Clear Sky Science · en
Programmable local immunochemotherapy for triple-negative breast cancer via spatiotemporally controlled release of CpG oligodeoxynucleotides, gemcitabine, and paclitaxel
Turning a Tough Breast Cancer into a Local Target
Triple-negative breast cancer is one of the most difficult forms of breast cancer to treat because it lacks the usual molecular switches that many modern drugs latch onto. Patients often must rely on powerful chemotherapy that surges through the whole body, bringing serious side effects and, too often, disappointing long-term control. This study explores a very different idea: instead of flooding the body with drugs, can we place a tiny programmable medicine depot next to the tumor, releasing chemo and immune-boosting agents in a carefully timed sequence right where they are needed most?
A Tiny Depot with Big Ambitions
The researchers created what they call a Programmable Local Immunochemotherapy, or PLICT, platform. Think of it as a soft, injectable bead that sits beside a breast tumor and slowly dispenses treatment over weeks. The bead combines a water-loving gel that quickly releases an immune stimulant and one chemotherapy drug, gemcitabine, with microscopic plastic spheres that slowly release a second drug, paclitaxel. The immune stimulant, a short stretch of DNA called CpG, is designed to wake up local immune cells, while the paired chemotherapies directly attack tumor cells and reshape the tumor environment so immune cells can move in and do their work.
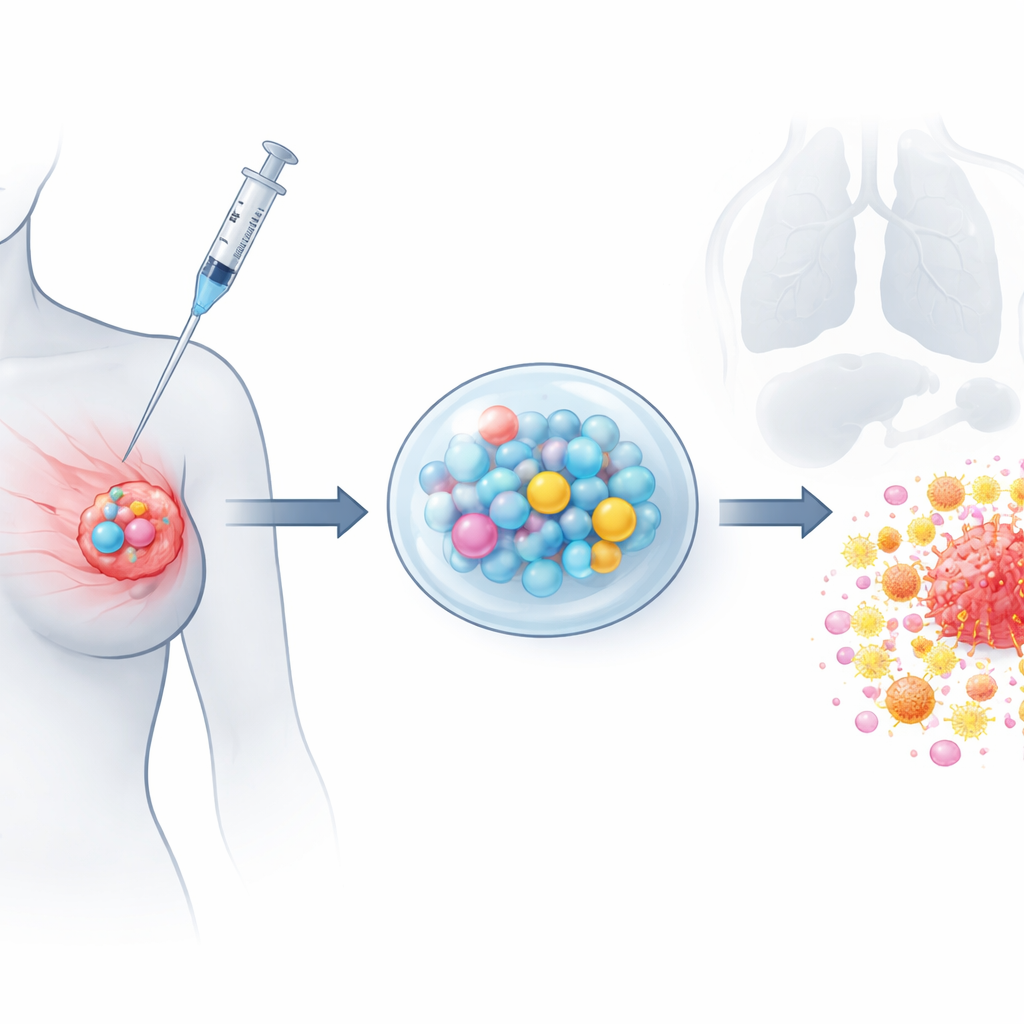
Engineering a Timed-Release Cancer Attack
To build this system, the team used a high-voltage “electrospray” method to form uniform paclitaxel-loaded microspheres from a biodegradable polymer. Imaging showed smooth, spherical particles about one-tenth the width of a human hair, and lab tests confirmed that paclitaxel leaked out steadily for at least a month without an early burst. Separately, they embedded gemcitabine and CpG into a temperature-sensitive gel that is liquid when cooled and becomes semi-solid at body temperature. Chemical fingerprinting verified that both drugs stayed intact inside their carriers. When combined, the gel provides a rapid, early dose of gemcitabine and CpG, while the microspheres offer a prolonged trickle of paclitaxel, mimicking the benefits of long-term, low-dose chemotherapy from a single local application.
Putting the Local Strategy to the Test in Mice
The team then tested PLICT in a mouse model of triple-negative breast cancer. Mice first developed small breast tumors, then received either standard injections of paclitaxel alone, paclitaxel plus gemcitabine, or a single local PLICT implant near the tumor. Over four weeks, tumors in the control group grew rapidly, those given standard chemotherapy grew more slowly, but the PLICT-treated tumors were dramatically smaller in both size and weight. Imaging of the whole animal and of removed organs showed that PLICT also cut down on the spread of cancer to distant sites, especially the lungs, more effectively than conventional drug injections. Measurements of drug levels revealed why: paclitaxel was highly concentrated inside PLICT-treated tumors but low in distant fat tissue and similar in the bloodstream, suggesting strong local trapping with fewer off-target exposures.
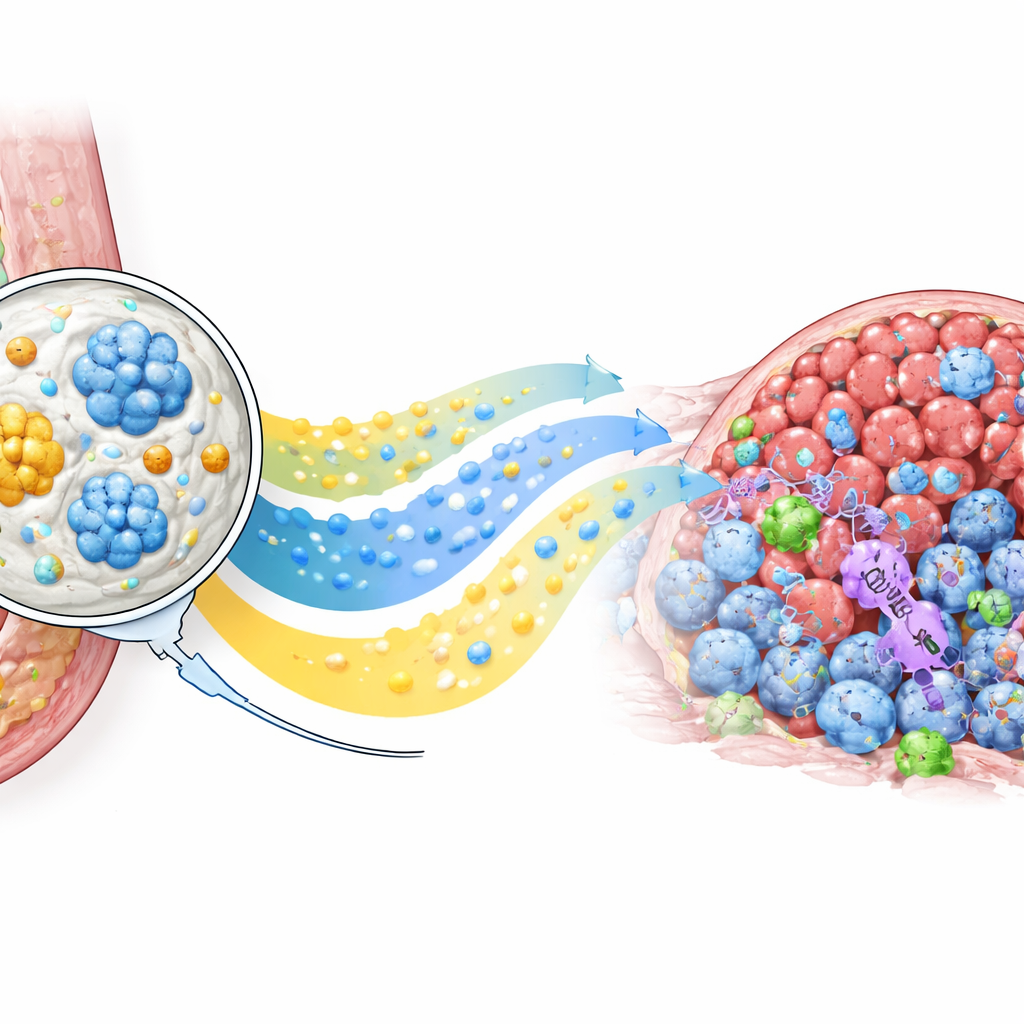
Recruiting the Body’s Own Defenders
Beyond shrinking tumors, PLICT altered the immune landscape inside them. Detailed cell analyses showed a sharp rise in cancer-killing CD8 T cells in PLICT-treated tumors compared with all other groups, along with a drop in certain regulatory T cells that normally dampen immune attacks. Tissue staining for CD69, a marker of recently activated T cells, confirmed that many of these infiltrating cells were switched on and ready to act. At the same time, standard chemotherapy caused noticeable damage to liver tissue, whereas organs from PLICT-treated mice looked nearly normal under the microscope. Together, these findings indicate that the local depot not only boosts direct drug exposure in the tumor but also sparks a more favorable, less suppressed immune response without adding extra stress to vital organs.
What This Could Mean for Future Cancer Care
For non-specialists, the message is straightforward: by turning chemotherapy and immune stimulation into a slow, precisely staged drip delivered right next to the tumor, this approach achieved stronger tumor control and fewer whole-body side effects in animals than standard drug injections. The work is still at the preclinical stage, and questions remain about how well such a system would perform in people or in different treatment settings. But the concept of an injectable, programmable “medicine depot” that quietly releases multiple agents in sequence could open new ways to treat aggressive cancers like triple-negative breast cancer—focusing the fight where it matters most while sparing the rest of the body.
Citation: Hsieh, CH., Hsu, MY., Lin, CF. et al. Programmable local immunochemotherapy for triple-negative breast cancer via spatiotemporally controlled release of CpG oligodeoxynucleotides, gemcitabine, and paclitaxel. npj Breast Cancer 12, 45 (2026). https://doi.org/10.1038/s41523-026-00910-7
Keywords: triple-negative breast cancer, local drug delivery, immunotherapy, controlled release, hydrogel microspheres