Clear Sky Science · en
Profound taxonomic and functional gut microbiota alterations associated with trichuriasis: cross-country and country-specific patterns
Why tiny gut residents matter for a common parasite
Hundreds of millions of people, mostly in tropical regions, live with a worm called the human whipworm, or Trichuris trichiura. While this parasite is well known for causing stomach troubles and anemia, scientists are now discovering that it also reshapes the vast community of microbes in our intestines. This study takes a deep look at how whipworm infection changes the gut “ecosystem” in people from three different countries, and what that might mean for health and treatment.
A worldwide look inside the gut
Researchers worked in Côte d’Ivoire in West Africa, Laos in Southeast Asia, and Tanzania in East Africa—three regions where whipworm is common. From nearly 9,000 people screened, they selected over 800 individuals, some infected and some not, and collected stool samples. Using a powerful technique called shotgun metagenomics, they read millions of DNA fragments from each sample to identify which bacteria were present and what they were capable of doing. This unified approach let them compare gut communities across very different diets, environments, and lifestyles while keeping the laboratory methods the same.
Different places, different microbes—same overall disturbance
The team found that the overall mix of gut microbes differed strongly among the three countries, even before considering infection. Age, diet, and geography clearly shaped which species were most common. When they compared infected and uninfected people within each country, the picture became more complex. In Laos, infected people actually showed higher microbial diversity, while in Côte d’Ivoire diversity was lower in those with worms, and in Tanzania it stayed about the same. Yet, despite these differences, infection consistently shifted the community’s balance: certain groups of bacteria became more common, others faded, and the way species were connected to each other changed in every country.
Fuel use in the gut shifts from food to the host
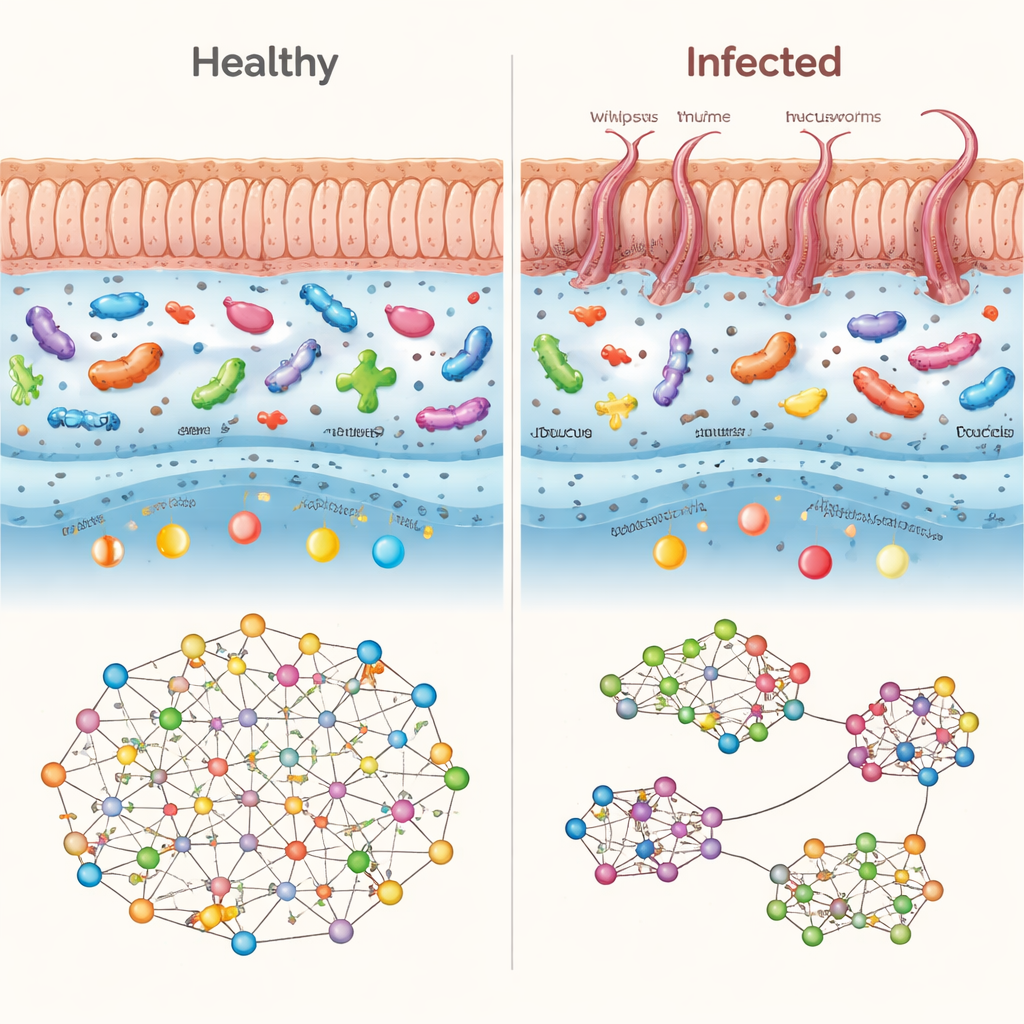
Beyond simply asking “who is there,” the scientists asked “what can they do?” by examining over 6,000 enzyme types and dozens of metabolic pathways. Across regions, people with whipworm showed a loss of microbes and functions that produce short-chain fatty acids (SCFAs)—small molecules made when bacteria ferment dietary fiber. SCFAs like acetate and butyrate help feed cells lining the colon and calm inflammation. Key SCFA-producing species, including certain Blautia and Holdemanella strains, were depleted. At the same time, microbes that specialize in eating the mucus layer that coats the gut, such as Ruminococcus and Bacteroides, became more common. Metabolic pathways for building protective bacterial sugars and exopolysaccharides declined, while pathways for breaking down host-derived sugars and mucins increased. In essence, the microbial community appeared to switch from living mainly on food passing through the gut to mining nutrients from the host’s own protective lining.
Fragile microbial networks and a new set of winners
To understand how these changes affect the stability of the gut ecosystem, the researchers mapped networks of “who co-occurs with whom” among bacterial species. In uninfected people, familiar gut bacteria such as Streptococcus, Clostridium, Dorea, and Blautia formed well-connected hubs, suggesting a cooperative, resilient community. In infected individuals, these hubs weakened or moved to the margins, and other species—most notably Segatella copri—rose to central positions. Overall, infected networks became more clustered but less globally connected, resembling a set of small islands rather than a single continent. Such fragmentation is often a sign of an ecosystem under stress and may make it harder for the gut to resist further disturbances like additional infections or dietary shifts.
What this means for people living with whipworm
For non-specialists, the message is that whipworm infection is not just a problem of a single parasite burrowing into the gut wall. It also appears to push the gut’s microbial community toward a state that thins the protective mucus layer, reduces beneficial fermentation products, and destabilizes microbial relationships. These combined effects may help the worm persist while leaving the host more vulnerable. Because these patterns were seen, in different forms, across Africa and Asia, the authors suggest that microbiome-based strategies—such as targeted probiotics, prebiotic fibers, or diet changes—could someday complement existing drugs. By restoring a healthier microbial balance and strengthening the gut barrier, such approaches might make it easier to clear infections and protect people in areas where whipworm remains a daily reality.
Citation: Schneeberger, P.H.H., Dommann, J., Rahman, N. et al. Profound taxonomic and functional gut microbiota alterations associated with trichuriasis: cross-country and country-specific patterns. npj Biofilms Microbiomes 12, 45 (2026). https://doi.org/10.1038/s41522-026-00911-1
Keywords: gut microbiome, Trichuris trichiura, parasitic infection, short-chain fatty acids, intestinal health