Clear Sky Science · en
Enzymes-enhanced antibiotic therapy reduces biofilms to undetectable levels in an implant-associated infection model
Why stubborn implant infections matter
Hip and knee replacements transform lives, but when bacteria colonize these metal implants they can form slimy communities called biofilms. These biofilms act like armored cities that shield germs from both the immune system and antibiotics, leading to painful, long‑lasting infections that are very hard to cure. This study explores a new, two‑pronged treatment that first helps dismantle the biofilm’s defenses and then bathes the area in high doses of antibiotics—an approach that could one day help save more implants instead of replacing them.
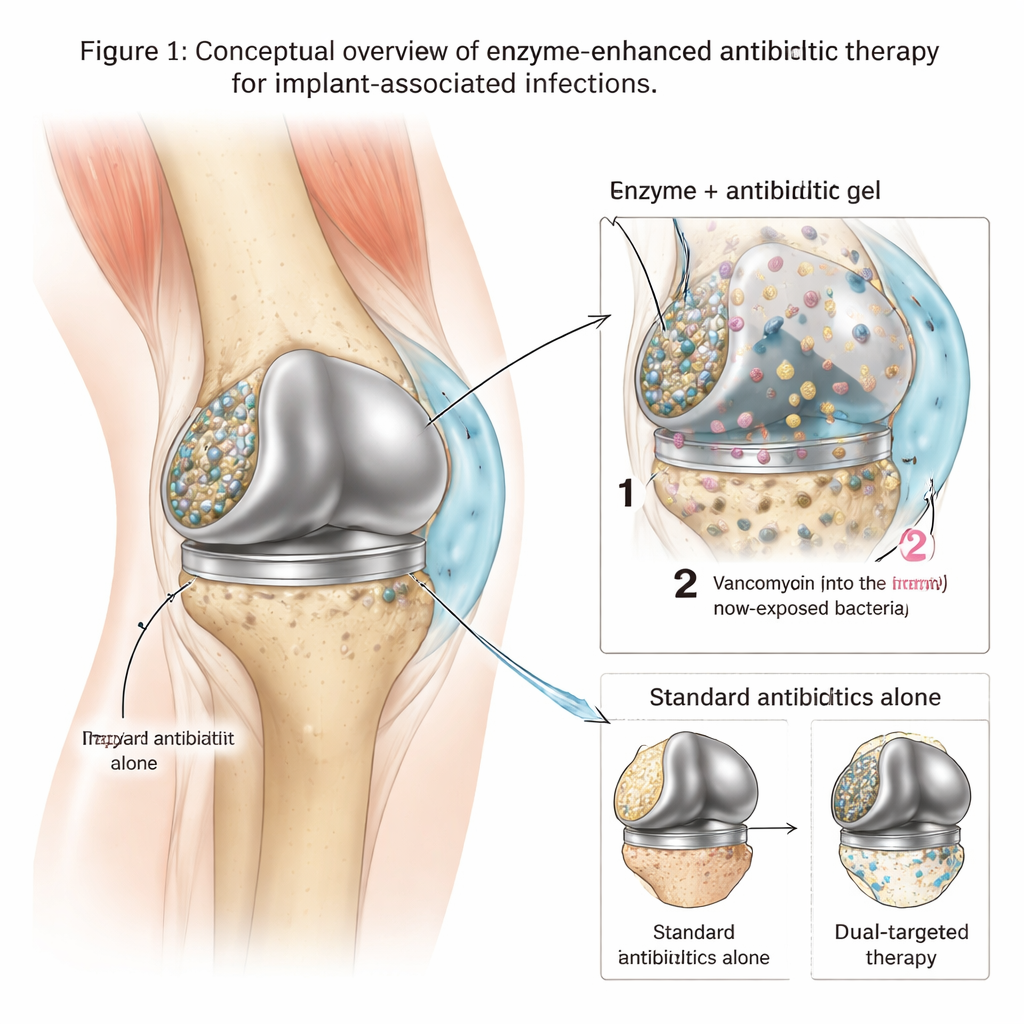
The hidden strongholds on metal implants
When bacteria such as methicillin‑resistant Staphylococcus aureus (MRSA) attach to an artificial joint, they quickly build a sticky matrix of DNA, sugars, and other molecules on the metal surface. Inside this shielded biofilm, bacteria grow slowly, change their behavior, and can tolerate antibiotic levels hundreds or even thousands of times higher than those that kill free‑floating germs. As a result, standard treatments—even strong drugs like vancomycin—often fail to fully clear the infection, forcing patients to undergo repeated surgeries or complete implant replacement. Doctors need tools that not only kill bacteria, but also break open their protective fortress so the drugs can reach them.
A smart gel that delivers enzymes and antibiotics
The researchers designed a local treatment built around a thermosensitive hydrogel made from a compound called poloxamer 407. This gel is liquid when cold, making it easy to inject around an implant, but quickly turns into a soft solid at body temperature, helping it stay in place. Into this gel, the team loaded two key ingredients: a powerful antibiotic (vancomycin) and a cocktail of three enzymes that cut up major components of the biofilm matrix. In lab tests, they tuned the gel so that the enzymes are released first—loosening and thinning the biofilm—while vancomycin is released more slowly over several days, maintaining very high local drug levels right where the bacteria live.
From lab dishes to guinea pigs
To see whether the strategy really worked, the team tested it first on biofilms grown on titanium pieces in the lab and then in a guinea‑pig model that mimics infected implants. In vitro, two applications of the enzyme‑antibiotic gel, 48 hours apart, reduced the number of live bacteria on metal surfaces by more than 100,000‑fold and nearly wiped out the biofilm mass. In animals, the researchers surgically placed perforated cages containing titanium beads under the skin, infected them with MRSA, and then treated them with different combinations of systemic antibiotics and local gels. The most effective regimen combined: (1) the local gel containing both enzymes and vancomycin, given twice, and (2) body‑wide treatment with vancomycin plus a second antibiotic, rifampicin, which is especially good at penetrating biofilms.
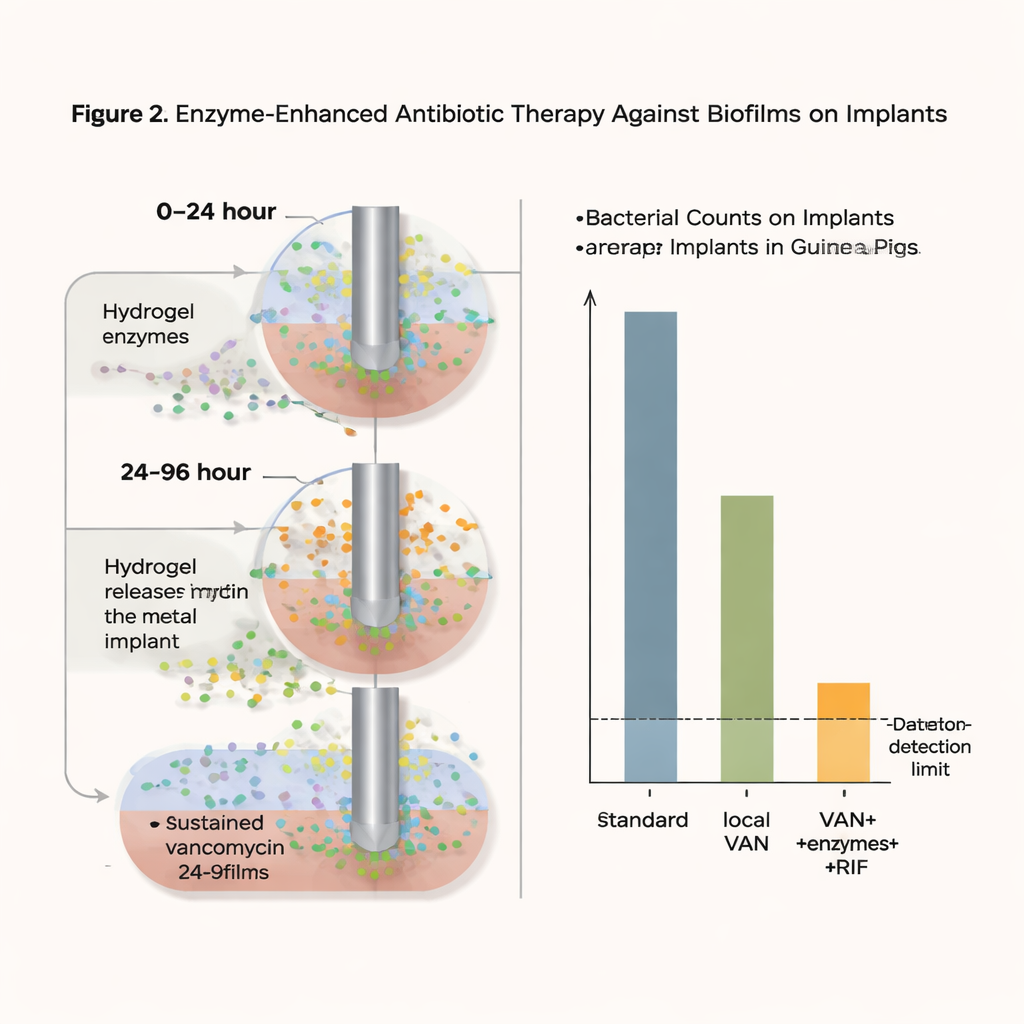
What the new approach achieved
With this intensive but localized strategy, bacterial counts on the implant material dropped to the detection limit—effectively “no recoverable bacteria” in this model—in 75 percent of treated cages one day after therapy ended, and in 37.5 percent even five days later. Importantly, no increase in antibiotic resistance to vancomycin was seen, and high local vancomycin levels appeared to prevent the emergence of rifampicin‑resistant strains. Safety tests using cultured cells and a reconstructed human skin model found no signs of toxicity or irritation from the gel formulations. While a mild regrowth of bacteria still occurred in some cases and the treatment period was shorter than what patients typically receive, the results show that combining biofilm‑disrupting enzymes with sustained local antibiotic delivery can dramatically shrink otherwise stubborn infections.
What this could mean for patients
For people facing implant‑related infections, this work points toward a future in which surgeons might inject a smart gel around an infected joint, rather than immediately removing or replacing the hardware. By first weakening the biofilm’s shield and then flooding the area with high, sustained doses of antibiotics—backed up by whole‑body treatment—this dual‑targeted strategy aims to push bacterial numbers down to undetectable levels and reduce the risk of resistance. Although more studies, longer treatments, and human trials are still needed, the approach offers a promising blueprint for turning some of the most persistent orthopedic infections into more manageable, and potentially curable, problems.
Citation: Buzisa Mbuku, R., Poilvache, H., Maigret, L. et al. Enzymes-enhanced antibiotic therapy reduces biofilms to undetectable levels in an implant-associated infection model. npj Biofilms Microbiomes 12, 44 (2026). https://doi.org/10.1038/s41522-026-00910-2
Keywords: biofilm infection, orthopedic implants, vancomycin, enzymatic therapy, rifampicin