Clear Sky Science · en
4A tetrahedron system: a synergistic framework for panvascular intervention empowered by flexible electronics
Rethinking Care for Our Blood Vessels
Panvascular disease—damage to blood vessels throughout the body—is now the world’s leading killer. It can strike the heart, brain, kidneys, and limbs, often with little warning. This article lays out a bold new blueprint for tackling these disorders using ultra-thin, bendable electronics and artificial intelligence. Instead of treating each procedure as a one‑off event, the authors imagine a lifelong, data‑driven partnership between patient and device, from diagnosis to surgery to long‑term follow‑up.
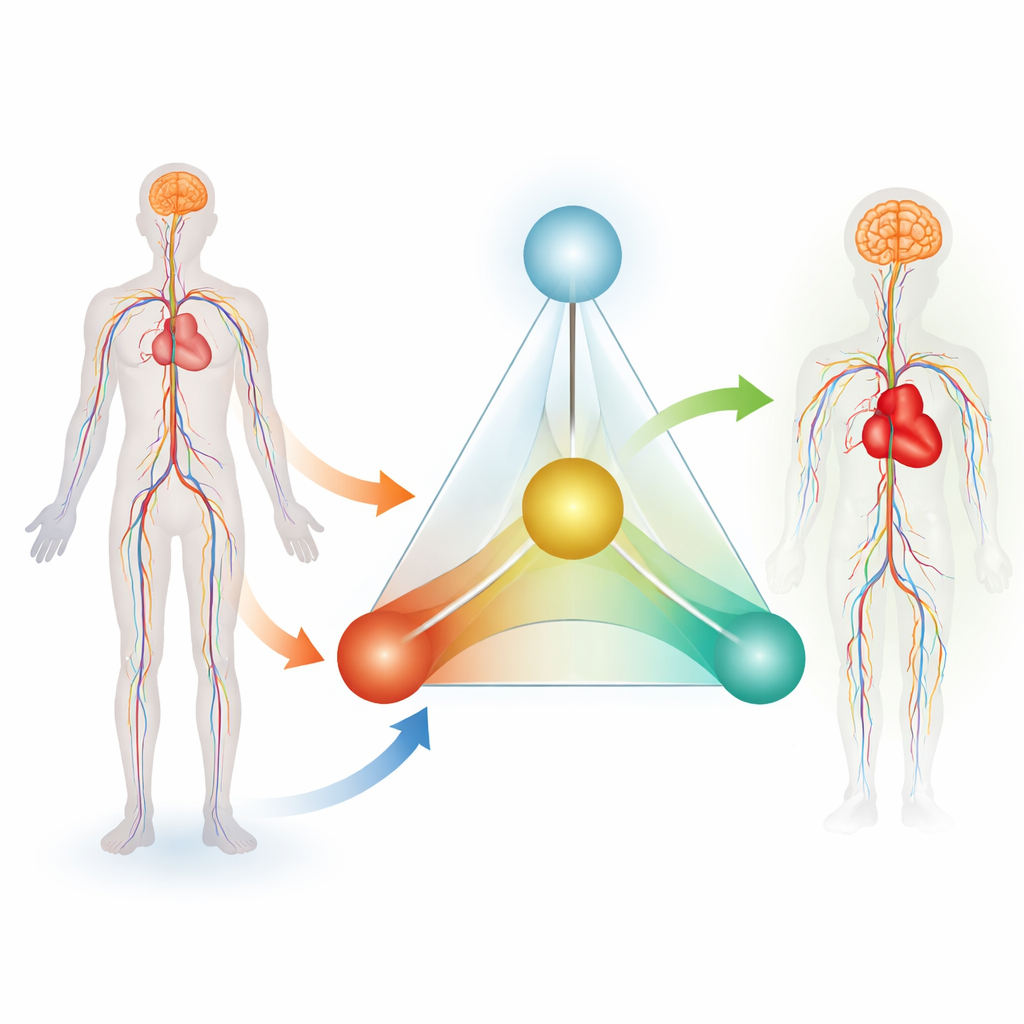
A Big Idea Built on Four Pillars
The authors propose what they call the “4A Tetrahedron System,” a four‑part framework for managing vessel disease as a continuous process. The four A’s are Assessment, Assistance, Aftercare, and AI‑retrofit. Assessment covers how doctors evaluate both the patient and the tools before a procedure. Assistance focuses on guiding instruments safely through tortuous arteries and veins during surgery. Aftercare deals with monitoring the patient afterward, ideally around the clock. Tying these three together is AI‑retrofit, in which artificial intelligence learns from all the data generated along the way and feeds the insights back to improve future devices and decisions. Together, these four elements are pictured as a three‑sided base (Assessment, Assistance, Aftercare) supporting an apex (AI), forming a tetrahedron that represents a closed, self‑improving loop of care.
Gentle Testing and Smarter Implants
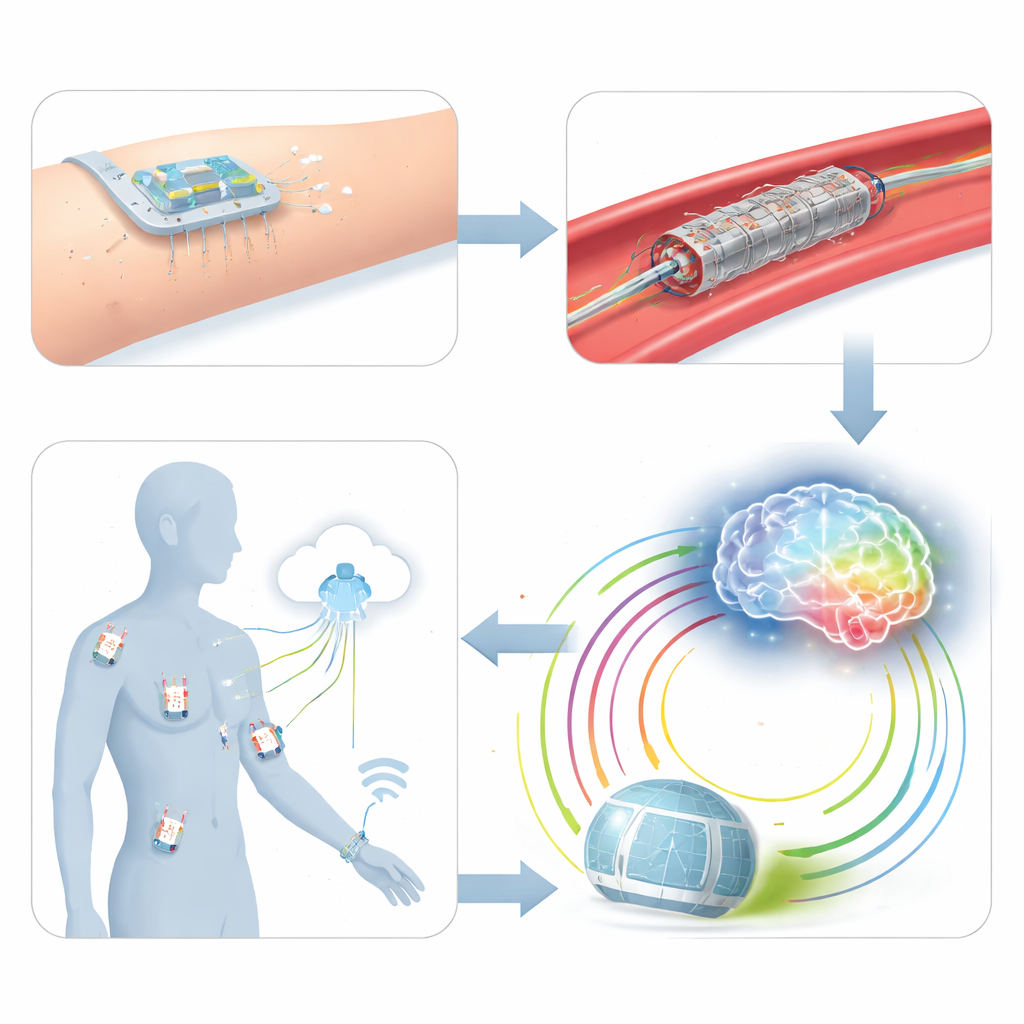
In the Assessment stage, the authors highlight flexible microneedle patches that barely penetrate the skin to sample the clear fluid between cells. This fluid closely mirrors blood chemistry, yet can be tapped in a nearly painless way. Embedded sensors in these tiny needles can track markers of inflammation, kidney function, blood sugar, and more, effectively shrinking parts of the laboratory onto a patch worn on the skin. At the same time, the review describes how traditional implants—such as stents, artificial valves, and heart defect plugs—could be upgraded with ultra‑thin pressure, chemical, and strain sensors. These “smart” devices would not just reopen blocked vessels; they would continuously measure the mechanical forces, cell behavior, and chemical environment at the implant site, revealing whether the vessel is truly healing or quietly drifting toward trouble.
Guiding Tools from the Outside In
The Assistance stage tackles a common problem in modern interventions: navigating guidewires and catheters through complex vessel networks while minimizing X‑ray exposure and dye use. The authors point to emerging flexible ultrasound patches and photoacoustic probes that can give real‑time pictures of vessels without radiation. They also describe magnetically steerable soft guidewires and micro‑catheters whose tips can be deflected by external magnetic fields and driven by robotic arms. Paired with flexible sensors and smart imaging, these systems aim to thread devices through hair‑thin or sharply bending vessels with sub‑millimeter precision, reducing the risks of vessel tears, strokes, or failed procedures.
Living with a Network of Silent Guardians
After a procedure, many patients today return to sporadic clinic visits and short check‑ups, which can easily miss silent restenosis or clot formation. In the Aftercare stage, the authors envision an “Internet of Medical Things”: a web of wearable, semi‑implantable, and fully implantable flexible sensors spread across organs and tissue depths. Skin patches might track heart rhythm and breathing, microneedles could watch glucose and inflammatory markers, and implanted sensors on stents or arteries would follow local pressure and flow. All of these would send data wirelessly to cloud systems, creating a “Medical Wireless Sensor Network” that keeps doctors virtually at the bedside and captures how disease unfolds across the whole body, not just at a single treated spot.
Letting Algorithms Close the Loop
At the top of the tetrahedron sits AI‑retrofit, where algorithms turn raw sensor streams into better care. Machine‑learning models can help design more reliable microneedles and smarter stents, recognize risky patterns in chemical and pressure data before symptoms appear, and guide surgical robots along safer paths. Deep reinforcement learning—software that learns by trial and error on historical data—could suggest how to adjust drugs or when to re‑intervene for each individual patient. Large language models may one day weave together sensor readouts, scans, and medical records into clear summaries for clinicians and understandable explanations for patients. In this vision, every procedure feeds information back into the system, sharpening future decisions.
From Concept to Everyday Care
To a lay reader, the core message is that vessel disease care can shift from isolated, reactive fixes to an intelligent, continuous partnership among the body, flexible electronics, and AI. Microneedle patches make testing gentler, smart implants watch over vulnerable sites from within, wearable and implanted sensors keep tabs on the whole body, and learning algorithms connect the dots over months and years. Many hurdles remain—from long‑term safety and power supply to data security and regulation—but the authors argue that over the next decade, this 4A Tetrahedron approach could turn today’s one‑time vascular procedures into an evolving, personalized protection system against the world’s deadliest group of diseases.
Citation: You, L., Qu, Y., Chen, Y. et al. 4A tetrahedron system: a synergistic framework for panvascular intervention empowered by flexible electronics. npj Flex Electron 10, 35 (2026). https://doi.org/10.1038/s41528-026-00537-5
Keywords: flexible electronics, vascular disease, smart implants, wearable sensors, medical AI