Clear Sky Science · en
Targeted elimination of Staphylococcus aureus mastitis infections with synthetic phage-based CRISPR-Cas delivery systems
Fighting a Costly Dairy Infection
Milk from cows and other farm animals can be compromised by a painful breast infection called mastitis, often caused by the bacterium Staphylococcus aureus. Treating these infections typically requires long courses of antibiotics, which are expensive, stressful for animals, and contribute to the growing problem of antibiotic resistance. This study explores a radically different approach: turning a natural virus‑like parasite of bacteria into a precision weapon that can selectively cut the bacterium’s DNA and clear infections without spreading resistance genes.
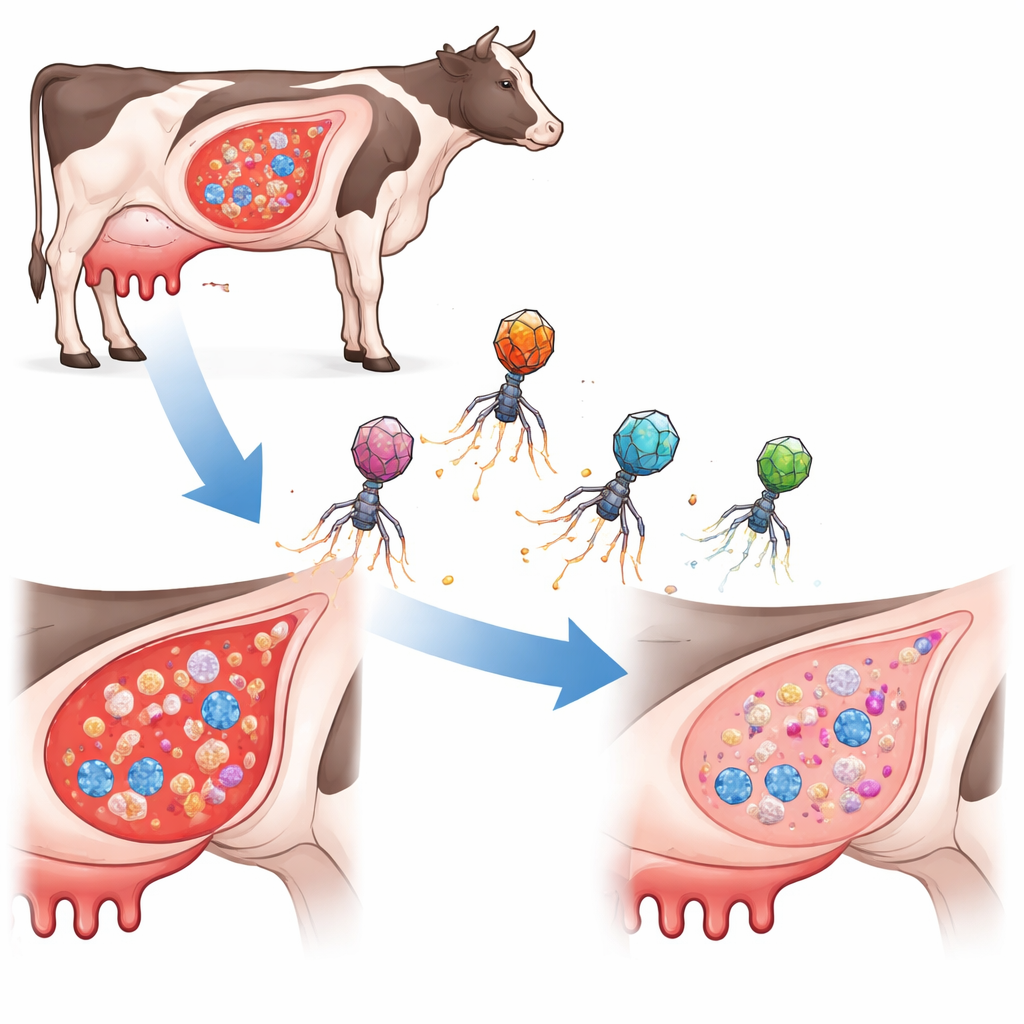
A New Type of Targeted Bacterial Hunter
Instead of using traditional antibiotics or ordinary bacteriophages (viruses that infect bacteria), the researchers engineered tiny DNA‑carrying elements known as phage‑inducible chromosomal islands. In nature, these islands hitchhike inside phage shells to jump between bacterial cells. Here, the team re‑built them as “ePICIs” that carry the gene‑editing tool CRISPR‑Cas9 plus short guide sequences aimed at three small but important regulatory RNAs in S. aureus. When an ePICI enters a target cell, Cas9 is produced and cuts the bacterium’s chromosome at the chosen site, killing the cell from within. Crucially, these engineered particles do not replicate on their own, which limits their spread and reduces the risk of accidentally moving harmful genes around.
How the Bacterial Shield Changes the Battle
S. aureus often survives treatment by forming biofilms—sticky communities where cells are embedded in a self‑made matrix. The team compared two common types of matrix: one built mainly from a protein called Bap, and another made from a sugar polymer known as PIA/PNAG. In lab tests, Bap‑rich biofilms strongly protected bacteria against the ePICIs and against a standard helper phage, acting like a dense armor that blocked infection. In contrast, biofilms based on the sugar polymer made the bacteria easier to kill, likely because enzymes on the viral shells can chew through these sugars and help the particles reach their targets.
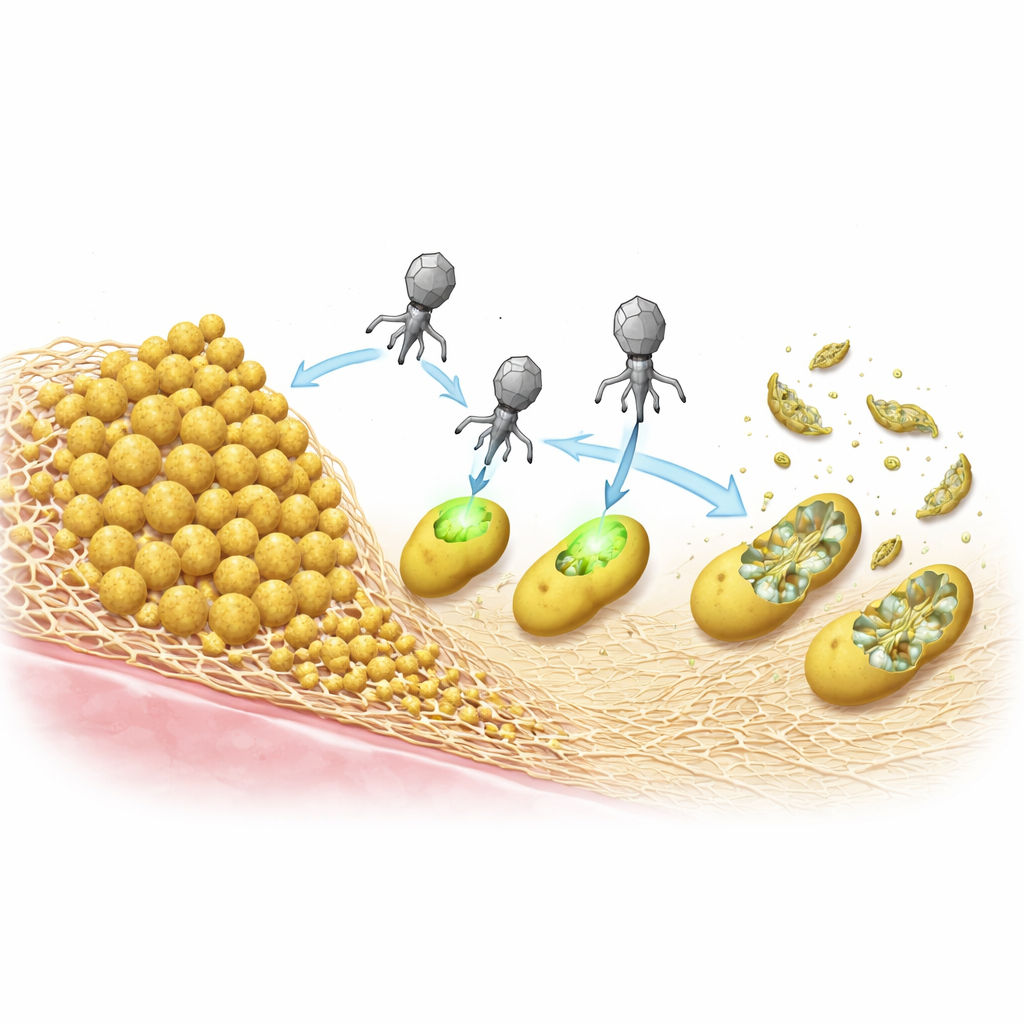
Killing Bacteria Without Joining Their DNA
Once inside a cell, ePICIs can integrate into the bacterial chromosome, but the researchers asked whether that step is actually necessary for killing. They created a version that could no longer integrate and showed that, as long as the particle was produced correctly in the source strain, it was just as lethal to S. aureus as the original. This means the CRISPR system can be expressed directly from the incoming circular DNA, do its job, and then be lost as the cell dies. That integration‑free action further lowers the chances of unintended gene transfer and distinguishes these particles from classic phage therapy, which relies on viral replication and cell bursting to spread.
From Petri Dish to Infected Mammary Glands
Because Bap‑producing strains are common in dairy mastitis, the team tested their best‑performing construct, called ePICIrsaE, in a mouse model that mimics udder infection. In milk and in lab cultures, Bap‑rich biofilms clearly blunted ePICI activity. Yet inside the mammary gland, repeated doses of ePICIrsaE sharply reduced bacterial numbers for all tested strains, including those that made Bap. Its performance was comparable to vancomycin, a powerful last‑line antibiotic. The results suggest that conditions in living tissue—such as lower Bap production, the presence of free‑floating bacteria, or helpful components in milk—allow the engineered particles to work far better than in static lab biofilms.
What This Could Mean for Farms and Beyond
The study shows that non‑replicating, phage‑packaged CRISPR systems can selectively eliminate S. aureus in a difficult, biofilm‑linked infection without spreading themselves or rupturing huge numbers of cells at once. Although high particle doses are needed and scaling from mice to cows will be challenging, these particles offer a promising, controllable alternative to antibiotics for localized infections such as mastitis. More broadly, the work highlights that treatments that look disappointing in simple lab tests may still succeed in the complex environment of an animal, and it opens the door to libraries of customized ePICIs tuned to different bacteria, viral shells, and guide sequences.
Citation: Garmendia-Antoñana, N., Dorado-Morales, P., Gil, C. et al. Targeted elimination of Staphylococcus aureus mastitis infections with synthetic phage-based CRISPR-Cas delivery systems. npj Biofilms Microbiomes 12, 67 (2026). https://doi.org/10.1038/s41522-026-00931-x
Keywords: bovine mastitis, Staphylococcus aureus, CRISPR therapy, phage-based treatment, biofilm infections