Clear Sky Science · en
The gut microbiota mediates depression-like behaviors in mice with chronic Echinococcus multilocularis infection
When a Hidden Infection Affects the Mind
Most people think of parasites as a problem for the gut or liver, not the mind. Yet many patients with long-lasting infections report low mood, fatigue, and trouble enjoying life. This study explores a surprising link: how a chronic tapeworm infection that lodges in the body cavity and liver might, through changes in gut bacteria and chemistry, drive depression-like behavior in mice. Understanding this chain of events could eventually point to new ways to protect mental health in people living with stubborn infections.
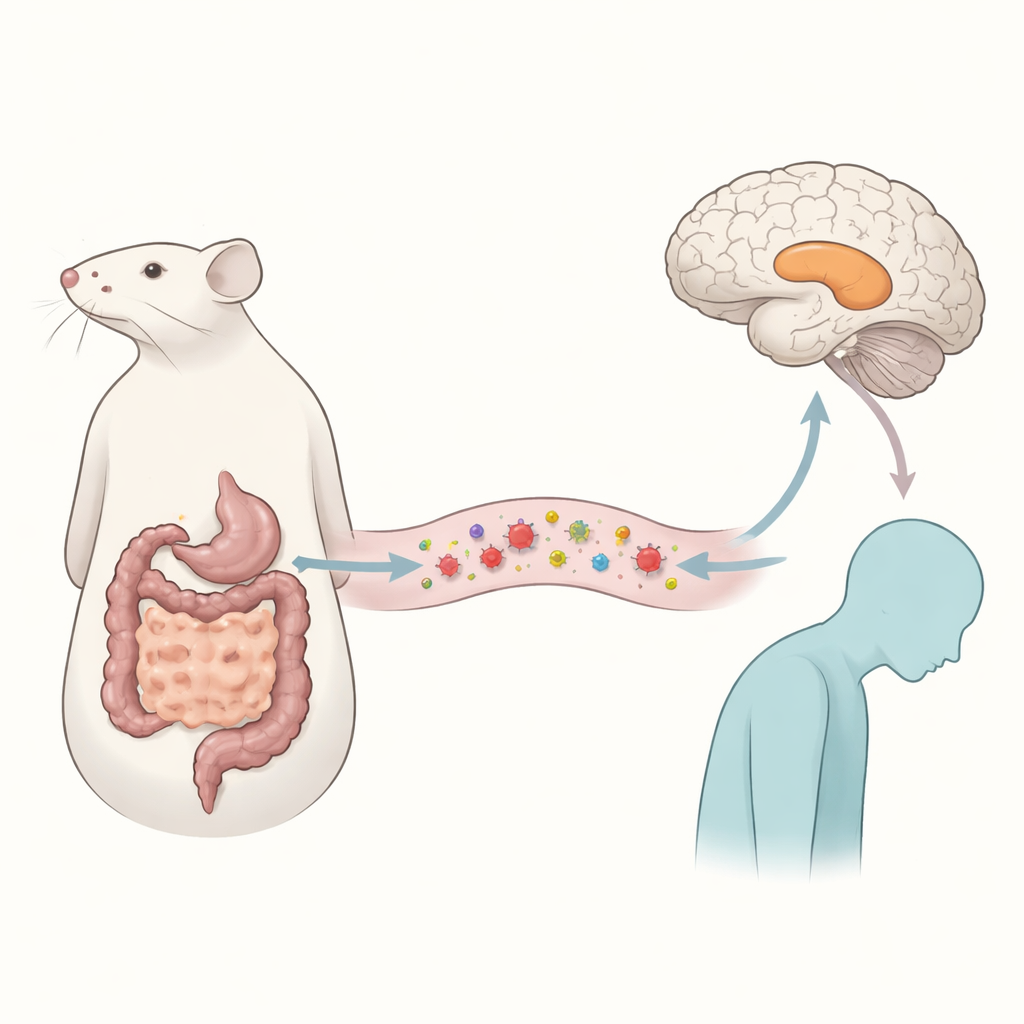
A Parasitic Passenger and Changes in Mood
The researchers focused on Echinococcus multilocularis, the parasite behind alveolar echinococcosis, a severe disease that mainly attacks the liver. Patients with this condition often report poor mental well-being, but the reasons have been unclear. To probe this, the team infected mice with the parasite and waited two months, long enough to mimic a chronic infection. The animals then completed a battery of behavioral tests designed to distinguish between simple sickness and more specific mood changes. Infected mice moved just as much as healthy ones and learned and remembered tasks normally, but they showed clear signs of depression-like behavior: less interest in sweet rewards and more immobility in stress-based tests that are widely used as readouts of despair-like states in rodents.
Subtle Brain Changes and Immune Signals
Looking inside the brain, the scientists homed in on the hippocampus and amygdala, regions deeply involved in emotion and stress. The overall structure of these areas remained intact, but under the microscope some neurons looked shrunken or oddly shaped, and the outer membrane of their nuclei appeared wrinkled. The brain’s resident immune cells, microglia, were more numerous in certain regions and had simpler, less-branched shapes—hallmarks of a shift away from their calm, watchful state toward an activated one. At the same time, both the gut tissue and the blood of infected mice carried higher levels of inflammatory molecules, especially the cytokines IL‑6 and MCP‑1, and the genes for some of these signals were more active in the hippocampus. Together, these findings point to a slow-burning inflammatory state that reaches from the intestine into the brain and nudges its immune cells out of balance.
Gut Bacteria and Lost Chemical Messengers
The team then asked whether the community of microbes living in the gut might be part of the story. Using DNA sequencing, they found that infection reshaped the intestinal microbiota: beneficial groups such as Lactobacillus and Bifidobacterium shrank dramatically, while other genera, including Ruminococcus and Prevotella, expanded. Mice with fewer friendly bacteria tended to have higher levels of inflammatory cytokines in both gut and blood. At the same time, blood tests and broad metabolite profiling revealed that key components of the tryptophan–serotonin pathway were depleted. Levels of tryptophan (a dietary amino acid), its intermediate N‑acetylserotonin, and the neurotransmitter serotonin (5‑HT) all dropped in infected animals. These changes mirror alterations reported in people with major depression and were strongly linked to the abundance of specific gut microbes.
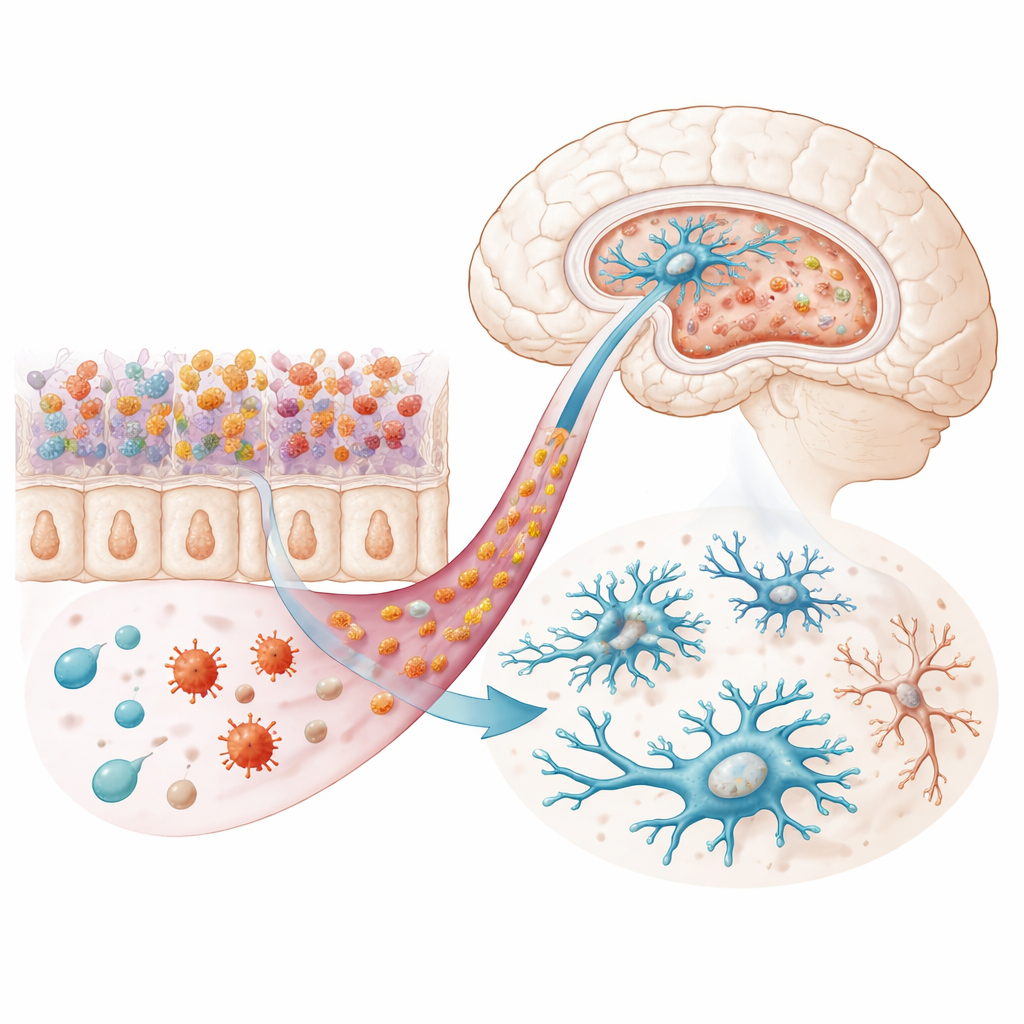
Transferring the Microbiome, Transferring the Mood
To test whether the altered microbiota was enough to change behavior, the researchers performed fecal microbiota transplantation. They first cleared the gut bacteria of healthy mice with antibiotics, then introduced stool from either infected or uninfected donors. Remarkably, recipients of microbiota from infected donors developed depression-like behaviors and anxiety-like avoidance of the center of an open arena, even though they themselves were never exposed to the parasite. These mice also showed heightened inflammation, reduced levels of tryptophan and serotonin-related molecules in the blood, and microglia that resembled those in directly infected animals—more abundant in certain regions, less branched, and filled with lysosome-like structures suggestive of increased cellular stress or cleanup activity.
What This Means for Human Health
For a layperson, the main message is that a chronic infection in the body can disturb the gut’s microbial residents and the chemical messengers they help produce, and that this, in turn, can influence the brain’s immune cells and mood circuits. The study does not claim that this tapeworm causes clinical depression in people, but it offers a detailed roadmap of how a long-term parasite, an inflamed gut, disrupted microbiota, and altered tryptophan–serotonin chemistry could work together to generate depression-like behaviors. This emerging picture suggests that treating mental health complications of chronic infections might one day include not only killing the parasite, but also calming inflammation and restoring a healthy gut microbiome.
Citation: Wen, R., Xin, Y., Bao, S. et al. The gut microbiota mediates depression-like behaviors in mice with chronic Echinococcus multilocularis infection. npj Biofilms Microbiomes 12, 63 (2026). https://doi.org/10.1038/s41522-026-00929-5
Keywords: gut–brain axis, microbiome, parasitic infection, depression, tryptophan metabolism