Clear Sky Science · en
Microbial regulation of stress-associated signaling molecules and its role in health and disease
How Tiny Gut Tenants Shape Our Stress
Most of us think of stress as something that happens in our heads, but this article shows that some of the most important players in our stress response live in our gut. The microbes that inhabit our intestines do far more than help digest food: they can make, break down, and reshape the very hormones that control how our bodies react to pressure, fear, and pain. Understanding this hidden partnership may open new ways to ease conditions like inflammatory bowel disease, depression, and other stress-linked illnesses.
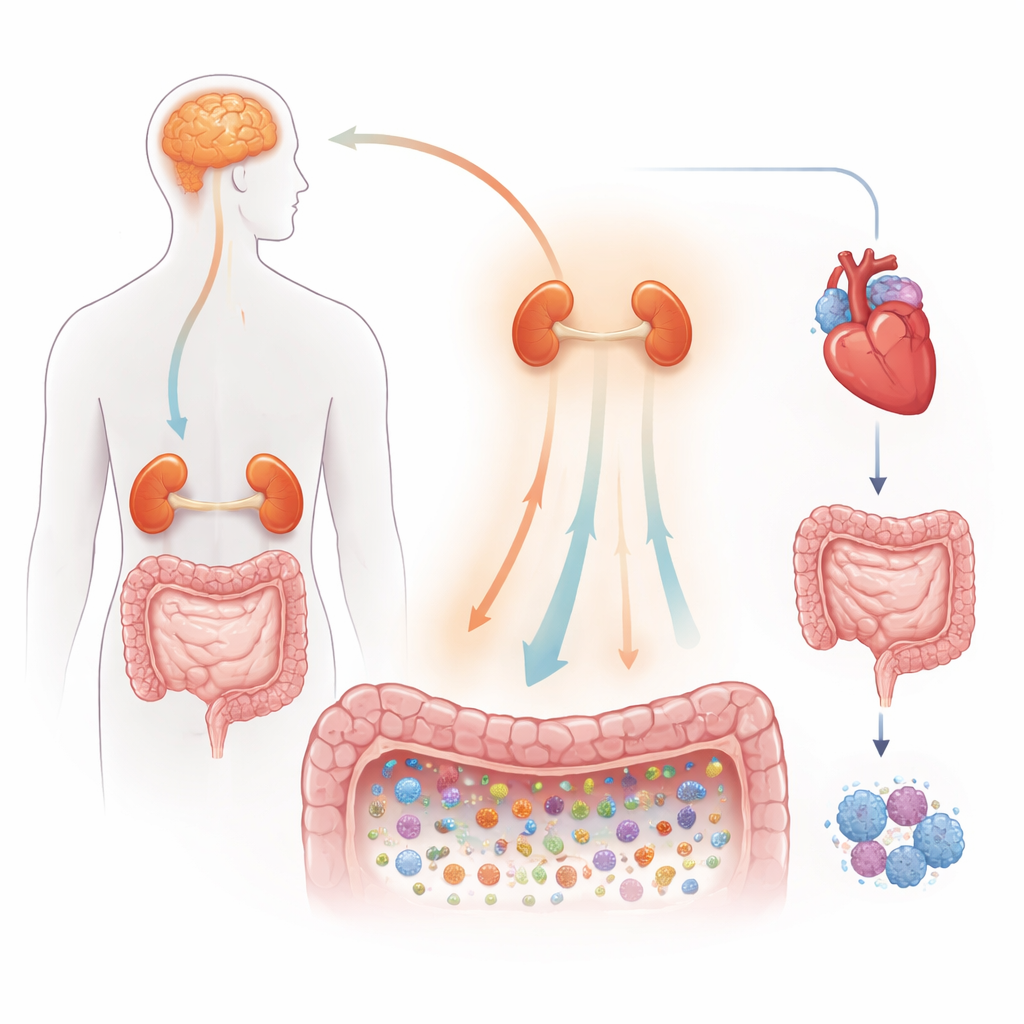
The Body’s Built-In Alarm System
The authors begin by outlining how the stress response normally works. When we face a challenge, the brain activates a chain of command that runs through the pituitary and adrenal glands, flooding the body with chemical messengers like adrenaline-like catecholamines and steroid hormones called glucocorticoids. These signals raise heart rate, shift blood flow, change gut movement, and tune the immune system for “fight-or-flight.” Nerves that wire directly into the digestive tract provide a fast track for signals to the intestine, tightening or loosening the gut barrier and altering the local environment where microbes live. This makes the gut a key crossroads where stress biology and the microbiome continually meet.
Microbes Listening and Talking to Stress Signals
Research over several decades reveals that many bacteria can sense and use our stress chemicals. Early experiments showed that certain gut and disease-causing bacteria grow faster and become more aggressive when exposed to catecholamines such as norepinephrine and epinephrine. Other work demonstrated that bacteria can take up these molecules, convert them into new forms, or even produce similar compounds themselves from amino acids like tyrosine. At the same time, host enzymes often “switch off” catecholamines by attaching small chemical groups to them; gut bacteria equipped with the right tools can snip these groups off again, restoring the active form. In mice without any microbes, most stress messengers in the gut are locked in inactive form, but introducing specific bacteria restores the active pool, showing that microbes directly control how much of these signals are available.
Reworking Stress Steroids from the Inside
The review then turns to glucocorticoids, the steroid hormones best known for taming inflammation and helping the body cope with ongoing stress. Small amounts of these hormones enter the gut from the bloodstream or are made locally in the intestinal lining, where they encounter dense bacterial communities. Many gut microbes carry specialized enzymes that can trim, rearrange, or reduce these steroid molecules. Some chop off side chains to turn them into androgens, which act more like sex hormones and can influence blood pressure and even hormone-sensitive cancers. Others remove particular chemical groups, weakening the hormone’s ability to bind its receptor, or further reduce the structure so it can no longer be recycled into powerful forms. Still others reverse the body’s own “inactivation” steps by removing attached sulphate or sugar-like groups, shifting steroids back into active circulation. Together, these actions make the microbiome a moving control knob on steroid strength and balance.
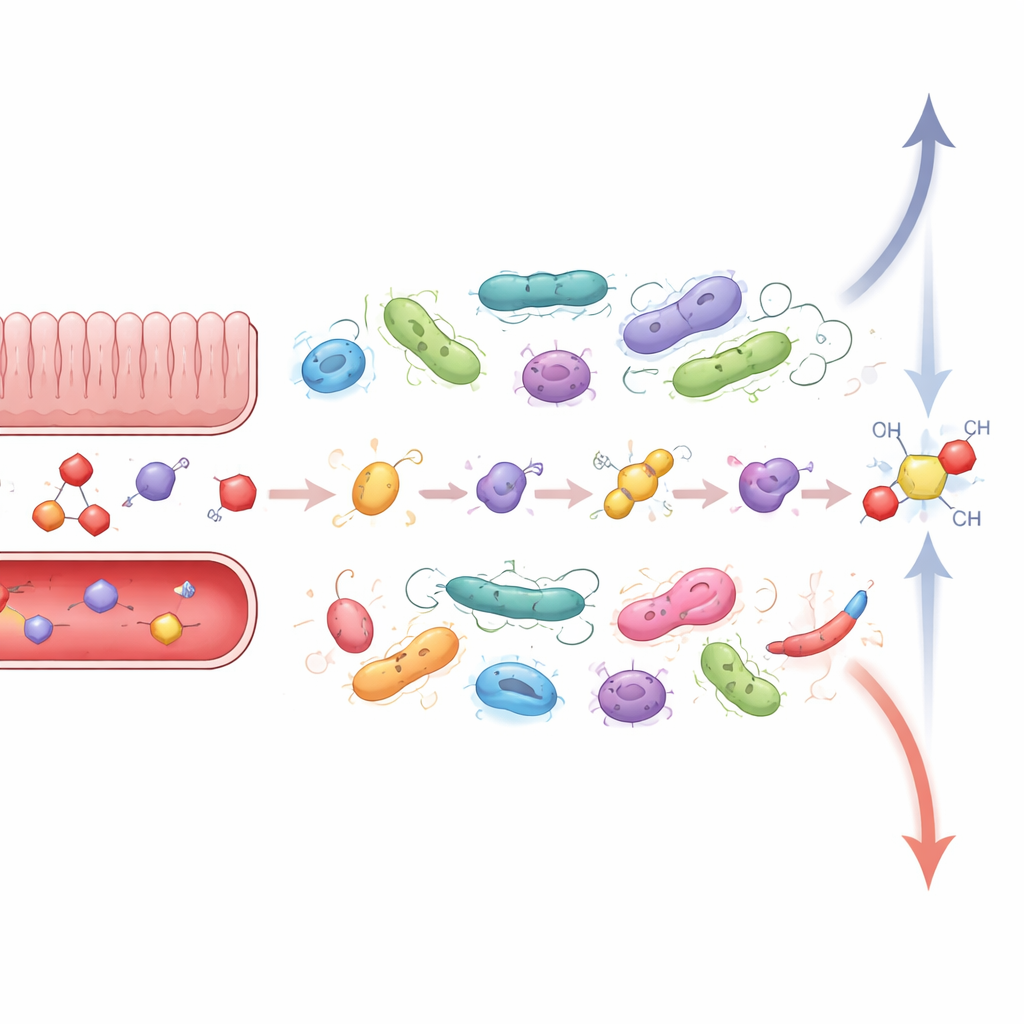
When Stress, Inflammation, and Microbes Feed Each Other
These intimate chemical exchanges matter most in diseases where both stress and gut microbes are already disrupted. Inflammatory bowel disease (IBD) is a prime example: patients show chronic gut inflammation, altered microbial communities, and heightened sensitivity to life stress. Stress hormones can loosen the intestinal barrier, allowing more microbes and their products to slip across and stir the immune system. At the same time, IBD is associated with blooms of species that can remodel cortisol and related steroids, as well as losses of bacteria that make short-chain fatty acids, small fermentation products that normally help seal the barrier and calm inflammation. Animal studies suggest that these microbial shifts can amplify hormone imbalances and immune activity, locking patients into a vicious cycle where stress worsens gut disease and gut disease heightens stress.
New Ways to Calm the Gut–Stress Loop
Because microbes sit at the junction of stress signals and inflammation, they are attractive targets for new therapies. The article highlights work showing that diets rich in fibers such as inulin can boost beneficial fermentation products, ease gut inflammation, and blunt some stress-related damage in IBD models. Probiotic strains of Lactobacillus have been found to tighten the gut barrier, reduce stress-induced hormone spikes, and lower pain sensitivity in animals. Direct delivery of short-chain fatty acids to the colon can reduce anxiety-like behavior in mice and dampen cortisol responses to social stress in humans. These early successes suggest that carefully designed probiotics, prebiotics, and microbial metabolites could help reset the stress response by reshaping hormone handling in the gut.
What This Means for Everyday Health
In closing, the authors argue that gut microbes should be viewed as active partners in our stress biology, not passive bystanders. By making, modifying, and recycling stress-related signaling molecules, the microbiome can influence how strongly we react to challenges, how quickly we recover, and how likely stress is to tip us into chronic disease. This does not mean that a single “magic” microbe will cure stress, but it does suggest that protecting and nurturing a healthy gut ecosystem may be one of the most powerful tools we have for building resilience. As researchers map these microbe–hormone interactions in more detail and test them in people, everyday strategies that support gut health could become a core part of managing stress, mood, and inflammatory conditions.
Citation: Sie, C., Tropini, C. Microbial regulation of stress-associated signaling molecules and its role in health and disease. npj Biofilms Microbiomes 12, 65 (2026). https://doi.org/10.1038/s41522-026-00932-w
Keywords: gut microbiome, stress hormones, microbiota-gut-brain axis, inflammatory bowel disease, microbial endocrinology