Clear Sky Science · en
Variants in MTNAP1 underlie a neurodegenerative disorder by impairing mitochondrial stability
Why this story matters for brain health
Many families face the heartbreak of watching a child slowly lose developmental skills with no clear diagnosis. This study uncovers a new genetic cause of such a condition, tracing it all the way from a single faulty gene to damaged “power stations” inside brain cells and, ultimately, to shrinking brain tissue. Understanding this chain of events not only brings answers for affected families, it also sharpens our broader picture of how fragile the brain’s energy systems really are.
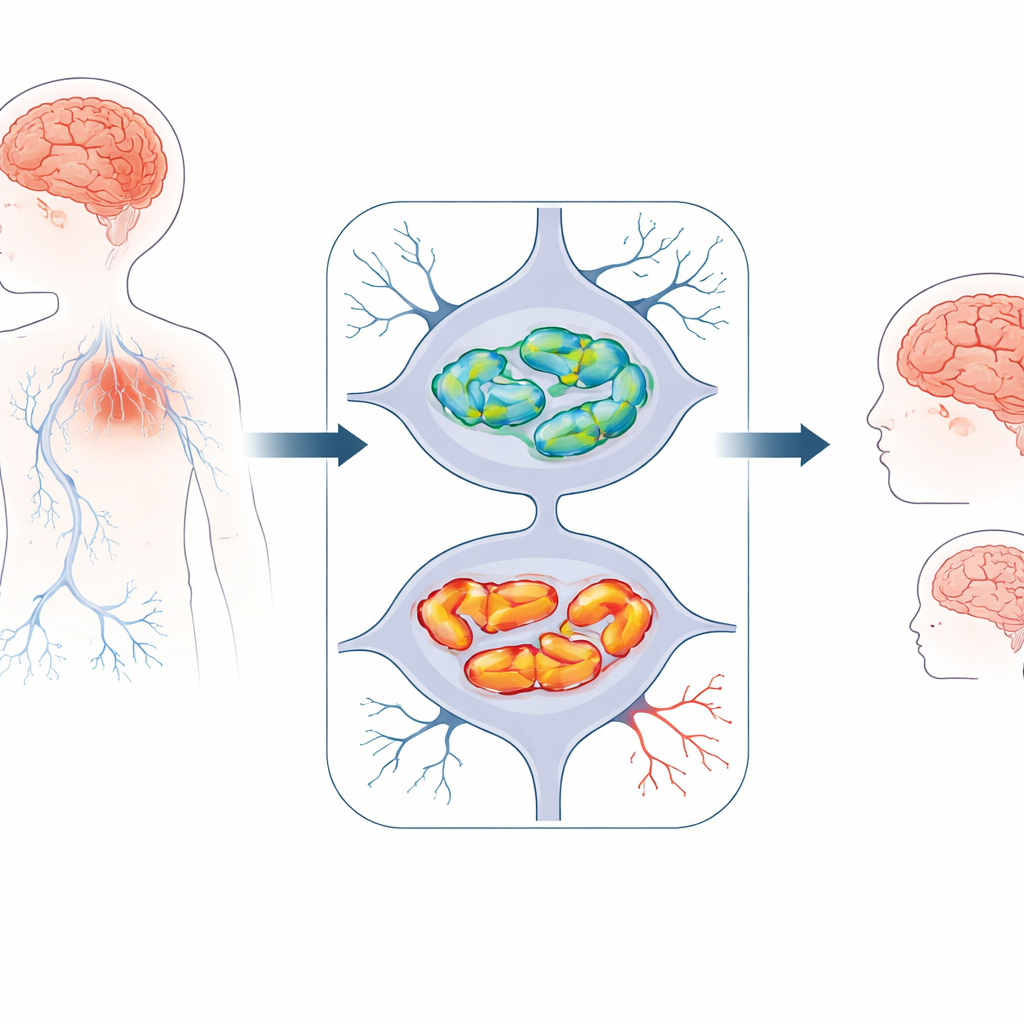
A newly recognized childhood brain disorder
The researchers studied three children from two unrelated families who all showed early developmental problems. They were small for their age, learned to sit, walk, and speak later than expected, and then gradually lost some of these abilities. All developed movement difficulties such as unsteady gait, muscle stiffness or low muscle tone, and seizures. Brain scans painted a consistent picture: the tissue of both the large brain (cerebrum) and the “little brain” at the back (cerebellum) was thinning over time, and a key connecting bridge of nerve fibers, the corpus callosum, was unusually slender. These features pointed to a progressive loss of nerve cells rather than a one-time injury at birth.
A tiny gene with big consequences
To look for an inherited cause, the team sequenced all protein-coding genes in the affected children and their parents. They homed in on a gene called MTNAP1, which helps organize the DNA inside mitochondria, the cell’s energy factories. Each child carried two faulty copies of MTNAP1, one from each healthy, carrier parent. In two siblings, a single “letter” change in the gene swapped one building block of the protein for another, subtly distorting its shape. In the third child, an early stop signal in the gene likely prevented the protein from being made at all. These changes were not seen in large population databases, strengthening the case that they are rare, harmful variants rather than harmless quirks.
Power stations under stress
Next, the scientists examined skin cells taken from the children and compared them with cells from healthy individuals. Under the microscope, normal cells showed long, thread-like mitochondria forming a connected network, while the children’s cells contained short, broken, and clumped mitochondria. When the researchers experimentally reduced MTNAP1 levels in a human nerve-like cell line, they saw the same breakup of the mitochondrial network, confirming that loss of this protein alone can disturb their structure. Measurements of mitochondrial activity revealed that key steps in energy production were weakened, and the cells produced excess reactive oxygen species—damaging oxygen by-products that act like molecular rust. The stressed cells stopped dividing properly, accumulated in a resting phase, and switched on markers of premature aging.
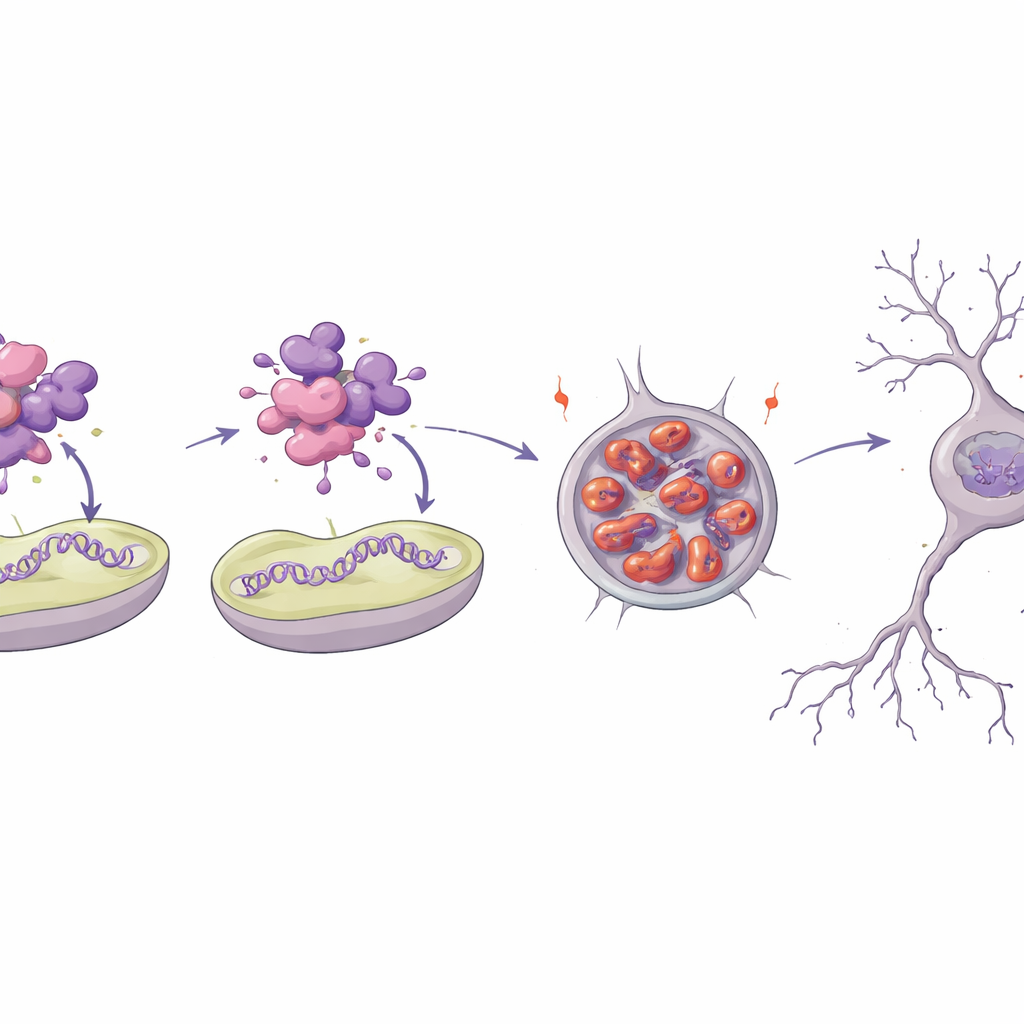
How a single change unravels a vital protein
To understand why one of the variants is so disruptive, the team modeled the 3D structure of the MTNAP1 protein and recreated it in the lab. The substituted building block sits in a tightly packed helical region that normally helps the protein interact with mitochondrial DNA and the inner membrane. Computer simulations and biophysical tests showed that the mutant protein is less stable, loses much of its ordered structure, and readily forms clumps. In test-tube experiments, the normal protein bound strongly to short pieces of mitochondrial DNA and to artificial membrane surfaces, while the mutant barely interacted and instead assembled into amyloid-like aggregates. When introduced into nerve-like cells, the mutant accumulated in large perinuclear clumps over time, a sign of protein quality-control systems being overwhelmed.
From damaged mitochondria to a failing brain
Putting the pieces together, the study outlines a stepwise model: faulty MTNAP1 weakens the scaffolding that helps organize mitochondrial DNA and anchor it to the inner membrane; this destabilizes mitochondria, causing them to fragment and lose their ability to generate energy efficiently; rising oxidative stress and cellular “aged before its time” signals then make nerve cells especially vulnerable, because they have high and constant energy demands and limited capacity to renew themselves. In the developing brain, this slow, ongoing energy crisis translates into stalled milestones, loss of learned skills, and gradual shrinkage of key brain regions. While more patients and animal studies are needed to fully map out the syndrome, this work firmly positions MTNAP1 as a crucial guardian of mitochondrial stability and highlights mitochondrial DNA organization as a central pillar of healthy brain development.
Citation: Kumar, A., Saha, S., Nasir, N. et al. Variants in MTNAP1 underlie a neurodegenerative disorder by impairing mitochondrial stability. npj Genom. Med. 11, 19 (2026). https://doi.org/10.1038/s41525-026-00554-3
Keywords: mitochondria, neurodegeneration, pediatric genetics, mitochondrial DNA, protein misfolding