Clear Sky Science · en
Atomistic understanding of hydrogen bubble-induced embrittlement in tungsten enabled by machine learning molecular dynamics
Why tiny bubbles can break tough metals
If you have ever seen a metal part crack unexpectedly, hydrogen might have been one of the hidden culprits. In extreme technologies such as fusion reactors, metal walls are bombarded with hydrogen, which can sneak inside and weaken them from within. This study uses advanced computer simulations to watch, atom by atom, how hydrogen gathers into bubbles inside tungsten—a leading candidate material for fusion reactors—and how those bubbles can trigger sudden, brittle failure. Understanding this hidden process is vital for building safer, longer-lasting machines in harsh environments. 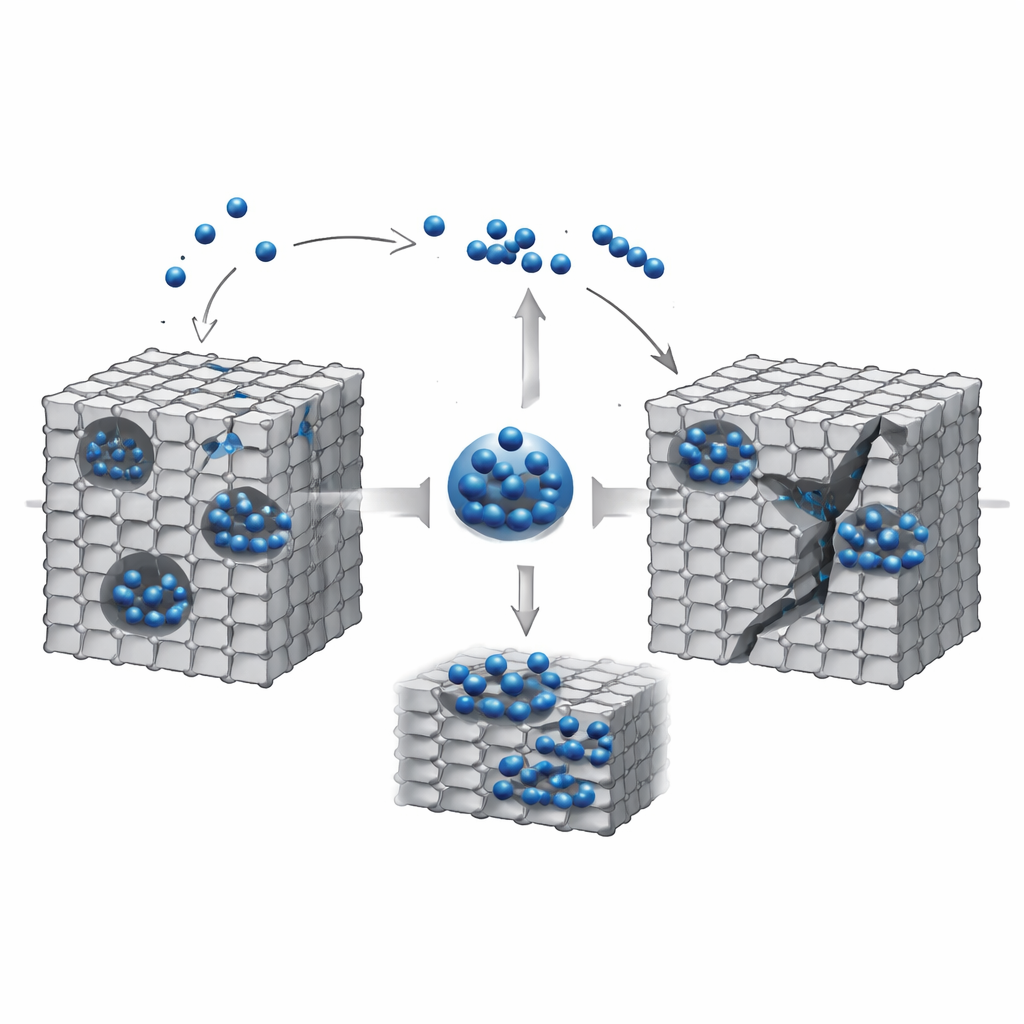
Following hydrogen atoms one by one
To track hydrogen inside a solid metal, scientists need tools that are both accurate and fast. Traditional quantum calculations are extremely precise but too slow to handle the millions of atoms involved in real bubbles, while simpler models are fast but often unreliable. The authors bridge this gap by training a machine-learning model, called NEP-WH, to mimic quantum-level physics for tungsten and hydrogen. They feed it a rich training set of atomic configurations—perfect crystals, defects, liquid-like states, surfaces, cracks, and voids—so that the model can recognize many environments. Tests show that NEP-WH closely matches quantum results for basic properties of tungsten, how hydrogen dissolves and moves in it, and how hydrogen molecules behave under very high pressure.
Seeing how bubbles form inside the metal
Armed with this new model, the researchers run large-scale molecular dynamics simulations to watch hydrogen gather inside tiny empty pockets—nanovoids—inside tungsten. As hydrogen enters, it first forms molecules that pack the center of the void, building up pressures of tens of billions of pascals, comparable to conditions deep inside planets. At the same time, some molecules break apart at the void surface, leaving individual hydrogen atoms that stick to the surrounding metal. The system eventually settles into a steady state where bubble pressure depends on the size of the void and follows a simple surface-tension-like rule: smaller voids reach higher pressures. This provides numerical support for long-standing theoretical ideas about how internal gas bubbles behave in solids.
Flat hydrogen sheets and hidden weak spots
Once the bubble pressure stabilizes, the hydrogen story does not end. Extra hydrogen atoms continue to seep out of the bubble and into the nearby metal, but not evenly. Instead, they gather into thin, sheet-like clusters along specific crystal planes, known as {100} planes, extending outward from the void surface. Within these sheets, the local metal structure is pushed toward more closely packed arrangements, and at the crossings of several sheets, small regions with yet another packing pattern appear. These hydrogen-rich features effectively carve invisible planes of weakness into the metal, concentrating stress along preferred directions. 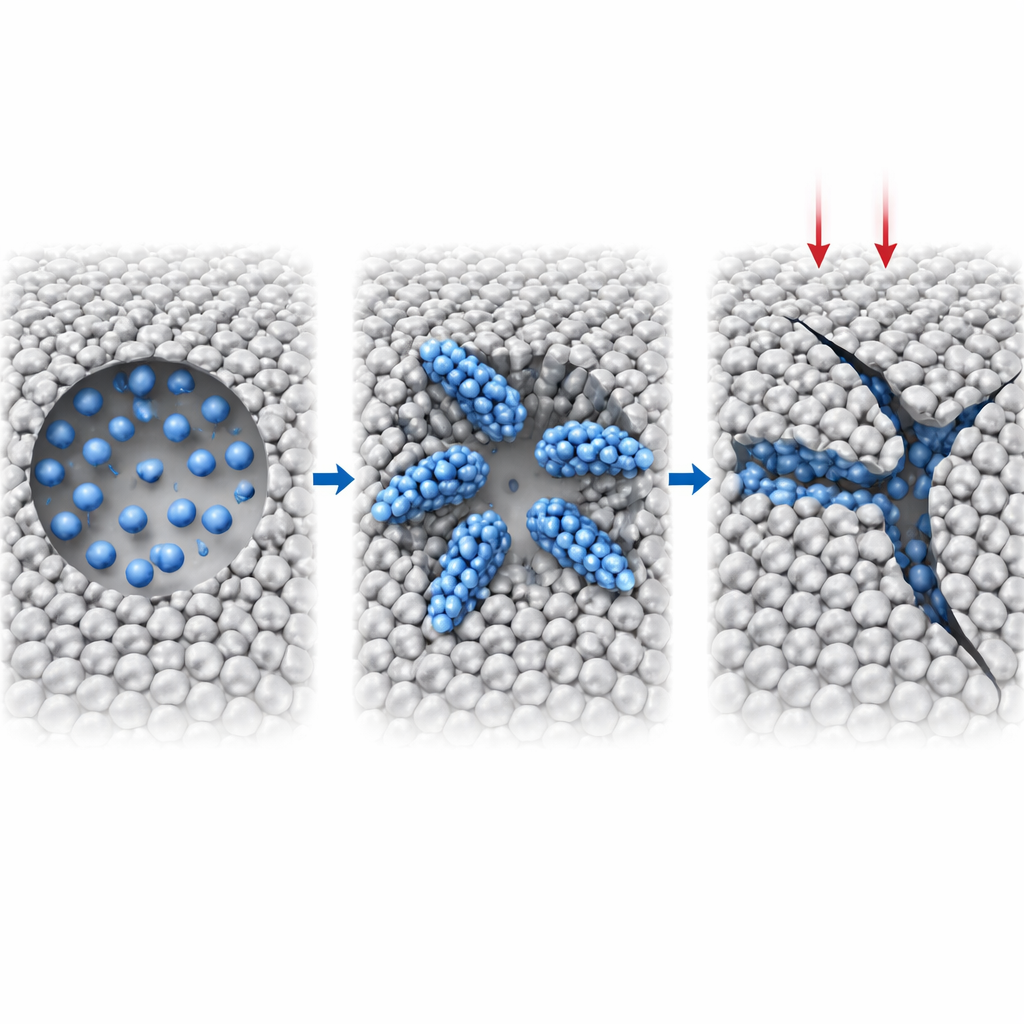
From smooth stretching to sudden snapping
To see how these hidden structures affect strength, the team stretches their simulated tungsten samples along one direction. A void with no hydrogen deforms in a relatively gentle, ductile way: dislocations—line-like defects—move and multiply, absorbing energy before the material finally breaks. Adding hydrogen changes the outcome dramatically. Even at modest hydrogen levels, cracks start from the bubble and run straight along the hydrogen-enriched planes, with far less dislocation activity. As hydrogen content increases further, pre-existing sheets and wedge-shaped hydrogen-rich regions guide the crack path, lowering the metal’s strength and turning its behavior from pliable to brittle. Instead of slowly yielding, the material snaps along sharp, flat fracture surfaces.
What this means for future extreme machines
For a non-specialist, the key message is that hydrogen does not just “make metal weaker” in a vague way. It organizes itself inside tungsten into pressurized bubbles and flat, hidden layers that act like built-in fault lines. The new machine-learning model lets researchers see this in unprecedented detail and at realistic scales, tying atomic movements to macroscopic cracking. These insights help explain puzzling surface blisters and internal cracks seen in fusion experiments, and they provide a roadmap for designing metals and operating conditions that can better resist hydrogen damage.
Citation: Bao, Y., Song, K., Liu, J. et al. Atomistic understanding of hydrogen bubble-induced embrittlement in tungsten enabled by machine learning molecular dynamics. npj Comput Mater 12, 108 (2026). https://doi.org/10.1038/s41524-026-01986-2
Keywords: hydrogen embrittlement, tungsten, nanovoids, machine learning potentials, fusion materials