Clear Sky Science · en
Spatial gene expression analysis reveals drivers of extremely early lymph node metastasis in breast cancer
Why tiny traveling cancer cells matter
When breast cancer spreads beyond the breast, it often stops first in nearby lymph nodes—small filters that help fight infection. Doctors know that finding cancer in these nodes usually signals a higher risk of the disease returning. But what actually happens in the very first moments when just a handful of cells arrive in a lymph node has been almost impossible to see in real patients. This study used a new kind of microscopic “map” of gene activity to watch those earliest steps in unprecedented detail, revealing which cells succeed, which fail, and why that matters for patient outcomes.
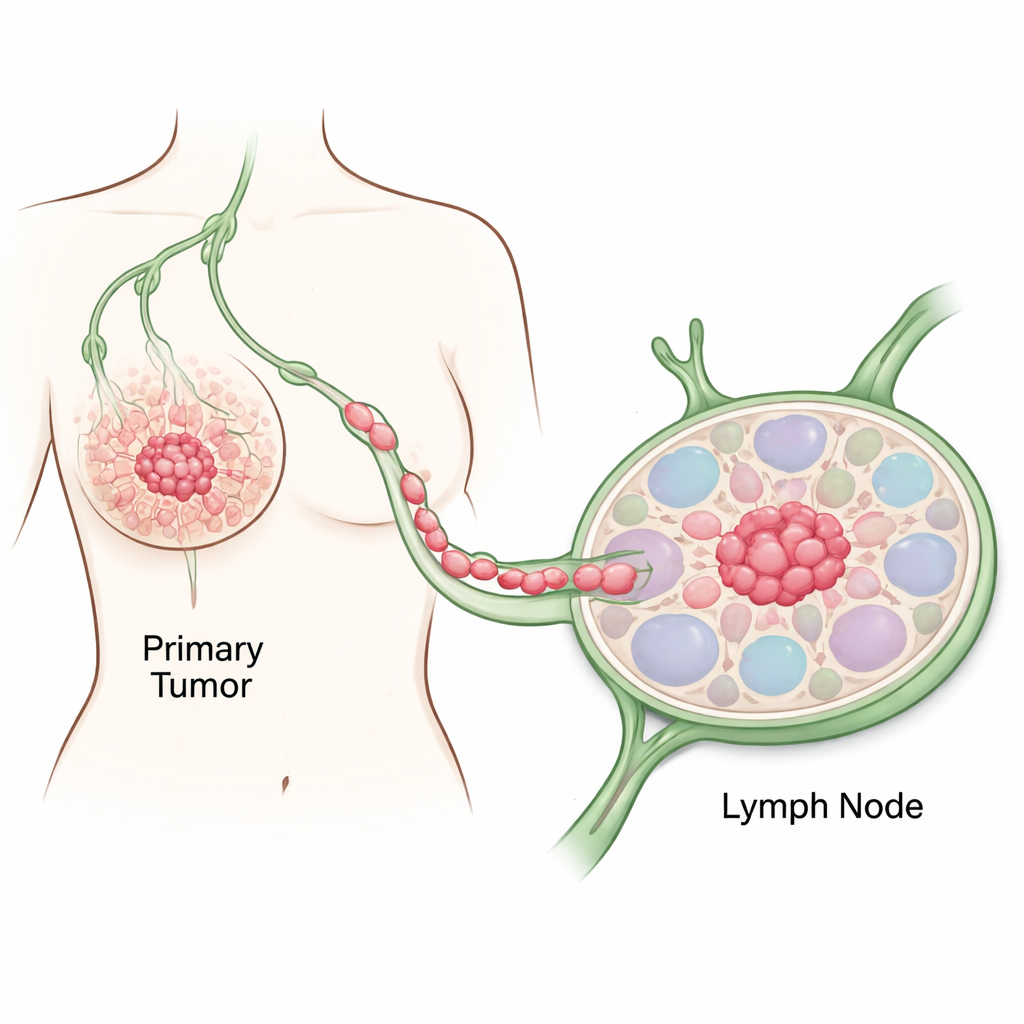
Following cancer’s first steps
The researchers focused on tissue from an 80-year-old woman with an aggressive, HER2-positive breast cancer. They sampled both normal-looking breast areas and nearby tumor regions, then used multiple DNA and RNA tests to trace how ordinary cells became cancerous. Compared with healthy tissue, tumor regions showed classic hallmarks of cancer: disrupted cell adhesion, hyperactive cell division, large-scale DNA methylation changes, and key mutations in genes that control growth and genome stability. These molecular shifts lined up with what pathologists saw under the microscope, confirming a stepwise march from normal breast cells to fully malignant ones.
Shape-shifting cells on the move
To understand how tumor cells start to travel, the team used a high‑resolution technique that reads gene activity in individual cells while keeping their exact positions in the tissue. This allowed them to reconstruct how cancer cells changed identity as they left normal breast structures and moved toward metastasis. They found that the cancer appeared to arise from one particular type of mature breast cell, which then gradually adopted features of a process called epithelial–mesenchymal transition—a kind of shape-shifting that helps cells loosen, move, and invade nearby tissue. Instead of a simple on-off switch, cells occupied a spectrum of mixed states, blending “epithelial” traits (orderly, stuck-together) with “mesenchymal” traits (mobile, invasive). Some of these hybrid cells dispersed through the tumor, while others clustered into more advanced cancer groups that were primed to spread to lymph nodes.
The first remote beachhead in a lymph node
In a lymph node that was judged metastasis-free by standard clinical tests, the scientists spotted something remarkable: just 30 stray tumor cells, occupying a region only about 200 micrometers wide—roughly twice the width of a human hair. These isolated tumor cells came from several distinct hybrid states, each with its own gene activity pattern. By grouping related cancer cells, the team defined six subtypes with different roles. One subtype, strongly “mesenchymal,” looked especially good at resisting cell death and escaping immune attack. Another subtype, which regained more epithelial features, showed a striking shift in its energy use, favoring fatty acid metabolism. Only this metabolically rewired, more epithelial-like group successfully formed small colonies in lymph nodes, suggesting that fuel choice—not just invasiveness—helps determine whether early arrivals can take root.
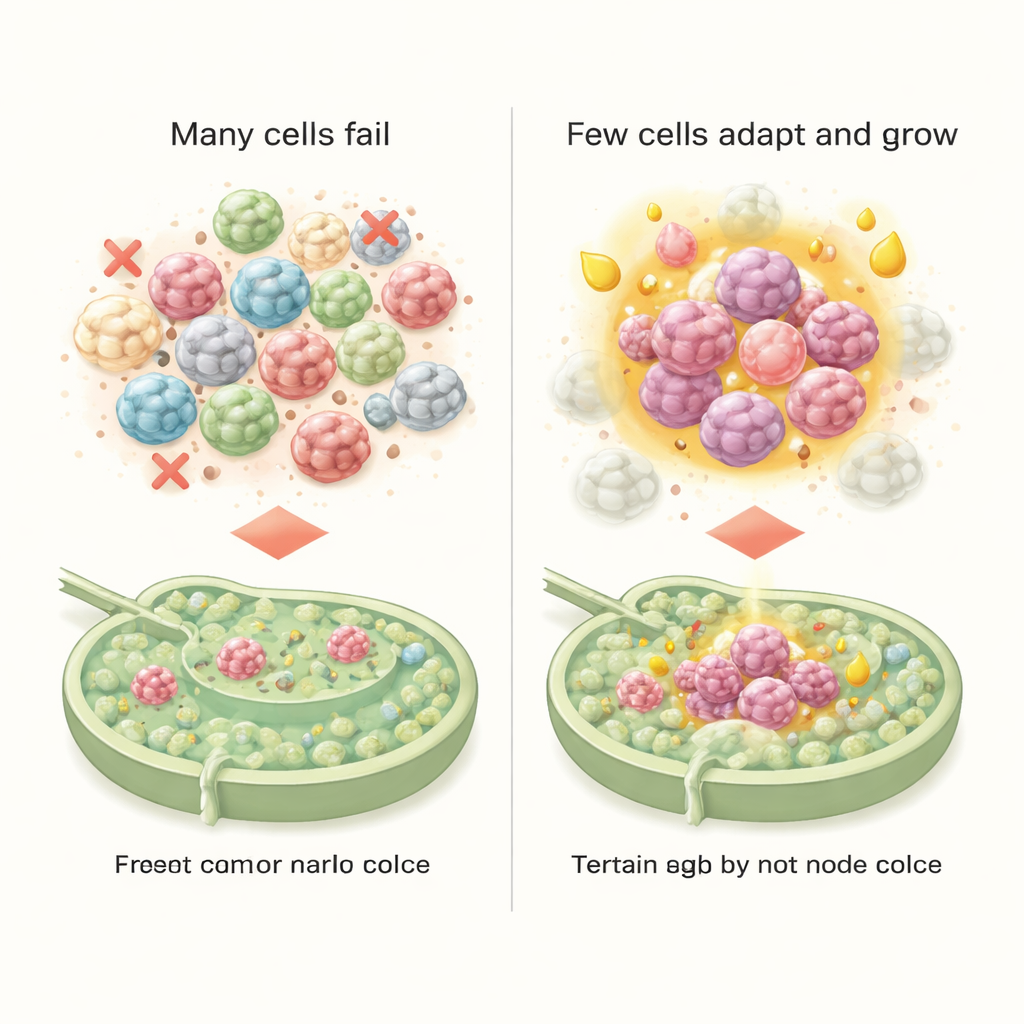
A lymph node that quietly helps cancer
The study also examined how the lymph node environment responded to these intruders. Even before a visible metastasis had formed, immune cells, blood vessel cells, and connective tissue were already sending signals that blunted immune reactions and supported tumor survival. Certain receptor–ligand pairs between immune cells hinted at early immune tolerance, while others encouraged new blood vessel growth and remodeling of the tissue scaffold. Together, these changes built a niche that was surprisingly welcoming to the metabolically adapted cancer cells, helping them survive, grow, and potentially seed future spread.
What this means for patients
By comparing their findings with large, existing patient datasets, the researchers made a counterintuitive discovery. The cells best at forming early lymph node colonies, and even those with the strongest mesenchymal features, did not correspond to the worst clinical outcomes. Instead, patients fared poorest when their tumors harbored certain aggressive hybrid cell types marked by high cell-cycle activity and sugar-burning (glycolysis) programs driven by genes like MYC. In other words, different shape-shifted cell states seem to specialize: some are experts at quietly colonizing new sites, while others drive rapid growth and recurrence. Understanding and targeting these distinct cell states and their metabolic preferences could lead to more precise treatments—ones that not only detect the earliest spread more reliably but also cut off the tumor cell types most likely to cause deadly relapses.
Citation: Nagasawa, S., Kajiya, K., Ishikawa, E. et al. Spatial gene expression analysis reveals drivers of extremely early lymph node metastasis in breast cancer. npj Breast Cancer 12, 28 (2026). https://doi.org/10.1038/s41523-026-00897-1
Keywords: breast cancer metastasis, lymph node, epithelial mesenchymal transition, spatial transcriptomics, cancer metabolism