Clear Sky Science · en
Probing multi-dimensional composition spaces in search of strong metallic alloys
Why Stronger Metals Matter
From jet engines to fusion reactors, the hottest parts of our technology are pushed close to their limits. Ordinary metals soften and fail when heated, so researchers are searching for new alloys that can stay strong at extreme temperatures. This study uses supercomputer simulations as a kind of virtual materials laboratory to explore a huge space of possible metal mixtures and to uncover why some combinations become surprisingly tough. The answers could guide the design of next‑generation structural materials without having to melt and test every candidate in the real world.
Mixing Metals Like a Powerful Cocktail
The work focuses on a new class of materials called refractory complex concentrated alloys—mixtures of several heavy, heat‑resistant metals such as iron, niobium, molybdenum, tantalum, and tungsten. Instead of tweaking one main ingredient with small amounts of others, these alloys mix elements in comparable proportions, opening an enormous design space. The team asked a simple question: can certain mixtures become stronger than any of their pure ingredients, a phenomenon often nicknamed the “cocktail effect”? Using detailed atomic‑scale simulations, they compressed virtual crystals and measured how much stress was needed to keep them flowing plastically, a measure of their practical strength at high temperature. 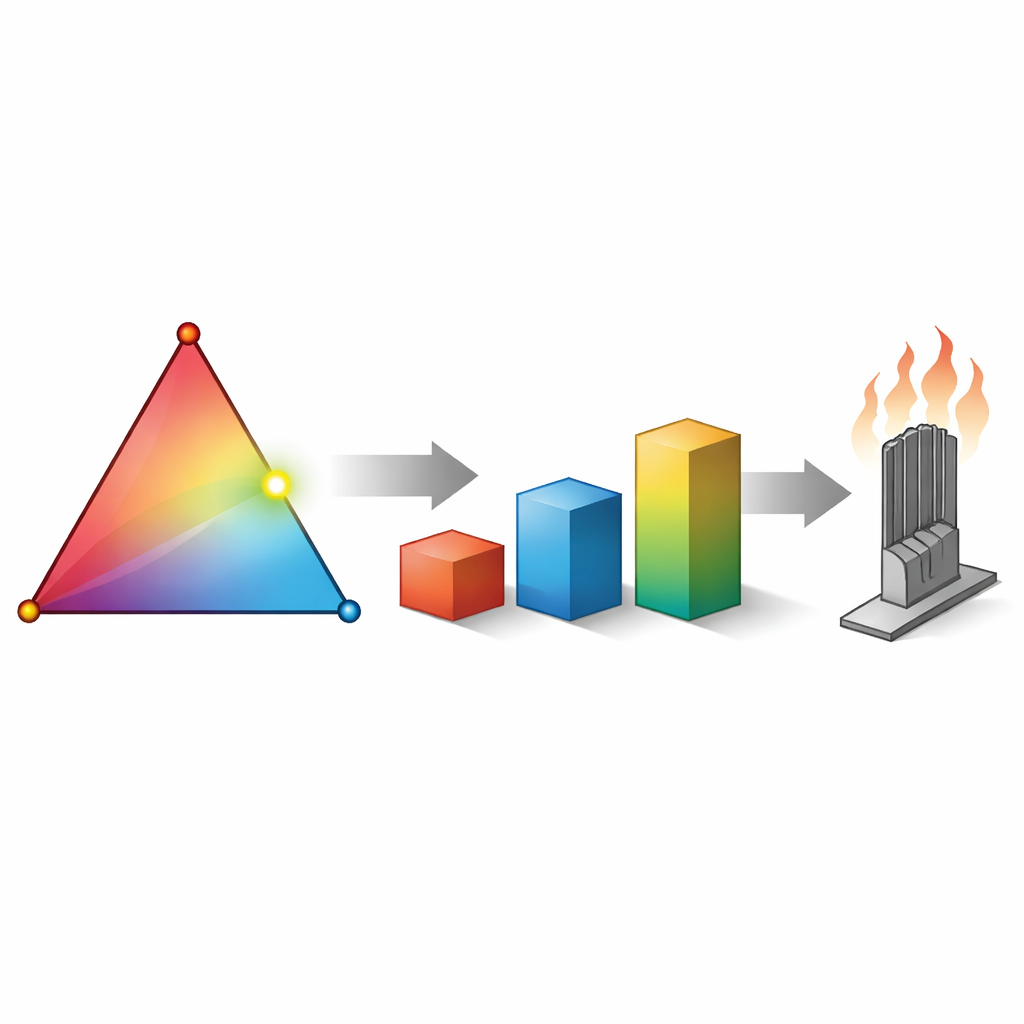
Letting a Learning Algorithm Hunt for the Best Mix
Because each simulation involves tens of millions of atoms and demands thousands of hours of supercomputer time, the authors could not simply test every possible recipe. Instead, they coupled their simulations to a statistical machine‑learning method called Gaussian process regression. After each batch of virtual experiments, this helper model guessed which new composition was most likely to be stronger and suggested it for the next simulation, gradually homing in on the best candidates. In one ternary family that combines iron, tantalum, and tungsten, this strategy rapidly converged on a mixture lying along a “binary edge” between iron and tungsten, rather than at the more intuitively appealing equal‑parts blend of all three. Similar searches in a four‑element family centered on niobium, molybdenum, tantalum, and tungsten pointed toward tungsten‑rich alloys and even pure tungsten itself as top performers, with little extra benefit from further mixing.
Peering Inside to See What Carries the Load
The simulations do more than just output strength numbers; they track every atom and every dislocation—tiny line‑like defects that carry plastic deformation in crystals. By examining the evolving network of these defects, the researchers could test competing theories of how complex alloys harden. One influential idea holds that so‑called edge dislocations, pushed through a random landscape of atomic sizes, dominate the strengthening. The virtual “micrographs” from this study tell a different story: screw‑type dislocations, which are inherently sluggish in body‑centered cubic metals, remain overwhelmingly prevalent in both pure tungsten and the strong alloys. As they move through the chemically disordered lattice, they repeatedly kink, tangle, and leave behind clouds of vacancies and interstitials, visible in the simulations as dense fields of debris.
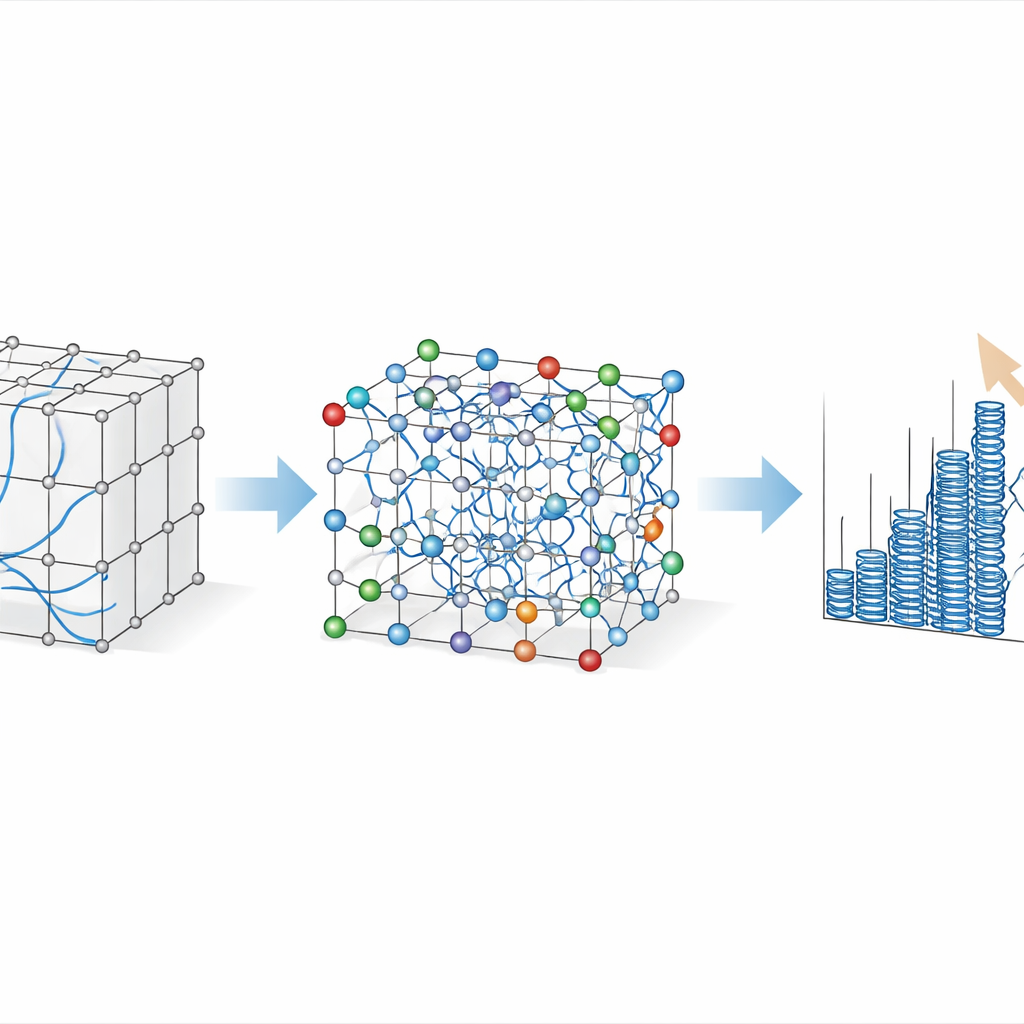
When Crowded Defects Do the Hard Work
These tangled dislocation networks reveal that collective interactions, not just the resistance felt by a single moving defect, are key to the alloys’ strength. The authors show that at large strains the material’s flow stress closely follows a classic relationship known as Taylor hardening, in which strength scales with the square root of the total dislocation density. In other words, as deformation proceeds, multiplying and intersecting dislocations form a forest that obstructs further motion. This pattern holds across pure body‑centered cubic metals and across all the complex alloys studied, with a single parameter describing the network’s effectiveness that matches values measured in experiments on simpler metals. Chemical disorder still matters: it raises both the intrinsic resistance to dislocation glide and the rate at which new dislocations are generated, but the dominant contribution at high strain comes from the crowded network itself rather than from isolated obstacles.
What This Means for Designing Future Alloys
For non‑specialists, the main takeaway is that making metals stronger at high temperatures is not simply a matter of adding more elements or maximizing randomness. The most robust mixtures the team found sit on the edges of the composition space or near the strongest pure metal, and their strength at large deformation is governed by how efficiently they build up and entangle dislocations. By marrying large‑scale atomistic simulations with smart, iterative search algorithms, the authors demonstrate a powerful route to explore vast composition spaces and to pinpoint the atomic‑level mechanisms that matter most. This approach will not instantly deliver ready‑to‑use superalloys, but it offers a clear roadmap: focus on how alloy chemistry controls both the ease of dislocation motion and the buildup of dislocation networks, because together these effects set the ultimate strength and durability of metals in extreme environments.
Citation: Zhou, X., Marian, J., Zhou, F. et al. Probing multi-dimensional composition spaces in search of strong metallic alloys. npj Comput Mater 12, 120 (2026). https://doi.org/10.1038/s41524-026-01975-5
Keywords: refractory alloys, high temperature strength, dislocations, molecular dynamics, materials design