Clear Sky Science · en
Oral microbiota–driven immune modulation along the oral–gut axis: from local signals to systemic inflammation
Why Your Mouth Matters to Your Whole Body
The bacteria living in your mouth do much more than cause cavities or bad breath. This review article explains how oral microbes can travel down into the gut, disturb its delicate defenses, and help set off inflammation that reaches organs like the liver and brain. Understanding this hidden “oral–gut axis” offers new clues about common problems ranging from fatty liver disease to Alzheimer’s, and points to surprisingly simple steps—like better dental care and diet—that may protect our overall health.
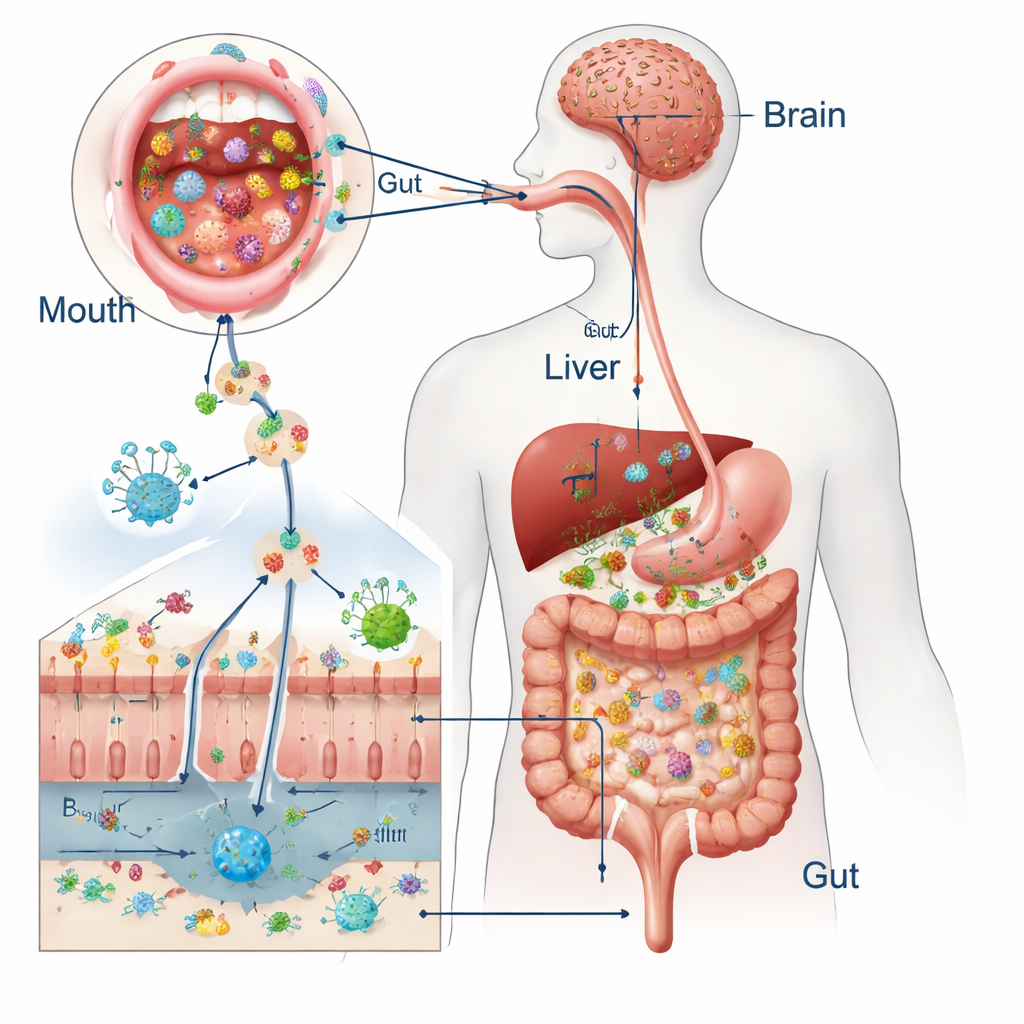
From Swallowing to System-Wide Effects
Every day, we swallow about a liter and a half of saliva packed with billions of mouth-dwelling microbes. In healthy people, strong defenses—saliva’s natural antimicrobials, stomach acid, bile, sticky mucus, and tightly sealed gut cells—keep most of these visitors from settling in. Studies comparing the germs in the mouth and in stool show that only a tiny fraction of gut microbes come directly from the mouth when everything is working well. But the two ecosystems overlap enough that oral species like Streptococcus and Veillonella can sometimes be detected in the intestine, raising the question of when they become more than just harmless passersby.
When Barriers Break Down
The picture changes under stress. Aging, dry mouth, poor oral hygiene, and chronic gum disease can overload the mouth with harmful bacteria. Medications such as antibiotics and stomach-acid blockers weaken gut defenses by thinning the mucus, loosening cell–cell seals, and allowing more microbes to survive the trip through the stomach. Oral infections and dental procedures can even push bacteria into the bloodstream. The review also highlights bacterial extracellular vesicles—nanosized “packages” shed by microbes—that can slip through tissues and carry inflammatory signals far from their source. Some oral bacteria, like Porphyromonas gingivalis and Fusobacterium nucleatum, even hide inside immune cells and hitch a ride through the body in a microbial “Trojan horse.”
How Mouth Bacteria Learn to Live in the Gut
Not every oral microbe that reaches the gut can stay there. Those that do tend to have special survival tricks. Certain strains remodel their cell membranes to withstand strong acid or bile; others feed on sugars from the mucus that coats the intestinal wall or join forces with resident gut bacteria to form clingy biofilms. At the same time, they learn to fly under the radar of the immune system. Some block the killing ability of natural killer cells and T cells; others interfere with early warning sensors that normally detect microbial components and trigger protective responses. By nudging the balance of immune cells toward more inflammatory types and fewer regulatory “brakes,” they create a niche where long-term, low-level colonization becomes possible.
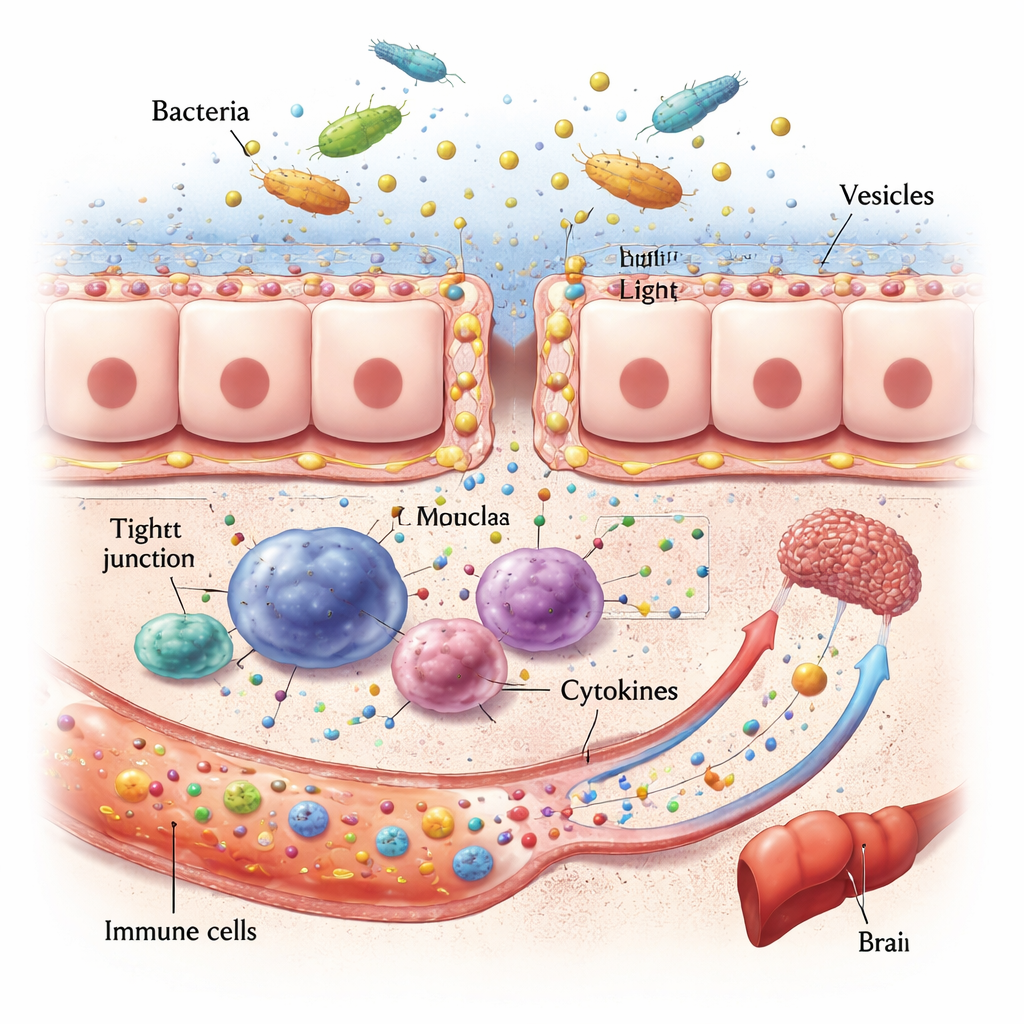
From Leaky Gut to Sick Organs
Once settled, invasive oral microbes and their vesicles can damage the gut’s front line. They chew through the proteins that seal neighboring cells together, thin the mucus layer, and can even kill intestinal cells outright. This makes the gut wall more permeable, allowing bacterial fragments and toxins to seep into the bloodstream. There, they activate immune cells and spark bursts of signaling molecules called cytokines. Because blood from the intestines flows straight to the liver, that organ is especially exposed: animal and human data link oral pathogens with fatty liver disease, scarring, and worsening inflammation. Similar pathways connect oral dysbiosis and gut disruption to the brain, where chronic inflammation and altered microbial metabolites appear to worsen accumulation of Alzheimer’s-related proteins and disturb mood and cognition.
New Ways to Intervene
Recognizing the oral–gut axis opens up multi-level strategies for prevention and therapy. At the mouth, routine periodontal treatment, light-based therapies, and natural antimicrobial peptides can lower harmful bacteria and reduce the number that reach the gut. In the intestine, probiotics, fiber-rich diets, and supplements that restore short-chain fatty acids help rebuild the gut barrier and calm overactive immunity. Looking ahead, sophisticated “organ-on-a-chip” devices that link miniature mouth, gut, liver, and brain tissues, together with big-data maps of which microbes provoke which immune reactions, may enable personalized approaches to preventing chronic inflammatory diseases.
What This Means for You
To a lay reader, the central message is that the mouth is not an isolated compartment: its microbes can shape gut health and, through the gut, influence the entire body. When oral bacteria bypass or erode natural barriers, they can help drive long-lasting inflammation in organs as distant as the liver and brain. The article concludes that protecting oral health, preserving the gut barrier, and gently steering our microbial communities with targeted treatments could become important tools in fighting modern chronic diseases.
Citation: Li, C., Fan, Y. & Chen, X. Oral microbiota–driven immune modulation along the oral–gut axis: from local signals to systemic inflammation. npj Biofilms Microbiomes 12, 46 (2026). https://doi.org/10.1038/s41522-026-00912-0
Keywords: oral microbiome, gut barrier, systemic inflammation, liver disease, brain health