Clear Sky Science · en
Estrogen receptor β target gene expression reveals novel repressive functions in aggressive breast cancer
Why this research matters
Inflammatory breast cancer is one of the most aggressive and deadly forms of breast cancer, often spreading quickly and leaving patients with few targeted treatment options. This study explores a lesser-known hormone sensor in breast cells, called estrogen receptor beta, and shows how it can act as a natural brake on tumor spread. Understanding how this built-in brake works could open the door to new drugs that slow or prevent metastasis in patients who currently have very limited choices.
A different face of inflammatory breast cancer
Unlike many breast cancers that depend on the classic estrogen receptor alpha, inflammatory breast cancer is usually missing this molecule and therefore does not respond well to standard hormone therapies. The authors focus instead on a second estrogen receptor, estrogen receptor beta (ERβ), which they had previously linked to fewer metastases in both patients and mouse models. Earlier work suggested that ERβ slows down the cell’s internal scaffolding and movement machinery. In this study, the team set out to map, across the whole genome, how ERβ changes gene activity and which downstream molecules might explain its protective, anti-metastatic role.
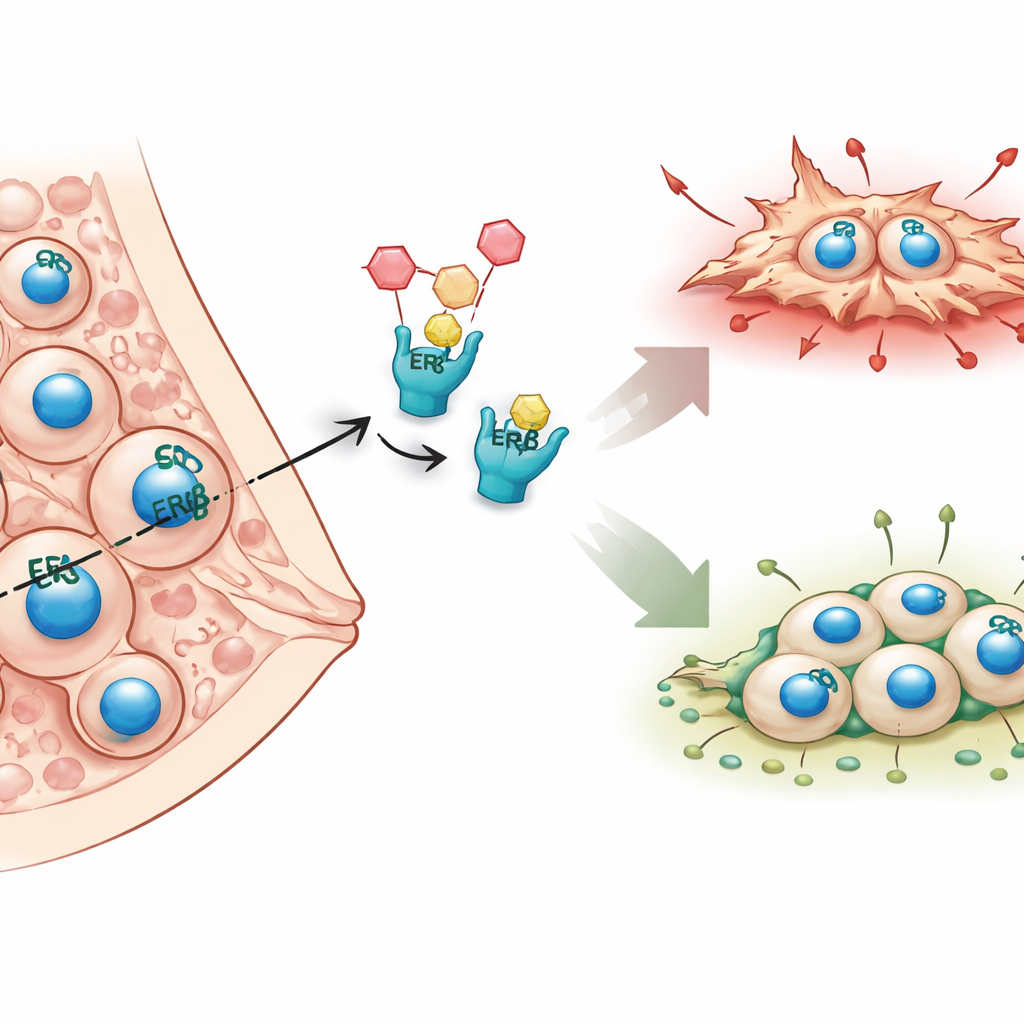
Reading the cancer cell’s instruction manual
To uncover ERβ’s influence, the researchers used two powerful sequencing approaches on inflammatory breast cancer cell lines. One method, ChIP-seq, pinpoints exactly where ERβ lands on the DNA, like flagging specific pages and lines in the cell’s instruction manual. The other method, RNA-seq, measures which genes are turned up or down when ERβ is present, absent, or switched on by a selective drug. Comparing these datasets, they found thousands of DNA sites bound by ERβ, most of them not just at gene on–off switches but deep within or between genes, suggesting wide-ranging control. They also discovered that ERβ often works together with families of other transcription factors, forming complexes that collectively decide which genes get silenced or activated.
Rewiring growth, movement, and metabolism
The integrated analysis showed that ERβ tends to dampen genes that promote cell movement, shape-shifting, and invasion, especially those tied to the actin skeleton and Rho GTPase signaling—pathways known to drive metastasis. At the same time, ERβ supports gene programs linked to tissue development and cell differentiation, nudging cancer cells toward a more settled, less aggressive state. The study also revealed a surprising role for ERβ in adjusting metabolic pathways, particularly those involving fatty acids and phospholipids, which are key fuel and building blocks for fast-growing tumors. Importantly, these effects were seen not only in cells engineered to overproduce ERβ but also in cells with natural levels of the receptor, underscoring its real-world relevance.
Small RNA messengers and clinical impact
Beyond protein-coding genes, the team examined microRNAs—tiny RNA molecules that fine-tune gene expression. They found dozens of microRNAs whose levels changed when ERβ was removed or restored, many of which have been linked to tumor suppression in breast cancer. By connecting these microRNAs to their target genes, the authors showed that ERβ controls some cancer drivers indirectly, through a layered regulatory network. They then turned to patient data, examining tumors from individuals with inflammatory and non-inflammatory breast cancer. Several ERβ-controlled genes were strongly tied to patient survival: high levels of some (such as SERPINA1 and HSPB8) predicted better outcomes, while high levels of others (such as PITX1 and HOMER3) were linked to worse survival. This positions these downstream molecules as potential markers for prognosis and as future drug targets.
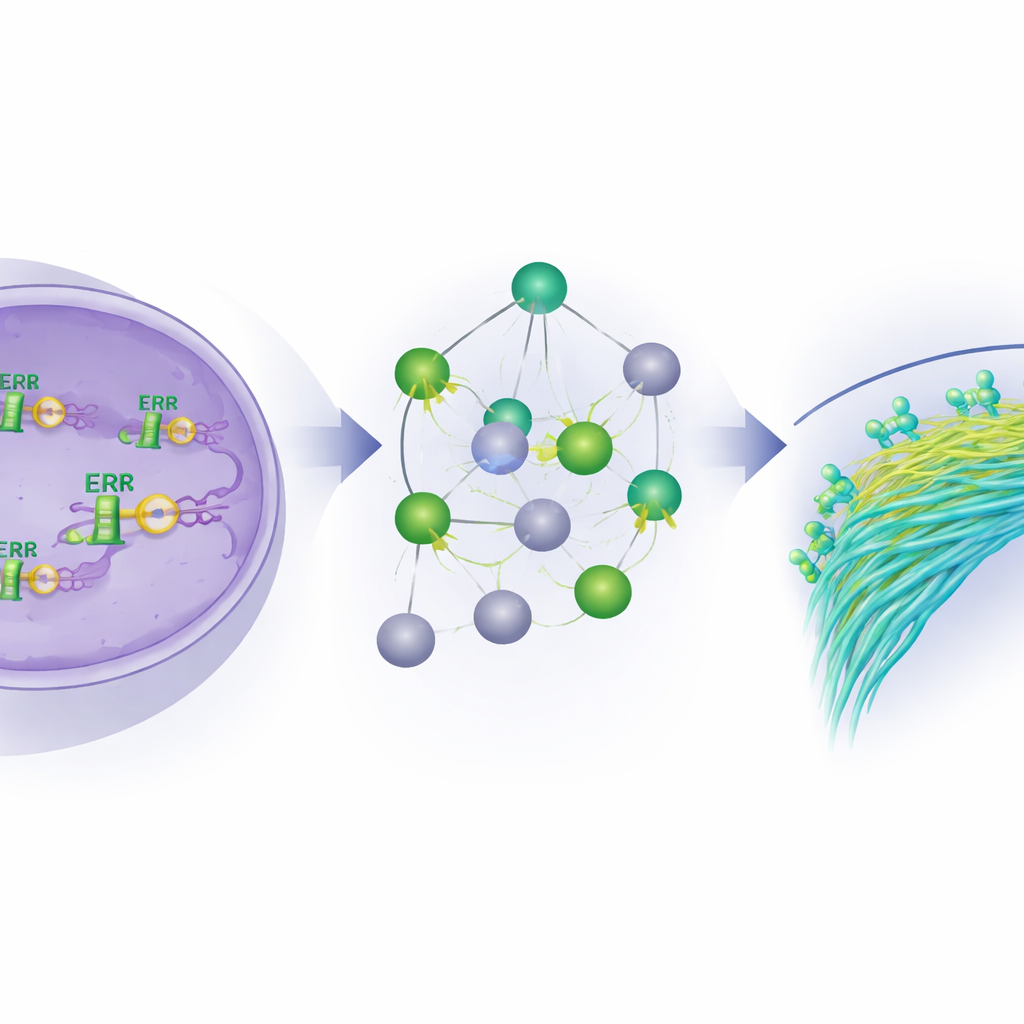
From molecular brake to future therapies
Altogether, the study paints ERβ as a central brake on the invasive behavior of inflammatory breast cancer cells. By binding to DNA, partnering with other regulators, and shaping both gene and microRNA networks, ERβ quiets programs that drive cell migration and metastasis while promoting more stable, differentiated states and re-tuning cancer metabolism. For patients, this suggests that drugs activating ERβ or inhibiting its key downstream targets—especially those involved in cell movement and fatty acid use—could form the basis of new, more precise treatments for this hard-to-treat disease.
Citation: Tastsoglou, S., Karagounis, I.V., Miliotis, M. et al. Estrogen receptor β target gene expression reveals novel repressive functions in aggressive breast cancer. npj Breast Cancer 12, 38 (2026). https://doi.org/10.1038/s41523-026-00905-4
Keywords: inflammatory breast cancer, estrogen receptor beta, metastasis, gene regulation, cancer metabolism