Clear Sky Science · en
The ppGpp-HpaR1-gum regulatory pathway modulates exopolysaccharides production in Xanthomonas campestris pv. campestris
Why sticky bacteria matter to crops
Many disease-causing bacteria survive and spread by wrapping themselves in slimy sugar coats known as biofilms. In vegetable crops like cabbages and broccoli, these coats can clog the plants’ water pipes, causing the notorious “black rot” disease and heavy yield losses. This study looks inside one such bacterium, Xanthomonas campestris, to reveal how an internal chemical alarm system controls the production of its sugary shield. Understanding this hidden control circuit could help scientists design better ways to protect crops or even harness these sugars for useful materials.
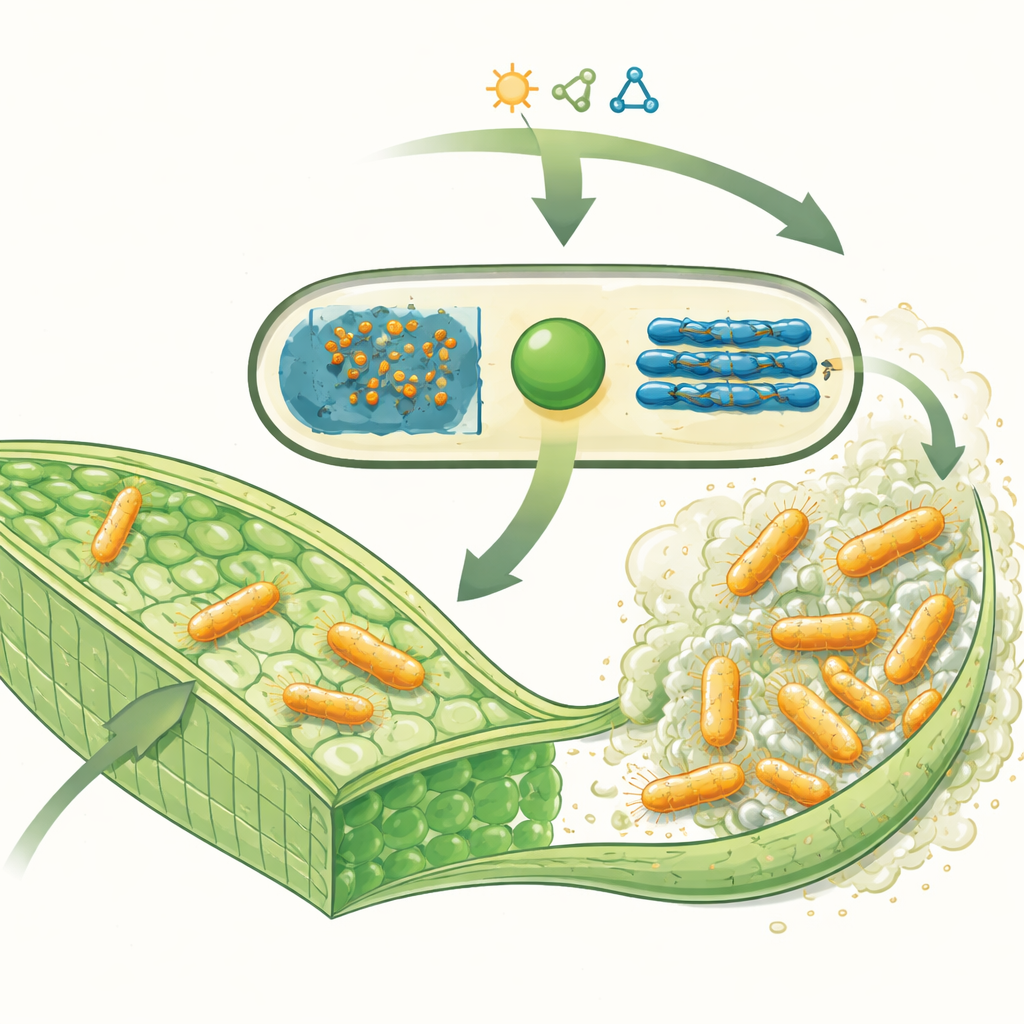
The plant-infecting microbe and its sugar armor
Xanthomonas campestris enters the veins of cruciferous vegetables—plants such as cabbage, cauliflower, and mustard—and produces huge quantities of a sticky substance called xanthan gum. This material is made of long chains of sugar units and forms a major part of the biofilm that helps bacteria attach to plant tissues, resist plant defenses, and survive harsh conditions. In the plant, these thick sugar layers can block water flow, leading to dead tissue and the characteristic V-shaped leaf lesions seen in black rot. Interestingly, the same xanthan gum is widely used as a thickener in foods and other products, making this bacterial sugar both an agricultural villain and an industrial asset.
An internal alarm that shapes bacterial behavior
Deep inside many bacteria, a small signaling molecule called ppGpp works like an emergency alarm. When nutrients run low or other stresses arise, ppGpp levels rise and trigger a broad “stringent response” that reshapes growth, metabolism, and survival strategies. Previous work showed that ppGpp can promote biofilm formation in several species and that removing it from Xanthomonas weakens the bacterium’s ability to form biofilms and cause disease. What had remained unclear was how this tiny molecule actually connects to the machinery that builds the thick exopolysaccharide (EPS) coat in this particular plant pathogen.
Tracing the path from signal to sugar
The researchers compared normal bacteria with mutants that cannot make ppGpp. On plates rich in sugar, mutants lacking ppGpp formed smaller, less mucoid colonies, and direct measurements showed they produced significantly less EPS. However, chemical fingerprinting with infrared spectroscopy and imaging with electron microscopy revealed that the basic composition and higher-order shape of the EPS remained the same: the mutants simply made less of it. Attention turned to the “gum” gene cluster, a group of genes that encodes the enzymes assembling xanthan gum. Using gene expression measurements and RNA sequencing, the team found that nearly all gum genes were turned down in ppGpp-deficient cells, placing ppGpp upstream of this cluster in the control hierarchy.
A key middleman: the HpaR1 switch
Between the ppGpp alarm and the gum genes sits a regulatory protein called HpaR1, a transcription factor that binds DNA and boosts gum gene activity. The study showed that ppGpp helps on two fronts. First, cells lacking ppGpp had reduced levels of the hpaR1 gene’s activity, meaning less of the HpaR1 protein was available. Second, in purified laboratory tests, adding ppGpp directly strengthened HpaR1’s grip on the DNA regions controlling the gum genes and even on its own control region. At moderate ppGpp levels this binding was clearly enhanced, though extremely high levels partly weakened the effect, hinting at a finely tuned balance. When the scientists artificially increased HpaR1 levels, EPS production rose again—even in bacteria missing ppGpp—confirming that HpaR1 is the crucial middleman between the alarm signal and the sugar-making machinery.
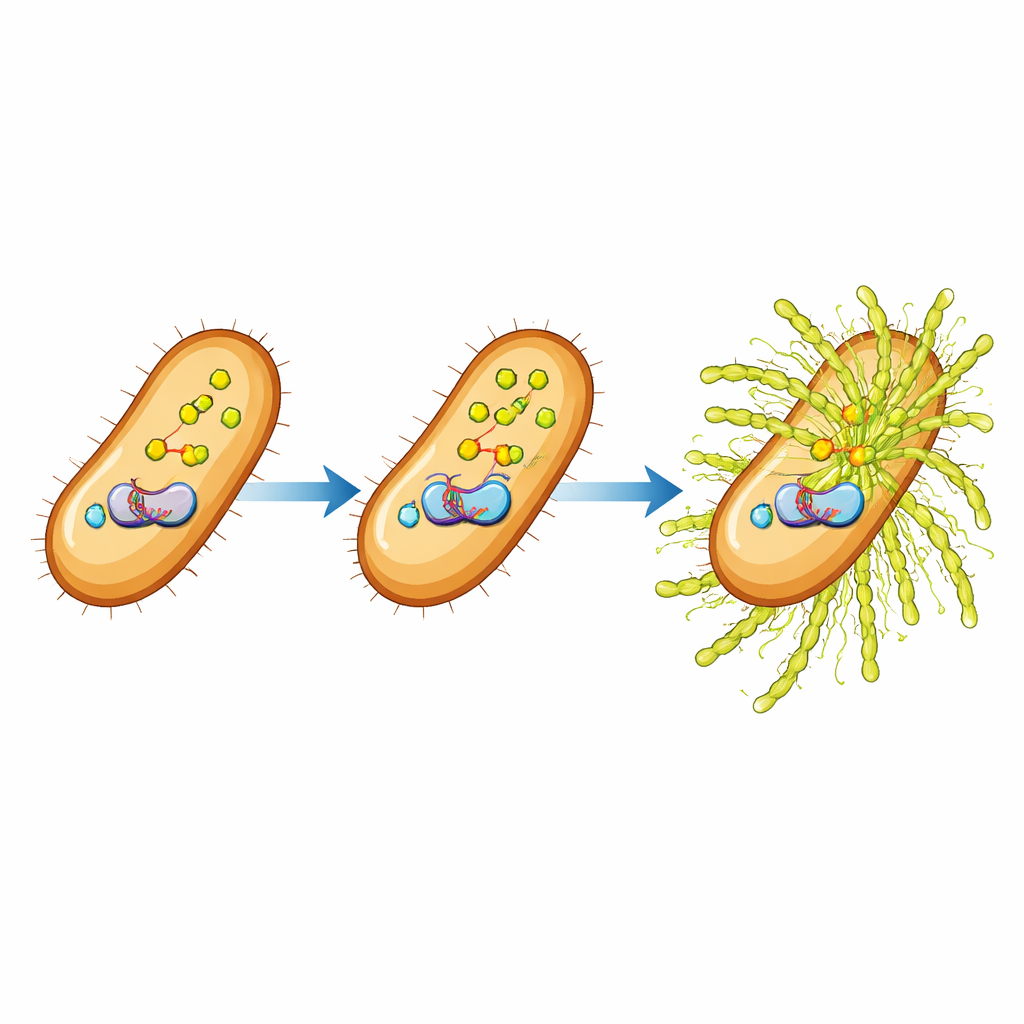
What this control circuit means for crops and beyond
In simple terms, this work uncovers a three-step relay inside a plant pathogen: an internal alarm molecule (ppGpp) boosts and strengthens a DNA-binding switch (HpaR1), which in turn cranks up a sugar-making factory (the gum genes), leading to a thicker protective coat of slime around the bacteria. By mapping this ppGpp–HpaR1–gum pathway in detail, the study explains how environmental stress cues can be converted into changes in biofilm matrix production. For farmers and plant scientists, these insights suggest new targets for disrupting the bacterium’s armor and reducing black rot damage. For microbiologists more broadly, the findings add an important piece to the puzzle of how universal stress signals control the formation of complex microbial communities.
Citation: Bai, K., Xu, X., Yu, C. et al. The ppGpp-HpaR1-gum regulatory pathway modulates exopolysaccharides production in Xanthomonas campestris pv. campestris. npj Biofilms Microbiomes 12, 60 (2026). https://doi.org/10.1038/s41522-026-00926-8
Keywords: bacterial biofilms, plant pathogens, xanthan gum, stress signaling, exopolysaccharides