Clear Sky Science · en
Flexible ferroelectric biomaterials for skin, neural, and musculoskeletal tissue repair
Smart materials that help the body heal itself
When we break a bone, tear a tendon, damage a nerve or develop a stubborn skin wound, doctors can often stabilize the injury—but getting the body to fully rebuild healthy tissue is still hard. This article explores a new class of “smart” materials that flex with our bodies and turn everyday movements into tiny electrical signals. Those signals imitate the body’s own bioelectric language and can gently nudge cells to grow, reorganize, and repair damaged skin, nerves, muscles, cartilage, and bone.
Why electricity matters for healing
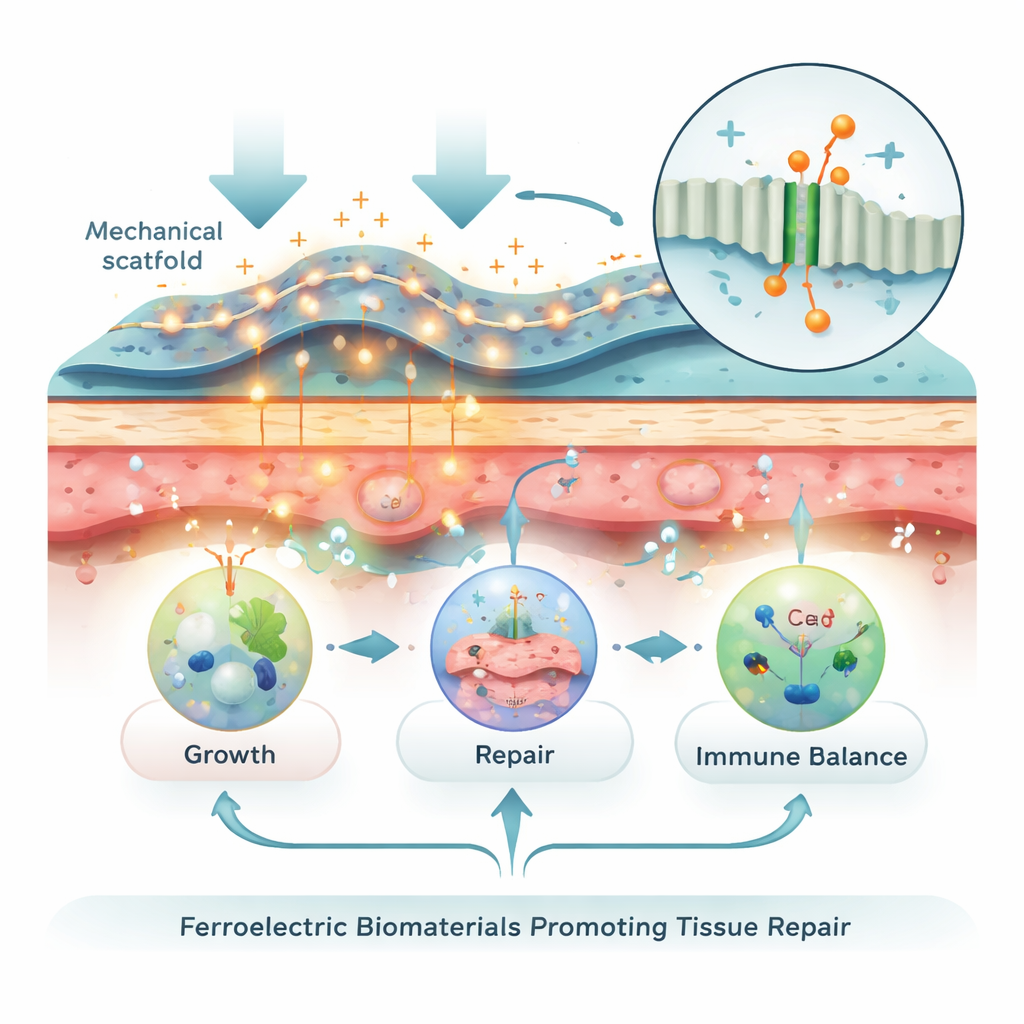
Every living tissue carries subtle electrical patterns. Nerves fire with voltage pulses, bones generate small charges when we walk, and skin forms natural electric fields around wounds. These signals guide cells—telling them when to move, divide, and specialize. Injury or chronic inflammation can scramble this electrical landscape, slowing or misdirecting repair. The review explains how flexible ferroelectric biomaterials are designed to restore or enhance these signals. They respond to bending, stretching, or temperature changes by producing tiny, localized voltages, essentially translating mechanical motion into the body’s own electrical “healing cues.”
What these smart materials are made of
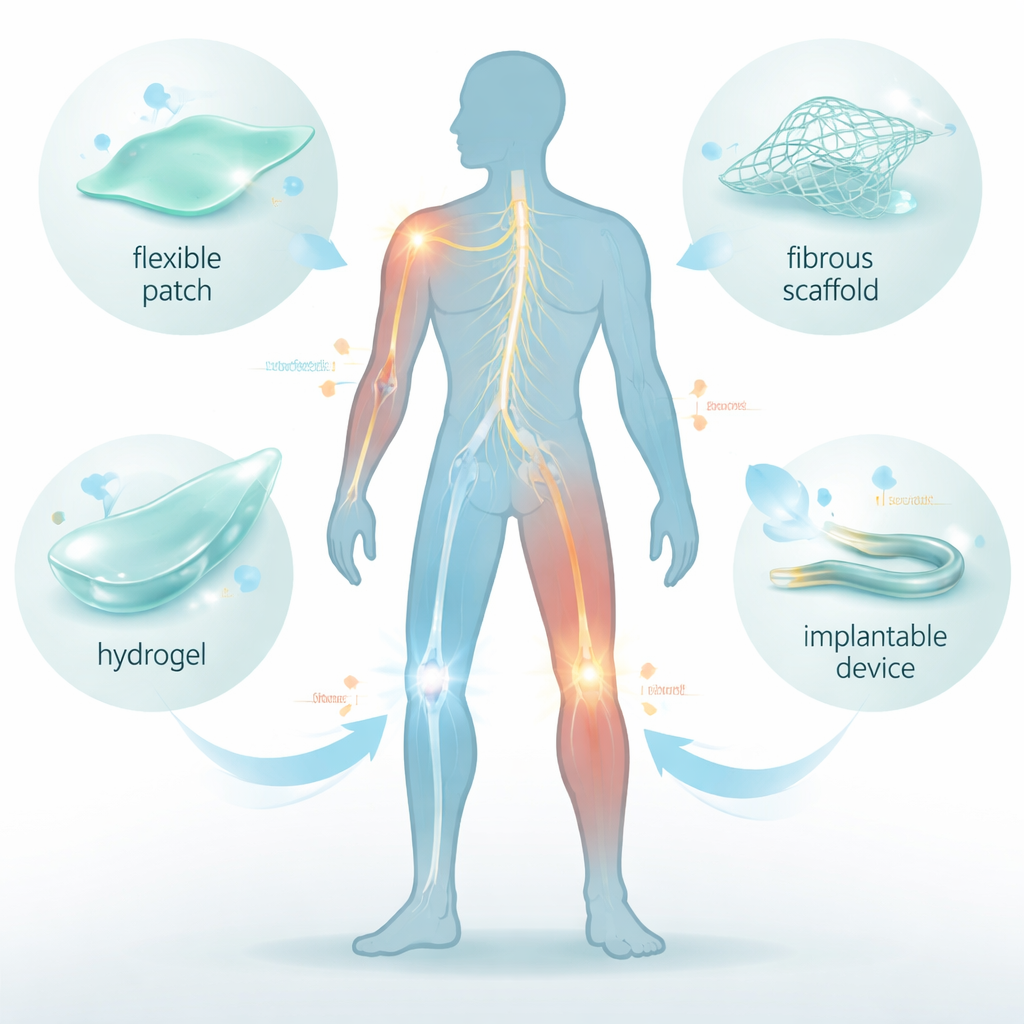
The authors focus on several families of ferroelectric materials that can be made soft and body-friendly. Polymers such as PVDF, its copolymer P(VDF-TrFE), and PLLA are plastics that, when processed correctly, behave like miniature generators: moving them creates charge. Ceramic particles such as barium titanate (BaTiO₃), bismuth ferrite (BiFeO₃), and potassium sodium niobate (KNN) offer strong electrical response but are brittle on their own, so they are blended into flexible polymers. By tuning crystal structure, fiber alignment, and porosity, researchers can make thin films, nanofiber meshes, 3D-printed scaffolds, and injectable hydrogels that conform to curved body surfaces while producing biologically relevant levels of electrical stimulation.
How movement-driven signals talk to cells
When these materials are pressed, stretched, or pulsed with ultrasound, they deliver tiny electrical pulses to nearby cells. Inside the cell membrane are ion channels that open in response to electrical or mechanical cues, allowing calcium ions to rush in. That brief calcium surge acts as a master switch, turning on networks that control cell survival, migration, growth, and specialization into bone, cartilage, nerve, or muscle cells. Electrical cues also rearrange surface receptors, influence how cells grip their surroundings, reshape energy use in mitochondria, and even steer immune cells away from prolonged inflammation toward pro-healing behavior. In this way, a simple mechanical step—such as walking after surgery—can, through these materials, be converted into meaningful biological instructions.
Real-world uses in bone, nerves, skin, and more
The review surveys rapid progress across many tissues. In bone and cartilage, ferroelectric scaffolds and hydrogels placed in defects generate small voltages under normal joint motion or focused ultrasound, boosting bone-forming gene activity and encouraging high-quality cartilage regrowth. In peripheral nerves, flexible conduits made from piezoelectric fibers guide regrowing axons and, when activated by movement or sound waves, provide gentle, continuous stimulation comparable to nerve grafts. For skin, self-powered dressings and 3D-printed patches amplify the body’s own wound currents, accelerating closure, blood vessel growth, infection control, and even reducing scar formation. Similar strategies help muscle fibers align and mature and support stronger tendon-to-bone attachment after rotator cuff or ligament injuries.
From lab promise to everyday therapies
Despite the excitement, the authors emphasize that most of these technologies are still in early animal or lab studies. Making these complex, layered materials reliably at scale, ensuring they survive sterilization and years inside the body, and matching their degradation to the pace of tissue healing are open challenges. Some widely used polymers, like PVDF, hardly break down in the body at all, raising questions about their long-term fate. Future work will need better control systems—potentially using AI—to adjust stimulation in real time, as well as new ferroelectric materials that are both effective and truly biodegradable. If these hurdles can be overcome, flexible ferroelectric biomaterials could enable implants, bandages, and soft electronic “skins” that quietly harvest our own motions to power precise, personalized repair of skin, nerves, muscles, and the entire musculoskeletal system.
Citation: Sheng, N., Wang, Y., Luo, X. et al. Flexible ferroelectric biomaterials for skin, neural, and musculoskeletal tissue repair. npj Flex Electron 10, 29 (2026). https://doi.org/10.1038/s41528-026-00532-w
Keywords: flexible biomaterials, electrical stimulation, tissue regeneration, piezoelectric polymers, wound healing