Clear Sky Science · en
First-principles computation of dislocation structures and stress-driven phase transformations in layered oxides for Na-ion batteries
Why tiny defects matter for future batteries
As the world looks beyond lithium toward cheaper, more abundant sodium-ion batteries, a hidden world inside the cathode materials becomes crucial: tiny crystal defects called dislocations. These line-like irregularities, only atoms wide, help the material flex as sodium ions move in and out—but they can also trigger the structural damage that shortens battery life. This paper uses quantum‑level computer simulations to uncover how dislocations form, move, and drive phase changes in layered sodium cathodes, offering guidance for designing longer‑lasting, more robust batteries.
Stacked atomic layers that must keep their shape
Many promising sodium-ion cathodes are built from stacks of flat atomic sheets. Sodium ions sit between transition‑metal–oxygen layers in an orderly “O3” arrangement when fully charged, but charging and discharging repeatedly push the structure toward a different stacking pattern, called “P3.” These shifts in how the layers line up—the stacking sequence—can be reversible and harmless, or they can trigger collapse, cracking, and loss of capacity. The authors focus on a family of layered oxides, Na(TM)O₂ with TM = Ti, Cr, Mn, Fe, Co, or Ni, and ask: how easy is it for these materials to rearrange their stacking, and what role do dislocations play when that happens? 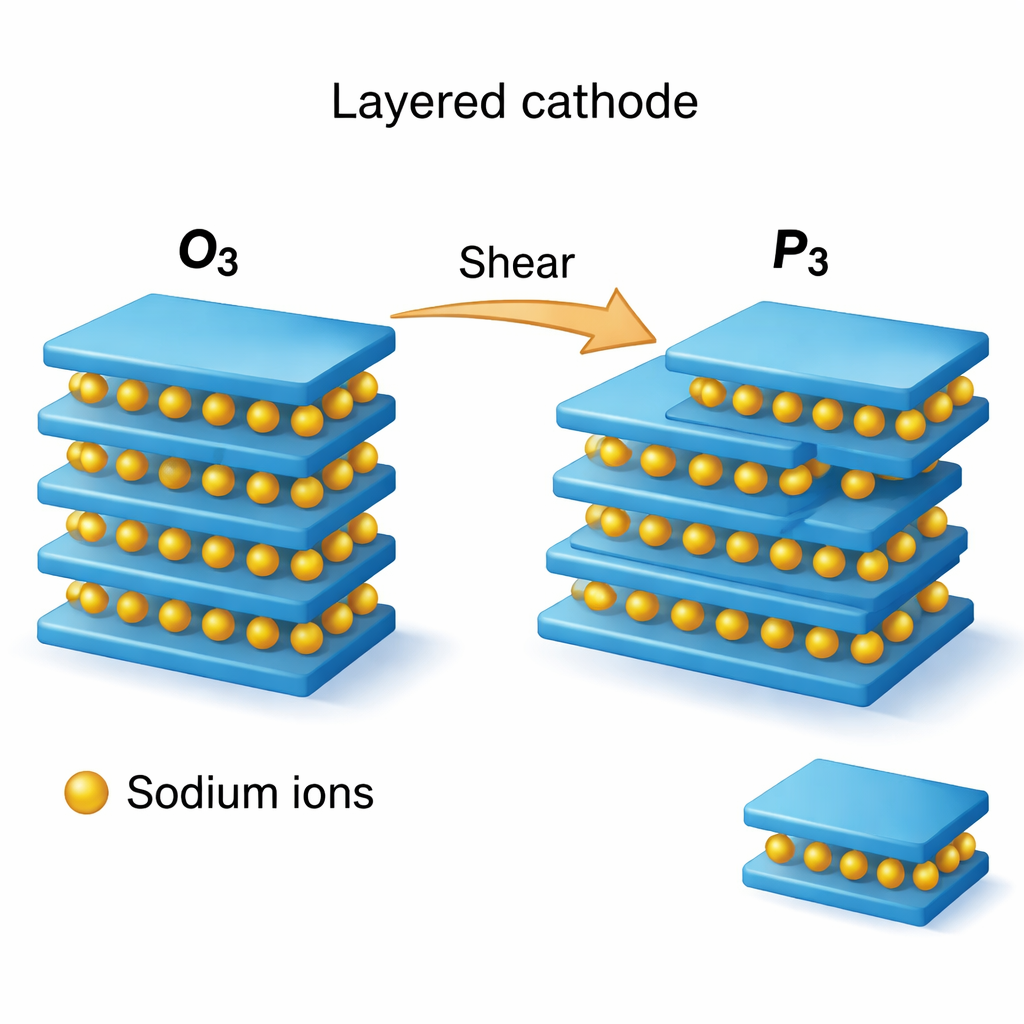
Mapping how layers prefer to slide
To answer this, the researchers first calculate so‑called generalized stacking fault energy surfaces. In simple terms, they take two halves of the crystal, slide one half over the other along different directions, and compute how much energy each shift costs. Low‑energy paths on this map reveal how the layers prefer to glide, and whether intermediate “faulted” states—local rearrangements of the stacking—are likely to form. Across all the compounds studied, they find that a P3‑like faulted state is possible, but it is especially favored in cobalt‑ and nickel‑based materials, which show deep energy minima for this configuration. In contrast, a more drastic O1‑type stacking does not appear as a stable state under the conditions they model, suggesting that the gentler O3↔P3 changes are inherently more accessible.
What dislocations look like inside these cathodes
Real crystals do not shear as perfectly rigid blocks; they deform through the motion of dislocations. Using a semi‑discrete Peierls–Nabarro model informed by their quantum‑mechanical data, the authors reconstruct the internal structure—or “core”—of both edge and screw dislocations on the key slip plane parallel to the layers. They find that dislocation cores are very narrow, only a few nanometers across, confirming that these materials are mechanically stiff. Edge dislocations tend to split into two “partial” dislocations separated by a thin strip that locally has P3‑type stacking, especially in Co‑ and Ni‑rich oxides where the P3 state is energetically favored. Screw dislocations usually remain more compact, but in some compositions (again, notably Co and Ni) they can also split and create narrow P3‑like regions.
How easily defects move under battery stresses
Next, the study estimates the Peierls stress—the minimum shear stress needed to start a dislocation moving through the lattice. This quantity acts like a microscopic yield strength for individual defects. For all the examined materials, the required stresses (a few to a few tens of megapascals) fall within the range of stresses expected when sodium ions are inserted and extracted during cycling. That means dislocation motion is not just possible but likely under realistic operating conditions. The computations also show that some structures, particularly monoclinic variants of Mn and Ni oxides, offer higher resistance to certain kinds of dislocation motion because their preferred low‑energy glide paths are more restricted. 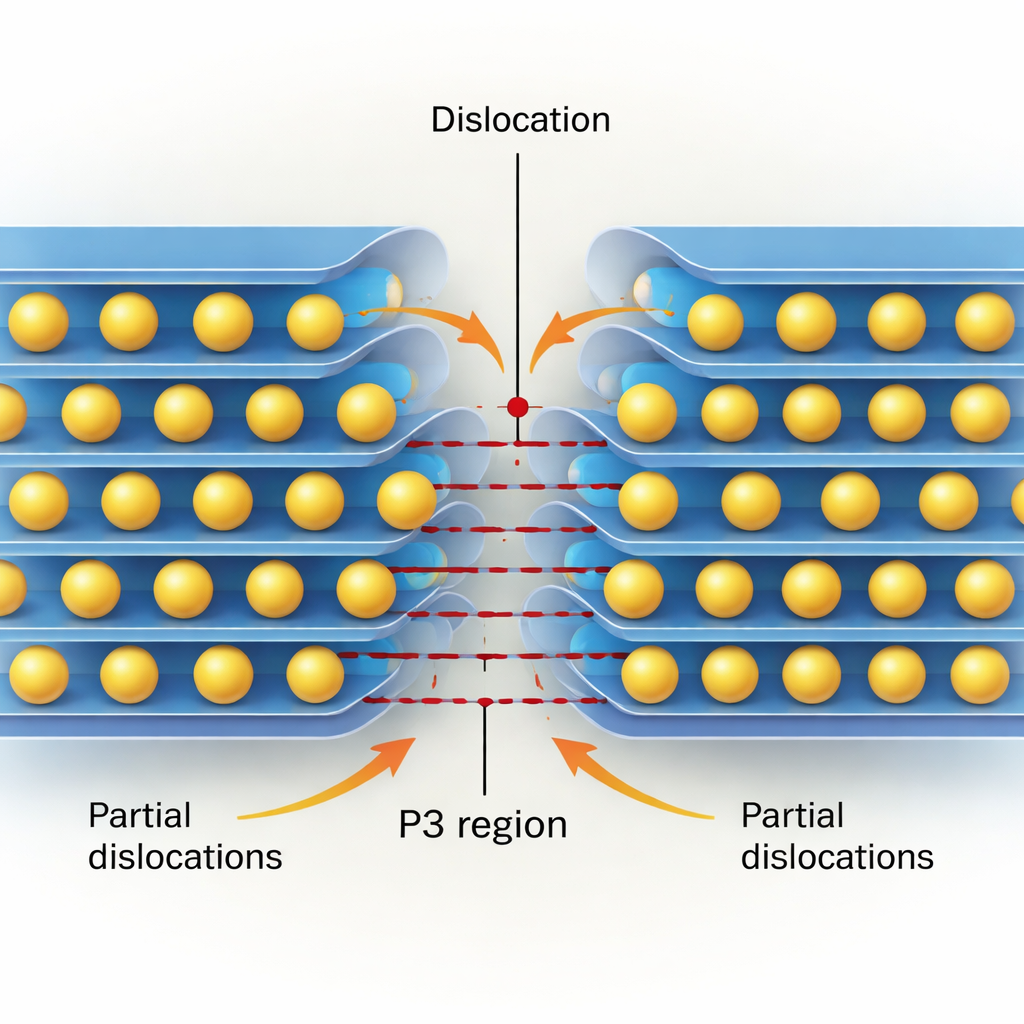
Dislocations as engines of phase change
Bringing these pieces together, the authors propose a picture in which dislocations actively drive the O3→P3 phase transformation. In a fully sodiated cathode, existing or newly formed dislocations can split into partials, seeding tiny P3‑like regions along their line. As sodium is removed, the local energy landscape changes so that the P3 configuration becomes increasingly stable. The P3 strip between the partial dislocations then widens, and sodium ions hop into the new prismatic sites, allowing the P3 region to grow and sweep through the particle. Over many cycles, the accumulation and motion of these defects can also contribute to microcracks and irreversible phases, linking atom‑scale processes directly to battery degradation.
Design rules for tougher sodium batteries
To a non‑specialist, the key message is that the lifetime of sodium‑ion batteries depends not only on which elements are chosen, but also on how their atomic layers prefer to slide and how easily dislocations can move. By mapping these behaviors from first principles, the study provides design cues: chemistries that keep stacking‑fault energies shallow and control dislocation motion can favor smooth, reversible O3↔P3 transitions and resist cracking. In practical terms, that means engineers can tune composition and structure to manage these tiny defects, paving the way to sodium‑ion batteries that are cheaper than today’s lithium cells yet durable enough for large‑scale energy storage.
Citation: Arcelus, O., Carrasco, J. First-principles computation of dislocation structures and stress-driven phase transformations in layered oxides for Na-ion batteries. npj Comput Mater 12, 96 (2026). https://doi.org/10.1038/s41524-026-01965-7
Keywords: sodium-ion batteries, layered cathodes, dislocations, phase transformations, materials degradation