Clear Sky Science · en
Dietary fiber deficiency exacerbates intestinal inflammation via miR-6240-enriched gut extracellular vesicles
Why the Fiber in Your Diet Matters More Than You Think
A growing number of people eat highly processed foods that are stripped of natural fiber. This study in mice digs into what that missing fiber actually does to the gut. It shows that low-fiber diets don’t just starve friendly microbes; they also trigger tiny particles released from the gut lining that reprogram immune cells and worsen inflammation. Understanding this hidden chain of events helps explain why modern diets are linked to bowel diseases and may point to new ways to treat them.
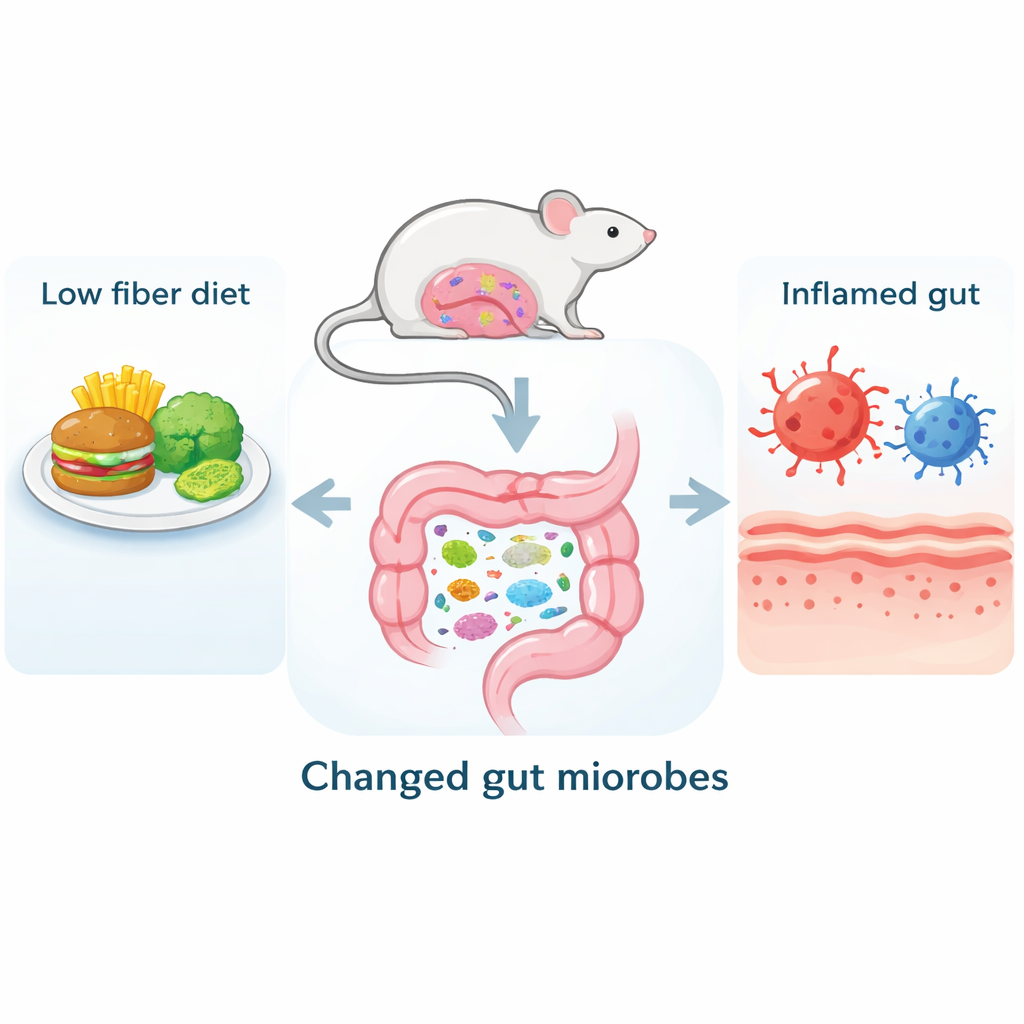
From Empty Plates to an Angry Gut
The researchers started by feeding mice either a normal diet or a fiber-free diet for four weeks. The fiber-deprived mice quickly developed signs of gut trouble: their colons became shorter and more damaged, and the mucus layer that protects the gut surface thinned out. Under the microscope, the tissue showed more injury and fewer mucus-producing cells. Blood and colon samples revealed higher levels of inflammatory molecules and lower levels of calming, protective ones. Immune cells in the gut shifted toward a more aggressive, attack-ready state, and the molecular seals that hold gut cells together weakened, suggesting a leakier intestinal barrier.
Gut Microbes Carry the First Message
Because fiber is a major food source for gut bacteria, the team also examined how the microbial community changed. Mice on the fiber-free diet had a reshuffled mix of microbes: helpful groups such as Bifidobacterium and Prevotella shrank, while several opportunistic or mucus-eating species expanded. To see if microbes alone could cause disease, the scientists transferred gut bacteria from fiber-deprived or normal mice into new animals whose own microbes had been wiped out. Even though these new mice all ate the same standard chow, those receiving bacteria from fiber-free donors developed colon damage, inflammation, and barrier problems similar to the original fiber-deprived animals. This showed that a low-fiber lifestyle can "imprint" a disease-promoting microbiome that is able to transmit gut problems to others.
Tiny Vesicles Turn Diet Signals into Immune Trouble
The study then looked beyond microbes to the body’s own cells. Intestinal lining cells constantly release nanosized bubbles called extracellular vesicles, which carry molecular messages. The authors isolated these vesicles from normal and fiber-free mice and fed them to healthy animals. Vesicles from fiber-deprived guts were enough to trigger colon injury, loss of mucus, higher inflammatory molecules, and weaker barrier proteins, even without changing the animals’ diet. Inside these vesicles, the team found a distinct pattern of small regulatory RNAs, with one in particular—called miR-6240—standing out as strongly increased in the low-fiber condition. When this single RNA was introduced into immune cells in dishes, it pushed them toward a more inflammatory profile and indirectly damaged neighboring gut cells.
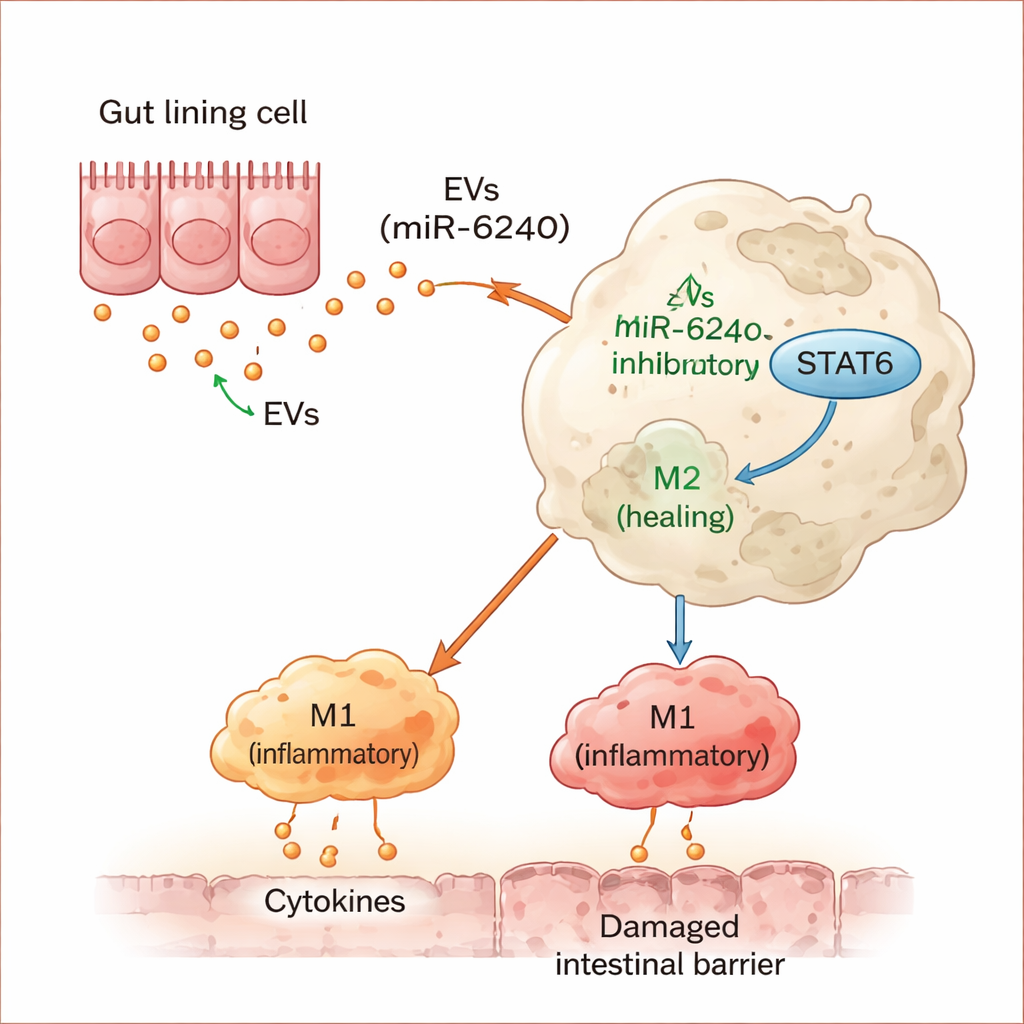
A Single Molecular Switch Tilts the Immune Balance
Digging deeper, the researchers discovered that miR-6240 directly targets a protein called STAT6, which normally helps steer macrophages—key immune cells—toward a healing, "cleanup" mode. By dampening STAT6, miR-6240 blocks this soothing pathway and shifts macrophages toward a harsher, tissue-damaging stance. Giving mice a synthetic form of miR-6240 reproduced many hallmarks of fiber deficiency in their colons, including inflammation, mucus loss, and a weakened barrier. Conversely, blocking miR-6240 in immune cells blunted the harmful effects of vesicles from fiber-deprived guts. When macrophages preloaded with miR-6240 were transferred into healthy mice, those recipients developed gut inflammation, underscoring how powerful this single signal can be.
What This Means for Everyday Eating
For non-specialists, the message is straightforward: skipping fiber doesn’t just change which bacteria live in your gut; it also changes how your own gut cells talk to the immune system. A low-fiber diet reshapes the microbiome and prompts gut lining cells to release vesicles loaded with miR-6240, which in turn turns down a protective switch (STAT6) in immune cells and fans the flames of inflammation. While this particular RNA may be mouse-specific, the general principle likely applies more broadly—what we eat can rewire molecular messages in the gut that decide whether the intestine stays calm or becomes inflamed. Restoring fiber, or targeting similar molecular pathways, could one day help prevent or treat diet-related bowel diseases.
Citation: Song, M., Zhou, W., Fan, J. et al. Dietary fiber deficiency exacerbates intestinal inflammation via miR-6240-enriched gut extracellular vesicles. npj Biofilms Microbiomes 12, 53 (2026). https://doi.org/10.1038/s41522-026-00918-8
Keywords: dietary fiber, gut microbiome, intestinal inflammation, extracellular vesicles, microRNA