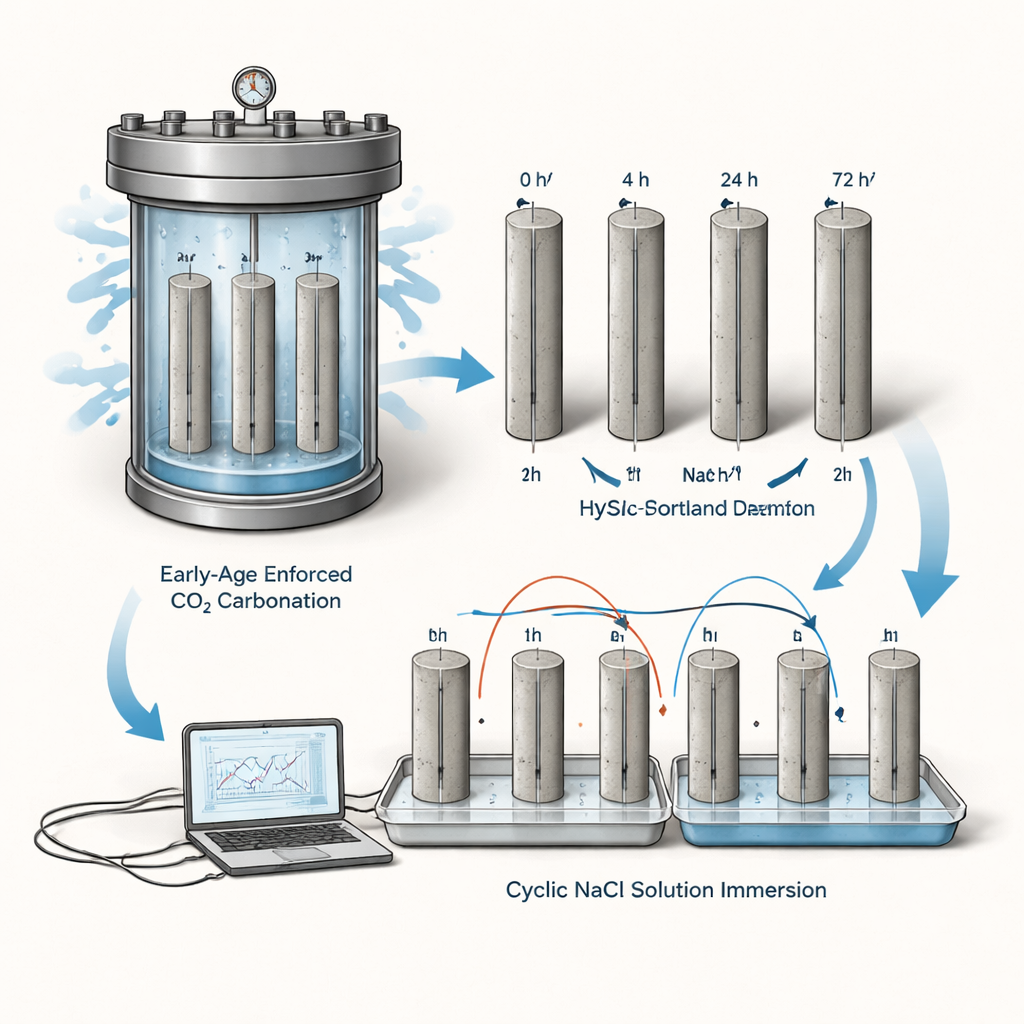
The authors focused on a hybrid mortar made from 75% calcium sulfoaluminate (CSA) cement and 25% ordinary Portland cement. CSA requires less energy and releases less CO2 when produced, but it also creates a less alkaline (less basic) internal environment than standard cement. That matters because steel rebars in conventional concrete are normally protected by a very alkaline pore solution that keeps their surface “passive” and resistant to rust. In this work, slender mortar cylinders, each containing a thin steel rod, were subjected either to no artificial carbonation or to 4, 24, or 72 hours of high-pressure pure CO2 at just one day old. Afterward, all specimens were cured to 28 days and then exposed to repeated cycles of immersion in salty water followed by drying for 43 weeks to mimic harsh, chloride-rich environments.
Watching steel lose its protective shield
Throughout the exposure, the team used electrochemical techniques to track the health of the steel—measuring its open-circuit potential, polarization resistance, and corrosion current density, which together indicate how actively the metal is dissolving. They also periodically measured the mortar’s pH. Even before the aggressive salt exposure, the bulk pH in this CSA-rich mortar was below the conventional threshold (around 11.5) needed for a robust passive film on steel. As the wet–dry cycles proceeded, pH dropped further, especially in the pre-carbonated samples. The corrosion current in carbonated mortars quickly rose to values about ten times higher than in the non-carbonated reference, corresponding to a “high” corrosion rate. In other words, while all bars were at risk, early-age enforced carbonation clearly pushed the steel into a more severe corrosion regime.
Rust that spreads and fills the concrete
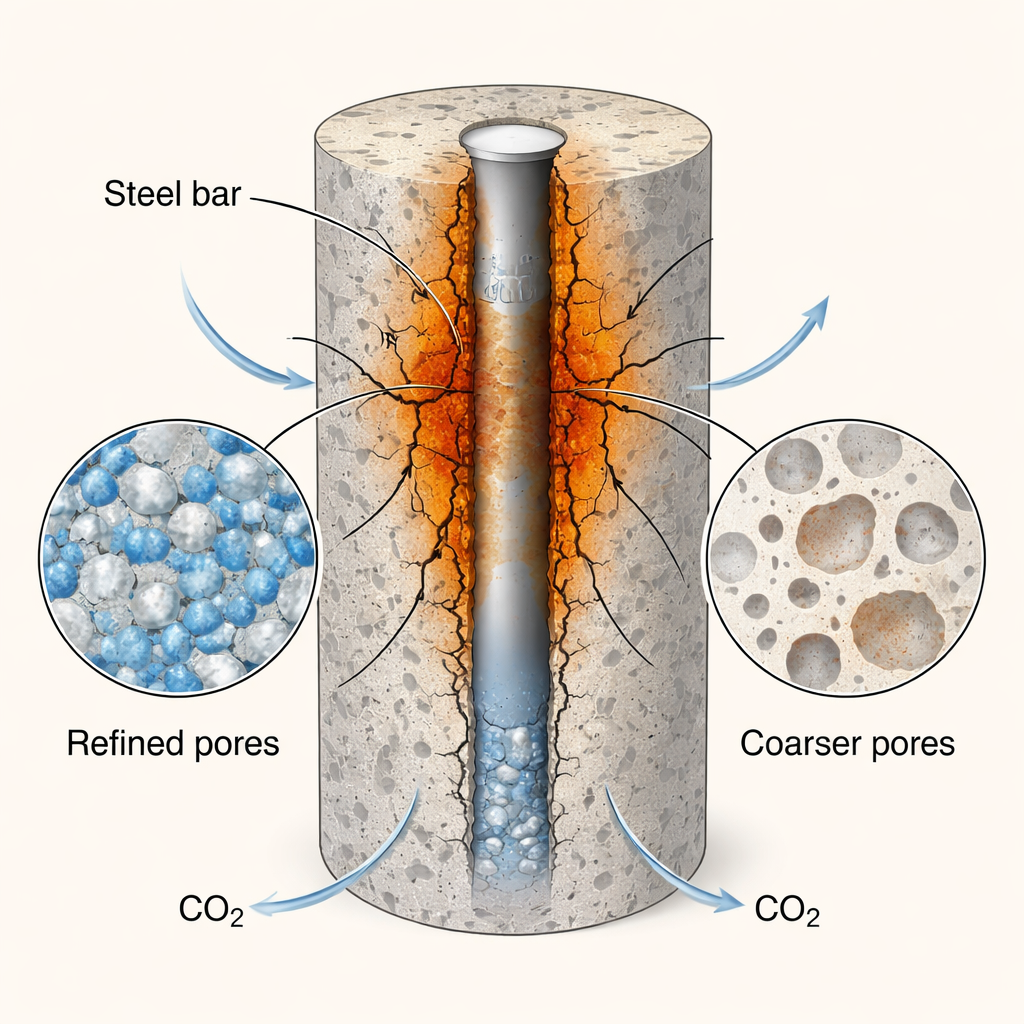
To see where and how the damage developed, the researchers turned to high-resolution imaging and chemical analysis. X-ray computed tomography provided 3D maps of rust-filled zones around the rebars, while backscattered electron microscopy and elemental mapping revealed how iron-rich corrosion products had migrated into the surrounding mortar. In the non-carbonated samples, only a thin rust layer clung to the steel, invading the mortar by just a few tens of micrometers. In contrast, the carbonated mortars showed much thicker and more irregular rust bands, with corrosion products penetrating up to about 2 millimeters into the matrix and forming clusters whose average volume roughly doubled after only 4 hours of early CO2 treatment. X-ray photoelectron spectroscopy confirmed that the steel surface in carbonated specimens contained more high-valence iron oxides and hydroxides and more bound water—signatures of a thicker, more active rust layer that is prone to further attack.
A denser material that still corrodes faster
Paradoxically, the same carbonation that sped up corrosion also made the mortar’s microstructure denser. Thermal analysis and nitrogen sorption measurements showed that outer regions of the cylinders developed more calcium carbonate and a shift from larger pores to finer micro- and mesopores, while inner regions near the steel were altered both by carbonation and by the inward growth and outward migration of rust. Overall, the pore network became tighter, which should, in principle, slow the movement of aggressive ions like chlorides and limit how far corrosion products can spread. The study indeed saw that extending carbonation from 4 to 72 hours did not greatly increase the total volume of rust, but mainly changed how it was distributed—more numerous, shallower rust zones rather than a few large ones—because the refined pores hindered further penetration.
What this means for greener concrete
For a non-specialist, the key message is that pumping extra CO2 into young, steel-reinforced concrete is a double-edged sword. It does help lock away carbon and makes the material’s internal pores smaller and more tightly packed. However, in a low-alkaline system like this CSA–Portland blend, deep early carbonation also strips away much of the chemical protection that normally keeps steel from rusting. The result is more frequent corrosion initiation and more rust spreading into the concrete, even if the dense microstructure limits how deep that rust can go. The authors conclude that while enforced early-age carbonation has clear environmental and mechanical advantages, it can seriously compromise the long-term durability of reinforced members unless the chemistry and design are very carefully controlled.
Citation: Qiang, Z., Yan, L., Yue, Q. et al. The carbon sinking-corrosion dilemma in concrete: insights from early-age CSA-PC mortar. npj Mater Degrad 10, 24 (2026). https://doi.org/10.1038/s41529-026-00737-4
Keywords: concrete carbonation, steel corrosion, calcium sulfoaluminate cement, CO2 sequestration, reinforced concrete durability