Clear Sky Science · en
Transcriptomic analysis to uncover the mechanism of radiosensitization of AR-positive triple-negative breast cancers with AR inhibition
Why this study matters for people facing breast cancer
Radiation therapy is a mainstay of treatment for many people with aggressive breast cancers, but some tumors are naturally harder to kill with radiation than others. This study asks a practical question with real clinical stakes: can a class of drugs that block male hormone signals, already used in prostate cancer, make a subset of triple-negative breast cancers more vulnerable to radiation—and why does this work in some cases but not others?
A tough-to-treat form of breast cancer
Triple-negative breast cancers lack three common molecular targets—estrogen receptors, progesterone receptors, and HER2—so patients often must rely on surgery, chemotherapy, and radiation alone. However, a portion of these tumors do carry the androgen receptor, a protein that responds to hormones similar to testosterone and can drive tumor growth. Earlier work showed that blocking this receptor can slow these cancers and may enhance the effects of radiation, but the details of how this happens, and which drugs and tumors benefit most, were unclear.
Testing modern hormone blockers with radiation
The researchers studied several laboratory models of triple-negative breast cancer cells that differed mainly in how much androgen receptor they carried. They focused on two newer anti-androgen medicines, apalutamide and darolutamide, and combined short exposures to these drugs with standard doses of X-ray radiation. In cells with high levels of androgen receptor, apalutamide clearly increased radiation damage, leaving fewer cells able to form new colonies. Darolutamide, in contrast, had little or no impact on radiation sensitivity in the same cells, and neither drug helped in cells that expressed very little androgen receptor. These findings suggest that not all androgen-blocking drugs act the same way in breast cancer, and that tumors must strongly rely on this receptor for the combination to work.
Watching a key protein move inside the cell
To understand what happens inside cancer cells after hormone signals or radiation, the team tracked where the androgen receptor itself was located. When cells were exposed to a synthetic androgen, the receptor moved into the nucleus, the control center where genes are switched on and off. Radiation alone did not drive the receptor out of the nucleus; in fact, it remained there after treatment, poised to influence gene activity. However, when cells received the anti-androgen drug enzalutamide before radiation, nuclear levels of the receptor dropped and more of it stayed in the cell’s outer fluid. This supports the idea that blocking nuclear entry of the receptor interferes with its ability to launch a protective gene program after radiation.
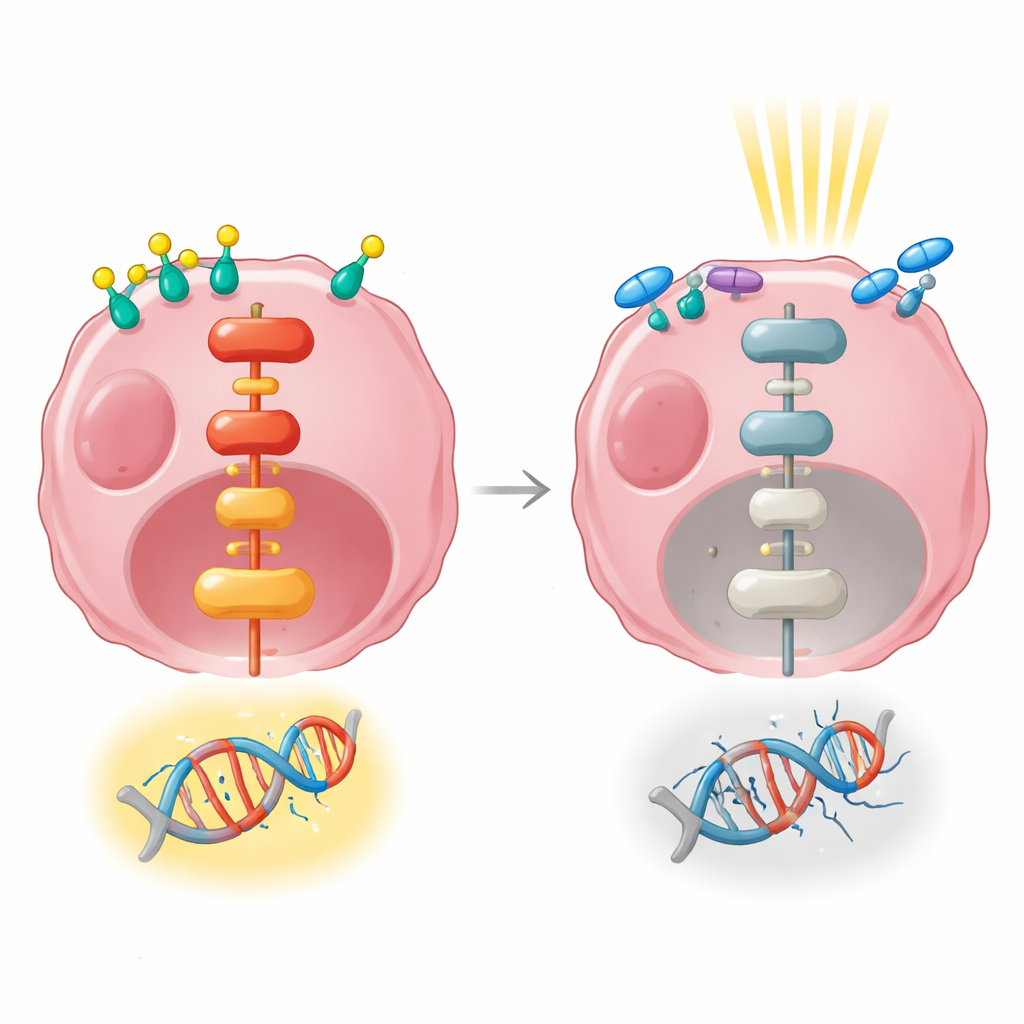
Gene signals that connect hormones to resistance
The investigators then used RNA sequencing to take a broad snapshot of which genes were turned up or down after hormone stimulation, radiation, or both. Hormone treatment alone changed the activity of hundreds of genes and strongly affected pathways that govern cell growth, attachment, and communication with the surroundings. Radiation given by itself, at an early time point, altered far fewer genes. When hormone stimulation and radiation were combined, many of the same growth-related pathways were activated, including a major signaling route known as MAPK/ERK, which relays signals from the cell surface to the nucleus. This pathway is known to help cells survive and repair DNA damage.
Probing a survival pathway that shields DNA
Because MAPK/ERK signaling kept surfacing in their analyses, the researchers tested whether boosting this pathway could undo the benefit of androgen blockade during radiation. They engineered cancer cells to overproduce an always-on version of ERK, a key component of the pathway, and then repeated the radiation plus apalutamide treatment. In this setting, apalutamide no longer made the cells more sensitive to radiation: the amplified ERK signal appeared to shield them. Together with protein measurements, these results point to MAPK/ERK signaling as a critical link between androgen receptor activity and the cell’s ability to repair radiation-induced DNA breaks.
What this means for future breast cancer care
In everyday terms, this work suggests that some triple-negative breast cancers survive radiation by using androgen-related signals to switch on an internal survival circuit that helps them mend damaged DNA. Drugs like apalutamide and enzalutamide can interrupt this circuit in tumors that strongly depend on the androgen receptor, making radiation more effective, while tumors with low receptor levels or alternative survival routes may not benefit. By mapping the gene pathways involved—especially the MAPK/ERK route—this study lays groundwork for more precise combinations of hormone blockers, pathway inhibitors, and radiation that could improve outcomes for patients with this hard-to-treat form of breast cancer.
Citation: McBean, B., Hauk, B., Michmerhuizen, A.R. et al. Transcriptomic analysis to uncover the mechanism of radiosensitization of AR-positive triple-negative breast cancers with AR inhibition. npj Breast Cancer 12, 50 (2026). https://doi.org/10.1038/s41523-026-00916-1
Keywords: triple-negative breast cancer, androgen receptor, radiation therapy, radiosensitization, MAPK ERK signaling