Clear Sky Science · en
Harnessing piezoelectric poly L lactic acid for enhanced sensing in aortic annuloplasty
Listening to the Heart’s Stitches
When surgeons repair a leaking heart valve, they often reinforce the base of the aorta with a supporting ring. That repair has to withstand millions of heartbeats, but today doctors mostly check how it is doing with snapshots from scans long after surgery. This study explores a new kind of temporary, body‑friendly electronic ring that can feel the heart’s movements and convert them into tiny electrical signals, offering a way to “listen” to the repair in real time without leaving permanent hardware behind. 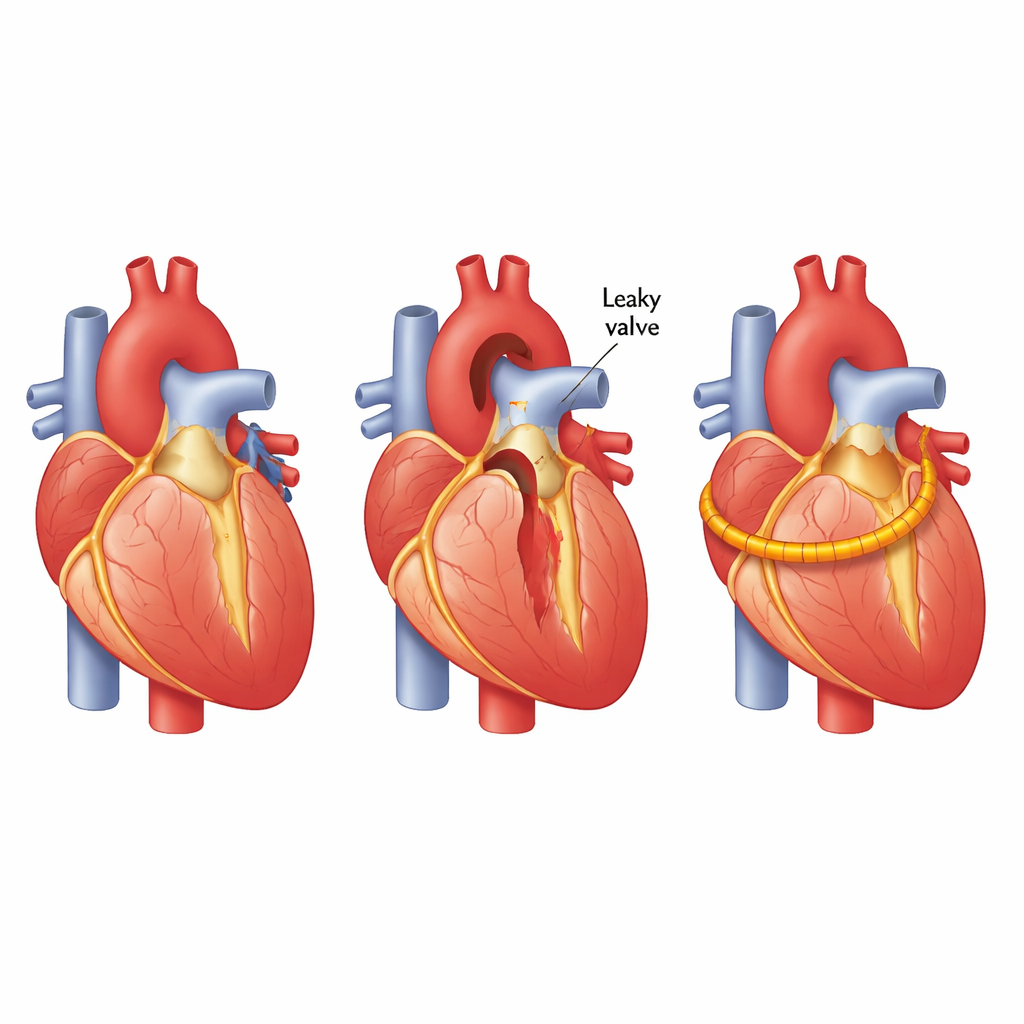
Why Mending Heart Valves Is So Tricky
The aortic valve controls blood flow from the heart to the body. In some people, the base of the aorta stretches or the valve becomes leaky, forcing the heart to work harder and eventually leading to serious illness. Surgeons can avoid replacing the valve with a mechanical one by tightening the area using an annuloplasty ring. This preserves the patient’s own valve and avoids lifelong blood‑thinning drugs. However, once the chest is closed, doctors have little direct information about the actual forces on that ring as the heart beats. Existing measurement tools are bulky, non‑biodegradable, and not suitable to leave inside the body for long periods, leaving an information gap about how the repair behaves over time.
A Plastic That Feels and Then Disappears
The researchers turn to poly‑L‑lactic acid (PLLA), a plastic already used in medical sutures and implants because the body can safely break it down over months or years. PLLA has another useful trait: when its internal molecules are lined up in the right way, it becomes piezoelectric, meaning it generates a small voltage when squeezed, stretched, or bent. On its own, though, raw PLLA does not produce a strong enough signal to be useful as a sensor. The team used a simple, energy‑efficient recipe: they dissolved PLLA, cast it into thin films, stretched those films so they became twice as long, and gently heated them. This treatment reorganized the material’s microscopic structure, boosting its ability to convert mechanical motion into electrical signals while preserving its strength and biodegradability.
Putting the Smart Ring to the Test
To see how well the processed PLLA worked, the team subjected the films to different kinds of movement: repeated stretching, tapping, bending, and controlled vibrations. Unprocessed films produced almost no electrical response, but once stretched and heat‑treated, the same plastic generated much stronger voltages and currents. The more the film had been stretched, the larger the signals became, confirming that the microscopic rearrangement of the material really did turn it into a sensitive motion detector. These experiments also showed that the films responded predictably as forces and vibration frequencies changed, an important requirement for use in the constantly moving environment of the heart. 
Simulating a Beating Heart in the Lab
Building on these results, the researchers fashioned a ring‑shaped sensor from the most responsive PLLA film and added thin silver electrodes so they could collect the tiny voltages it produced. They then mounted this flexible ring around a 3D‑printed model of the aortic root in a laboratory setup that mimicked a human left heart. By pumping fluid to create realistic blood pressures, they could compare pressure waves in the model “aorta” with the ring’s electrical output. As they increased the simulated blood pressure from normal to high values, the PLLA ring produced larger voltage swings, from about minus 0.5 to plus 0.5 volts at low pressure up to about minus 1.1 to plus 1.3 volts at the highest pressure. The signals were stable, repeated with each beat, and closely tracked the timing and size of the pressure pulses.
What This Could Mean for Future Heart Surgery
For non‑specialists, the key message is that the team has created a thin, flexible plastic ring that can feel how hard the heart is pulling and pushing on a repair and translate that into simple electrical signals. Because the material is both biocompatible and biodegradable, such a ring could, in principle, be left in place only as long as needed and then safely disappear as the patient heals. While this work was done in a realistic lab model rather than in people, it shows that a dissolving sensor can reliably monitor heart‑like pressures. In the future, similar devices might guide surgeons as they adjust valve repairs and keep watch afterward, offering continuous feedback without permanent electronics inside the body.
Citation: Merhi, Y., Montero, K.L., Johansen, P. et al. Harnessing piezoelectric poly L lactic acid for enhanced sensing in aortic annuloplasty. npj Flex Electron 10, 31 (2026). https://doi.org/10.1038/s41528-026-00533-9
Keywords: aortic valve repair, biodegradable sensors, piezoelectric plastics, cardiac surgery monitoring, flexible electronics