Clear Sky Science · en
The CXCL10/CXCR3 axis is essential for sustaining immunological dormancy in triple-negative breast cancer
Why some cancers sleep instead of spread
Breast cancer can sometimes behave like a sleeper agent. After initial treatment, a few cancer cells may hide in the body for years, not growing enough to be seen on scans—but still capable of waking up later and causing life‑threatening metastases. This paper explores why that “sleep” state, called dormancy, happens in an especially aggressive form of the disease, triple‑negative breast cancer, and uncovers a chemical signal that helps the immune system keep these cells in check.
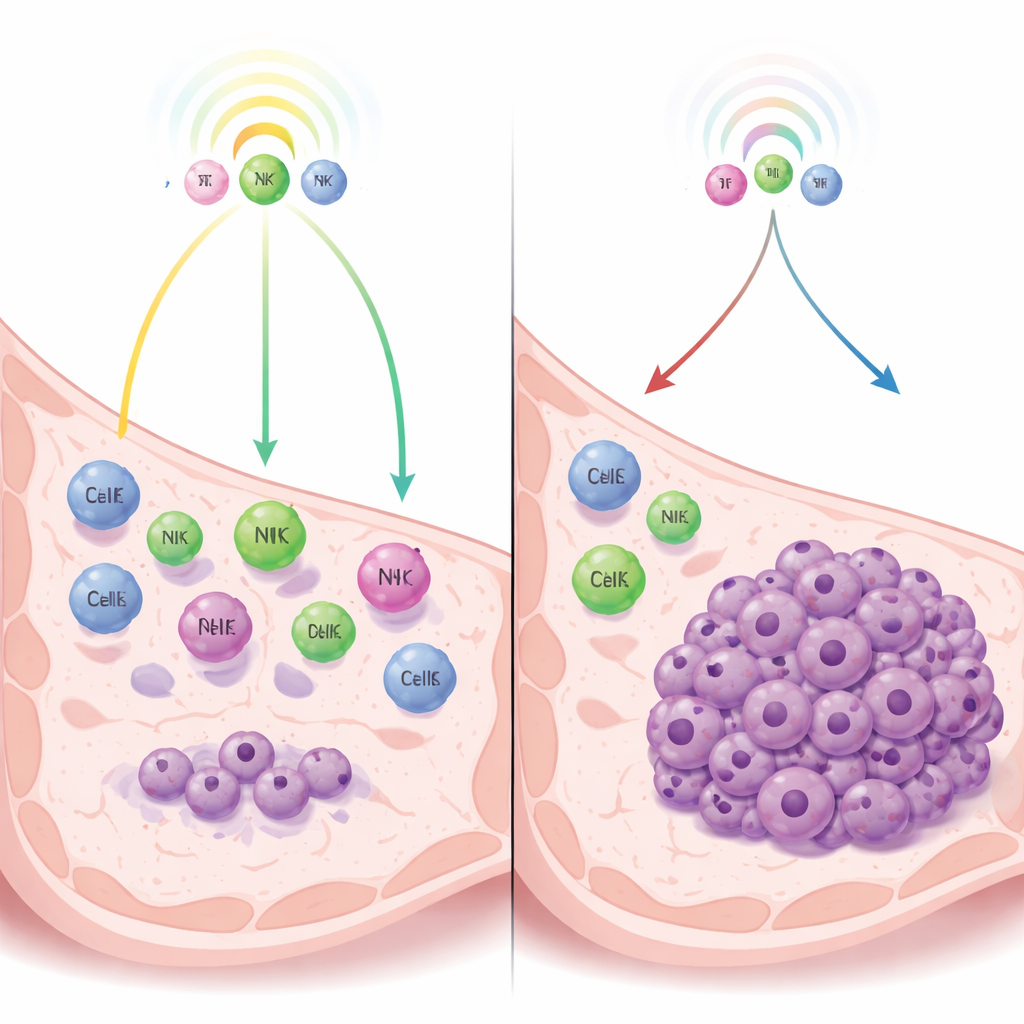
A tug‑of‑war between cancer and the immune system
The authors focus on a kind of dormancy controlled not by lack of blood supply or internal cell brakes, but by immune surveillance—the constant watch kept by immune cells that can hold tumors at bay without wiping them out. Using mouse models of triple‑negative breast cancer, they compare tumor cells that naturally remain dormant with closely related cells that form fast‑growing tumors. By examining which genes are switched on, they find that dormant cells strongly activate interferon pathways—immune alarm systems that stimulate the release of chemical messengers attracting immune cells into the tumor area.
A key chemical messenger that keeps tumors quiet
Among the many molecules turned on in dormant cells, one stands out: CXCL10, a small protein that acts like a beacon for immune cells carrying its partner receptor, CXCR3. Dormant cancer cells produce much more CXCL10 than their aggressive counterparts. When the researchers deliberately switch off CXCL10 in dormant cells and implant them into mice with intact immune systems, the once‑quiet cells rapidly form tumors. Blocking the CXCR3 receptor with an antibody has a similar effect. In both cases, cancer cells escape the immune stalemate and start to grow, showing that the CXCL10/CXCR3 axis is not just correlated with dormancy—it is required to maintain it.
How changing the local neighborhood tips the balance
Turning down CXCL10 does more than alter a single pathway; it reshapes the entire local immune landscape. In tumors that have lost CXCL10, helpful CD4 and CD8 T cells are reduced, while certain myeloid cells that can suppress immune responses become more abundant. Natural killer cells and dendritic cells also shift in number. This pattern appears both in primary tumors and in the lungs, where cancer cells can travel and lie low. Even when lung metastases are too small to see under the microscope, molecular markers reveal that more cancer cells succeed in seeding the lungs when CXCL10 is absent, and fewer T cells infiltrate those tissues. Yet in mice that lack a working immune system altogether, removing CXCL10 makes no difference—dormant cells grow regardless—underscoring that this signal acts through immune control rather than a direct brake on cancer cell growth.
Why more signal is helpful but not a magic switch
The team also tests the opposite idea: can boosting CXCL10 turn an aggressive tumor into a dormant one? When they engineer fast‑growing cancer cells to produce extra CXCL10, tumors grow more slowly and take longer to appear, and some immune cell types in the blood increase. However, full dormancy does not develop; eventually all mice still form tumors. This suggests that while CXCL10 is essential for maintaining dormancy when the rest of the immune machinery is in place, it cannot by itself build that complex state from scratch. Other pathways must cooperate to create a long‑lasting balance between immune attack and cancer cell survival.
From mouse experiments to patient outlook
To connect their findings to human disease, the researchers derive a “dormancy signature”—a set of genes that are consistently higher in dormant cells and drop when CXCL10 is silenced. They then look at large breast cancer patient datasets and ask how this signature relates to survival. In triple‑negative breast cancer, patients whose tumors express this dormancy signature at higher levels live longer overall and tend to remain relapse‑free for more years, consistent with an immune‑controlled dormant state delaying recurrence. The effect is not seen in the same way for hormone‑positive tumors, highlighting biological differences between breast cancer subtypes. Together, the results suggest that the CXCL10/CXCR3 axis is a central pillar of immune‑mediated dormancy in triple‑negative breast cancer, and that measuring this dormancy signature could help identify patients more likely to benefit from therapies that reinforce immune control—or, conversely, those who may need closer long‑term monitoring because their cancer is less well held in check.
Citation: Yilmaz, A., Haerri, L., Granda, M.E. et al. The CXCL10/CXCR3 axis is essential for sustaining immunological dormancy in triple-negative breast cancer. npj Breast Cancer 12, 36 (2026). https://doi.org/10.1038/s41523-026-00903-6
Keywords: triple-negative breast cancer, tumor dormancy, immune surveillance, chemokine signaling, CXCL10 CXCR3 axis