Clear Sky Science · en
Element mapping-based Bayesian optimization framework enabling direct materials design: a case study on NASICON-type cathode materials
Smarter Shortcuts to Better Batteries
Designing new battery materials has traditionally meant years of trial and error in the lab and on the computer. This study shows how a smarter search strategy can dramatically speed up that process, using statistics and chemistry together to pinpoint promising ingredients for next‑generation sodium‑ion batteries, a lower‑cost alternative to today’s lithium‑ion cells.
Why We Need New Battery Recipes
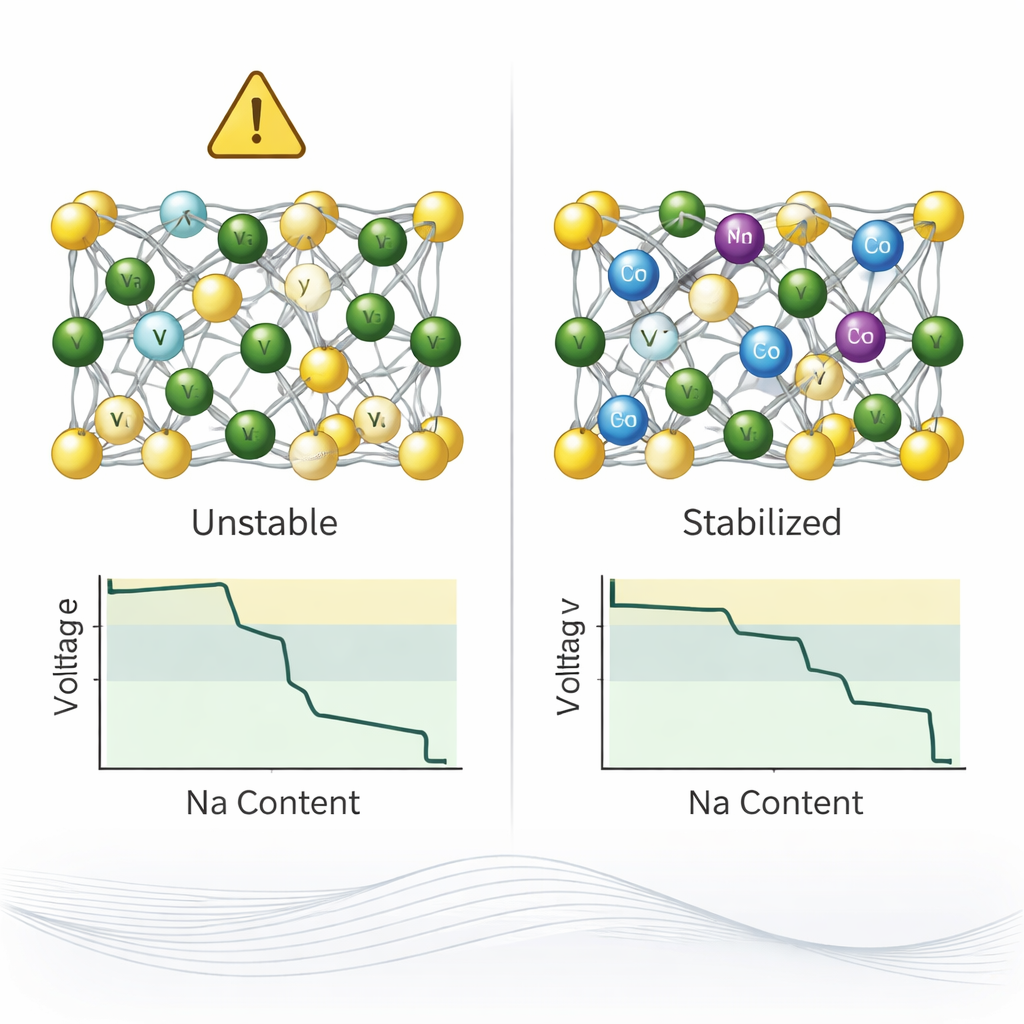
Lithium‑ion batteries power phones, laptops, and electric cars, but lithium is relatively scarce and expensive. Sodium‑ion batteries, which use common table‑salt sodium instead of lithium, are emerging as a cheaper and more sustainable option. One especially promising sodium‑based material, called NVPF, already offers fast charging and high operating voltage. However, it cannot fully use all the sodium it could, leaving valuable capacity untapped. When extra sodium is added, the material enters a "sodium‑excess" state that is thermodynamically unstable and operates outside the safe and practical voltage window used in real devices. Stabilizing this sodium‑rich state without damaging the crystal structure is a key challenge for making sodium‑ion batteries truly competitive.
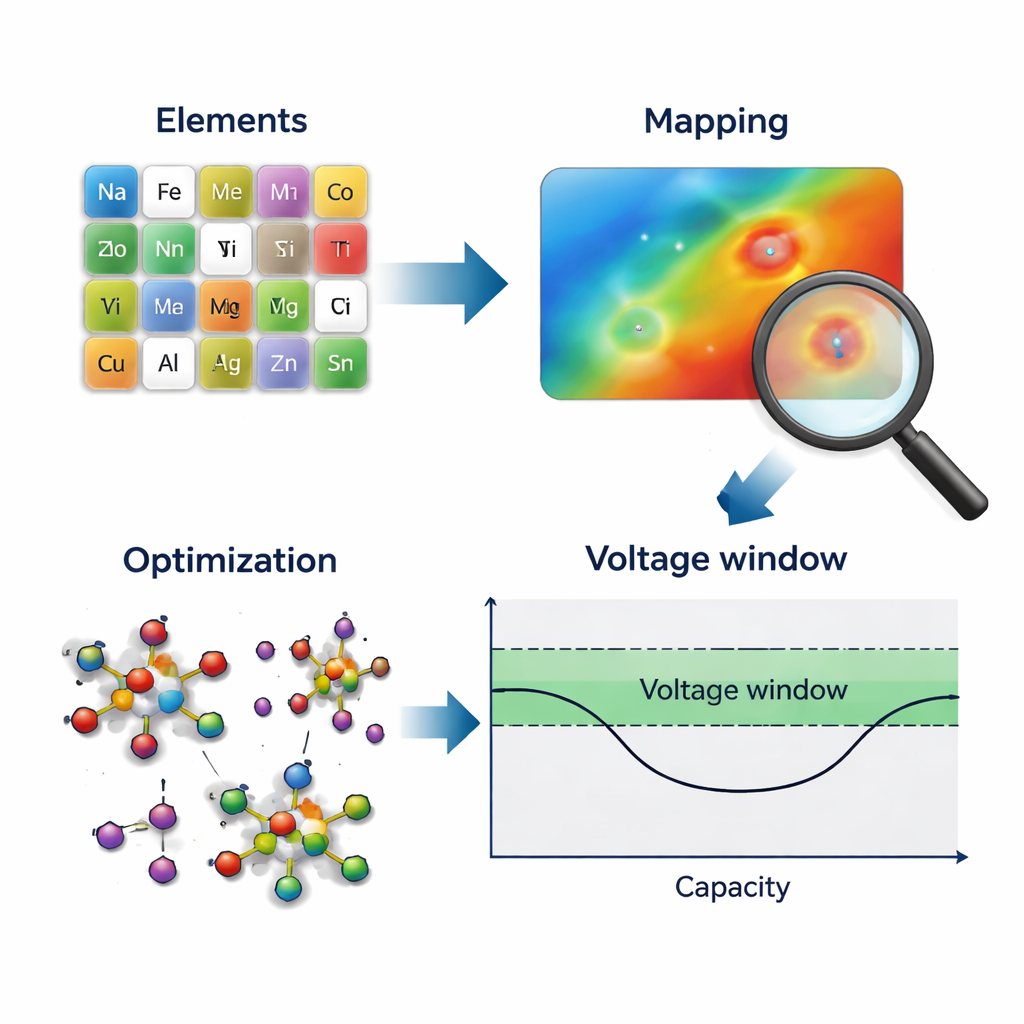
A Map for Exploring the Periodic Table
Searching for better versions of NVPF means trying many ways of swapping vanadium atoms in its structure with other metals. The number of possible element combinations quickly explodes, and testing each one with detailed quantum‑mechanical simulations is far too costly. The authors tackle this by using Bayesian optimization—a strategy that chooses the next most informative experiment based on what has already been learned. Yet standard Bayesian methods prefer smooth numerical inputs, not jumpy categorical ones like the names of elements. To bridge this gap, the team invented an "element mapping" scheme that translates each element into a continuous numerical score reflecting how it behaves when it replaces vanadium in NVPF. These scores, derived from quantum calculations, capture how easily each element accepts electrons during battery charging and discharging.
Turning Chemistry into a Smooth Landscape
With every element encoded as a continuous "unary score," the once‑discrete set of choices becomes a smooth chemical landscape that Bayesian optimization can navigate. The algorithm proposes a pair of elements to test, the researchers calculate how that combination affects the theoretical voltage profile of the material, and then a scoring function rewards cases where all battery voltages fall neatly inside the desired 2.5–4.3 volt window. This new data point updates the statistical model, which then suggests the next most promising combination. Because the unary scores are tightly linked to the actual charging behavior of the material, the resulting landscape is relatively smooth and easy to predict, allowing the optimizer to focus quickly on the most promising regions rather than wandering blindly.
Finding Better Cathodes with Fewer Guesses
Using this framework, the authors explored binary mixtures of 35 possible metals that could share vanadium’s role in the NVPF structure. Out of hundreds of theoretical combinations, their algorithm needed only 50 iterations to uncover 16 compositions whose calculated voltages all lie within the practical battery window. Many of these favorable recipes involved palladium, rhenium, tungsten, or lead in various ratios, but two combinations stood out as particularly realistic when cost, energy density, and toxicity were considered: one mixing manganese with vanadium, and another mixing cobalt with vanadium. Further electronic‑structure analysis showed that these substitutions help by accepting more electronic charge than pure vanadium does, especially in the sodium‑rich state, which helps stabilize the extra sodium instead of triggering damaging structural changes.
Beyond Trial and Error in Materials Discovery
For non‑specialists, the key takeaway is that the authors have built a kind of intelligent GPS for the periodic table. By converting each element into a chemistry‑aware number and feeding that into a Bayesian optimization loop, they can home in on high‑performance battery materials with far fewer expensive simulations than traditional grid searches or even some modern deep‑learning screeners. In their test case, this approach not only identified multiple new candidate cathode compositions for sodium‑ion batteries, but also explained why they work—because the chosen elements can safely host more electrons and sodium at useful voltages. The same strategy could be adapted to many other materials challenges, from catalysts to alloys, wherever scientists must search vast combinatorial spaces for rare, high‑performing needles in a haystack.
Citation: Park, S., Shim, Y., Hur, J. et al. Element mapping-based Bayesian optimization framework enabling direct materials design: a case study on NASICON-type cathode materials. npj Comput Mater 12, 92 (2026). https://doi.org/10.1038/s41524-026-01958-6
Keywords: sodium-ion batteries, Bayesian optimization, materials discovery, cathode design, element mapping