Clear Sky Science · en
CD52 signaling via macrophage Siglec-G represents a therapeutic target for cancer immunotherapy
Why some breast cancers hide from the immune system
Triple‑negative breast cancer is one of the most dangerous forms of breast cancer because it spreads quickly and lacks many of the common treatment targets. This study explores a newly discovered way these tumors can hide from the body’s defenses and shows how blocking that escape route could make immune‑based therapies work better.
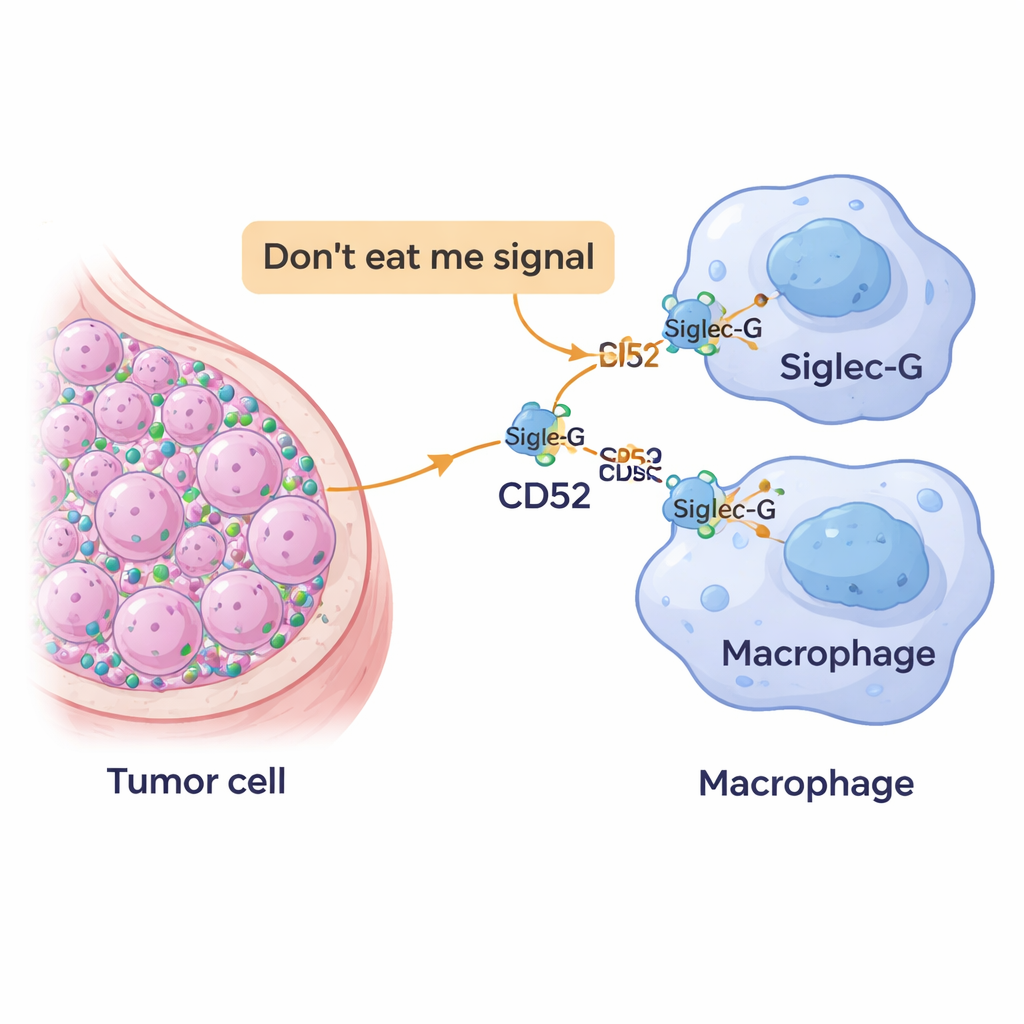
A new "do not eat me" flag on cancer cells
Our immune system relies on scavenger cells called macrophages to patrol tissues and swallow dangerous or abnormal cells. To avoid being mistakenly destroyed, healthy cells display surface markers that signal, in effect, “I belong here.” Cancer cells often hijack this system by overusing such protective signals, turning them into powerful “don’t eat me” flags. The researchers focused on a molecule called CD52, already known from blood cancers and autoimmune disease, and asked whether breast tumor cells also use it to escape macrophage attack.
Finding CD52 in aggressive breast tumors
The team analyzed large public cancer databases and tissue samples from patients. They found that breast tumors, especially triple‑negative ones, produced more CD52 than normal breast tissue. Moreover, tumors with higher CD52 tended to be less well‑differentiated, a feature often linked with more aggressive disease. At the same time, immune cells in and around the tumors carried high levels of a partner molecule called Siglec‑10 (and its mouse counterpart Siglec‑G), which sits on macrophages and can send calming signals inside these cells. Together, these patterns suggested that CD52 on tumor cells might be directly talking to Siglec‑G on macrophages to keep them from doing their job.
Switching off CD52 to free macrophages
To test this idea, the scientists used gene‑editing tools to remove CD52 from mouse triple‑negative breast cancer cells. When these modified cells were mixed with macrophages in the lab, the macrophages swallowed significantly more tumor cells than when they faced unmodified cancer cells. Importantly, the cancer cells themselves did not grow or die differently after losing CD52, indicating that the key effect was on immune recognition rather than on the tumor cells’ own biology. When the same tumor cells were implanted into mice, those lacking CD52 formed smaller tumors and were surrounded by macrophages that showed more evidence of having eaten cancer cells. If macrophages were depleted from the mice, this growth advantage disappeared, confirming that CD52’s main role was to shield tumors from being eaten.
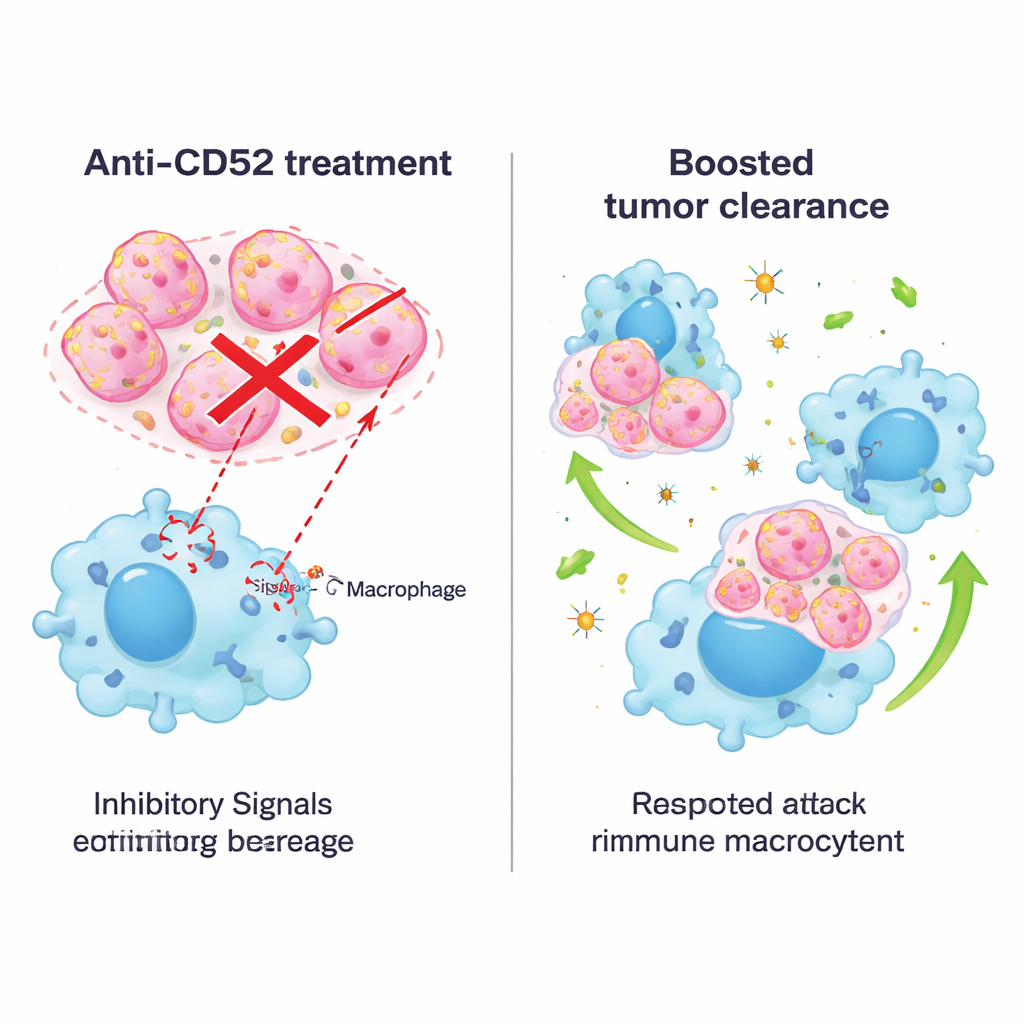
Blocking the signal with antibodies and combining treatments
Because drugs that target CD52 are already used in other diseases, the researchers next tried a blocking antibody in mouse models. Treating tumor‑bearing mice with an anti‑CD52 antibody reduced tumor growth, extended survival, and did not cause obvious organ damage or weight loss. In dishes, similar antibodies boosted the ability of both mouse and human macrophages to engulf breast cancer cells from several subtypes, not just triple‑negative. The team then combined CD52 blockade with anti‑PD‑1 therapy, a widely used checkpoint drug aimed at T cells. In a spontaneous mouse model of triple‑negative breast cancer, the combination worked better than either treatment alone: tumors shrank more, macrophages became more inflammatory and active, and more potent cancer‑killing T cells entered the tumor while fewer exhausted T cells remained.
What this means for future breast cancer care
This work uncovers CD52 as a previously unrecognized “don’t eat me” signal that triple‑negative breast cancer cells use to fend off macrophages. By interrupting the conversation between CD52 on tumor cells and Siglec‑G on macrophages, the researchers were able to reawaken both innate immune cells and, indirectly, T cells, especially when paired with PD‑1 blockade. While more safety and clinical studies are needed—because CD52 also appears on many normal immune cells—the findings point to a promising dual‑checkpoint strategy that could help turn hard‑to‑treat, immune‑resistant breast tumors into ones that respond better to immunotherapy.
Citation: Qin, X., Chang, Y., Qiu, Y. et al. CD52 signaling via macrophage Siglec-G represents a therapeutic target for cancer immunotherapy. npj Breast Cancer 12, 29 (2026). https://doi.org/10.1038/s41523-026-00895-3
Keywords: triple-negative breast cancer, macrophages, immunotherapy, CD52, checkpoint blockade