Clear Sky Science · en
Effect of electrochemical water treatment processes on carbon steel corrosion in urban water supply system
Why Your Tap Water Pipes Matter
Electrochemical water treatment systems are becoming more common in cities because they efficiently remove pollutants, soften water, and kill germs. But the same electrical reactions that clean the water can also quietly eat away at the metal pipes carrying that water to our homes. This study looks at how such treatment affects carbon steel, a common pipe material, and explains why corrosion can speed up dramatically when electricity and everyday tap water chemistry interact.
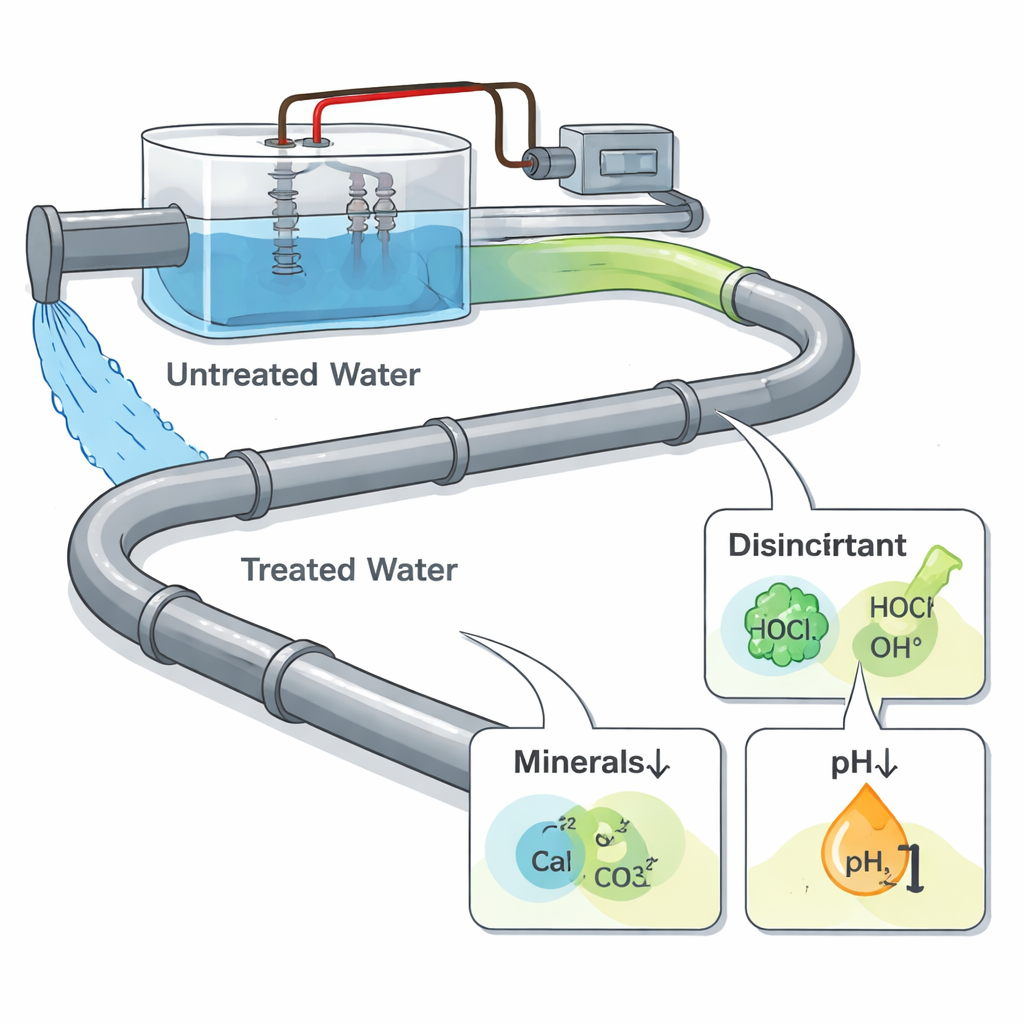
Cleaning Water With Electricity
Electrochemical water treatment passes an electric current through water using metal plates called electrodes. This can remove unwanted ions, break down organic pollutants, and generate disinfectants, making the process attractive for urban and industrial systems. Unlike traditional treatment, however, the pipes themselves are not wired into the system—they just see the treated water that leaves the unit. That means any extra corrosion is driven not by electricity directly hitting the pipe, but by changes in water chemistry: new oxidizing chemicals for disinfection, shifts in acidity, and movement of mineral ions that normally help form protective layers inside pipes.
Faster Metal Loss and Deeper Hidden Damage
The researchers ran a 90-day experiment with real city water, exposing carbon steel samples to different voltages from 0 to 9 volts, then tracking weight loss, surface damage, and electrochemical behavior. They found that even within the first three days, turning on electrolysis made corrosion worse, and higher voltage always meant more damage. At 9 volts, the general corrosion rate jumped more than 25-fold compared with water that had not gone through the electric process. Using 3D surface scans, they also showed that tiny pits in the metal grew much deeper as voltage increased. Under the highest voltage, the deepest pits were nearly five times deeper than in untreated water, sharply shortening the expected service life of the steel even if the average metal loss did not seem extreme.
Why Protective Layers Fail
Inside many water pipes, minerals from the water naturally build a thin, dense layer that helps shield the metal. In untreated water, calcium carbonate plays this protective role. In the electrochemically treated water, however, that mineral protection largely disappears. Because the cathode in the treatment unit attracts calcium and carbonate ions and encourages them to form deposits there, fewer of these ions remain to build a solid shield on the pipe walls. Microscopy and diffraction tests revealed that, instead of a tight mineral-rich barrier, the steel in treated water developed a loose, porous rust layer with small, unstable crystals. This weak coating let corrosive species and electrons move easily, so corrosion stayed fast instead of slowing down over time.
A Stronger Chemical Attack in the Water
To pinpoint what was driving the extra attack, the team separated different factors. When they ran electrolysis in a sulfate solution with no chloride, the by-products of splitting water—like oxygen, hydrogen, and short-lived radicals—barely changed the steel’s corrosion behavior. But when chloride, the same ion found in ordinary salts and many water supplies, was present, the story changed. Electrolysis converted part of the chloride into hypochlorite, a more reactive disinfectant. Electrochemical tests showed that as current increased, steel became much easier to corrode in chloride solutions. Computer simulations backed this up: hypochlorite lowered the energy barrier for iron atoms to leave the metal surface, making corrosion proceed more readily than with chloride alone. At the same time, the rapid formation of rust products consumed hydroxide ions, nudging the water toward lower pH, or more acidic conditions, which further favored corrosion.
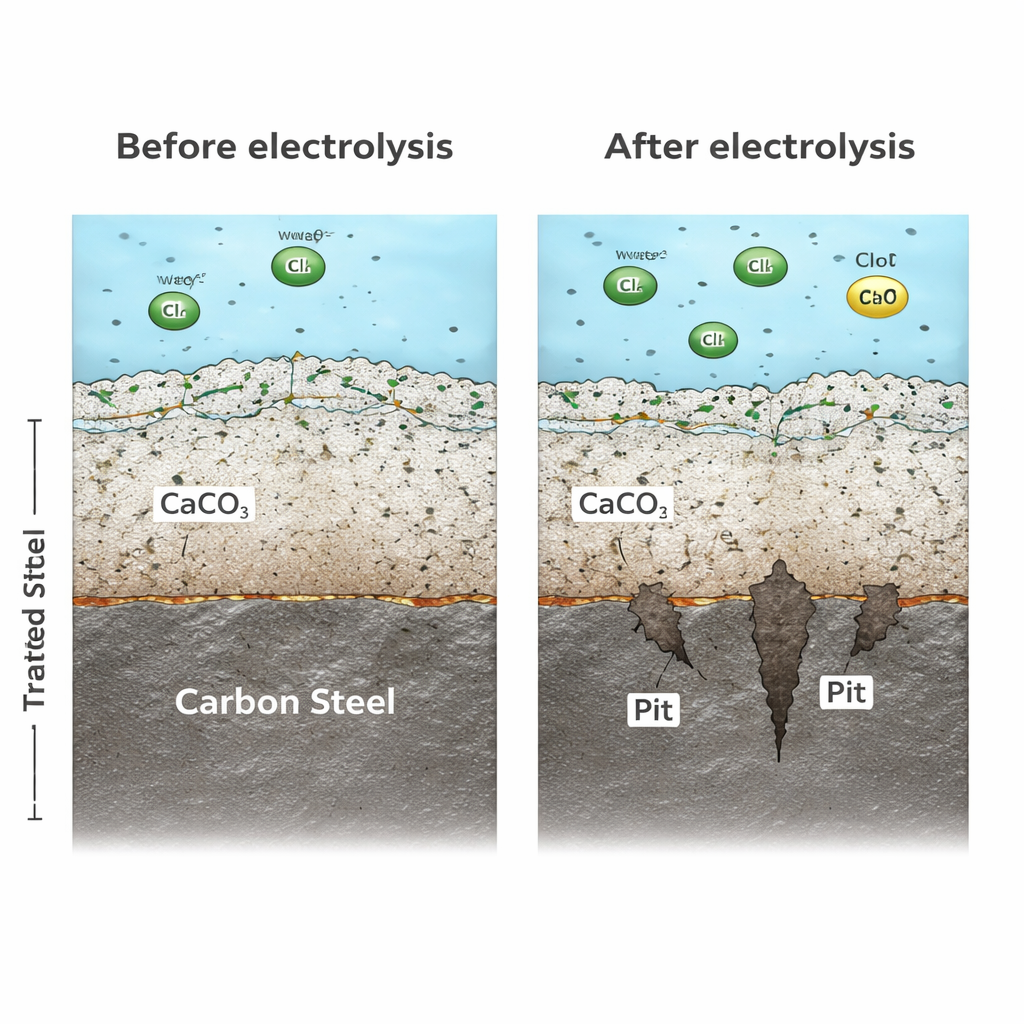
What This Means for Water Systems
For non-specialists, the takeaway is that cleaning and disinfecting water with electricity is not automatically gentle on the pipes that carry that water. In systems where the water contains chloride, electrochemical treatment can turn some of that chloride into a stronger, more corrosive disinfectant while stripping away mineral ingredients needed to build natural protection. The result is faster metal thinning and deeper pits that can shorten pipe life and raise the risk of leaks or contamination. The authors suggest that designers and operators of such systems treat corrosion as a central design concern—by limiting voltage, reducing chloride before treatment, choosing electrodes that form fewer aggressive by-products, and monitoring pitting and water chemistry over the long term—to ensure that cleaner water does not come at the cost of failing infrastructure.
Citation: Zhao, S., Jing, Y., He, X. et al. Effect of electrochemical water treatment processes on carbon steel corrosion in urban water supply system. npj Mater Degrad 10, 23 (2026). https://doi.org/10.1038/s41529-026-00736-5
Keywords: electrochemical water treatment, carbon steel corrosion, urban water pipes, chloride and hypochlorite, water infrastructure durability