Clear Sky Science · en
A theoretical exploration of protocols for treating prosthetic joint infections with combinations of antibiotics and bacteriophage
Why joint implant infections matter
Hip and knee replacements have transformed life for millions, letting people walk without pain. But when bacteria colonize these artificial joints, they can form slimy communities called biofilms that cling tightly to metal and plastic surfaces. Once a biofilm is established, normal doses of antibiotics and the body’s immune defenses often struggle to wipe it out, sometimes forcing patients to undergo more surgery. This study asks whether adding viruses that specifically attack bacteria—called bacteriophages, or phages—to standard antibiotics can offer a better way to treat these stubborn infections.
Tiny viruses that hunt bacteria
Phages are natural predators of bacteria. They latch onto a bacterial cell, inject their genetic material, turn the cell into a virus factory, and finally burst it open, releasing a swarm of new phages. Some phages also carry enzymes that can chew through the sticky material surrounding biofilms, potentially opening up protective layers that shield bacteria from drugs. Because of these features, many researchers have hoped that combining phages with antibiotics could provide a powerful one-two punch against prosthetic joint infections, especially when bacteria become resistant to the drugs.
Building infections inside a computer
Instead of going straight to animal experiments or clinical trials, the authors first built a detailed mathematical and computer model of a prosthetic joint infection. Their virtual system had two main zones: free-floating bacteria in body fluids and bacteria embedded in a biofilm on the implant surface. The model also tracked immune cells, nutrients for bacterial growth, antibiotics, and phages, all moving into and out of the joint. By changing conditions such as drug dose, timing, and the presence of antibiotic-resistant bacteria, they could explore many treatment strategies that would be difficult, slow, or expensive to test in real patients.
When medicines work alone and together
The simulations showed that neither the immune system nor antibiotics alone easily cleared biofilm infections. Antibiotics strongly reduced free-floating bacteria but barely dented the biofilm population. Phages could initially control the infection more effectively, but they introduced boom-and-bust cycles: as phages killed bacteria, their prey became too scarce, causing phage numbers to fall and allowing bacteria to rebound. When antibiotics and phages were given at the same time, the antibiotic tended to dominate the outcome, and adding phage often did little more than the antibiotic could accomplish on its own.
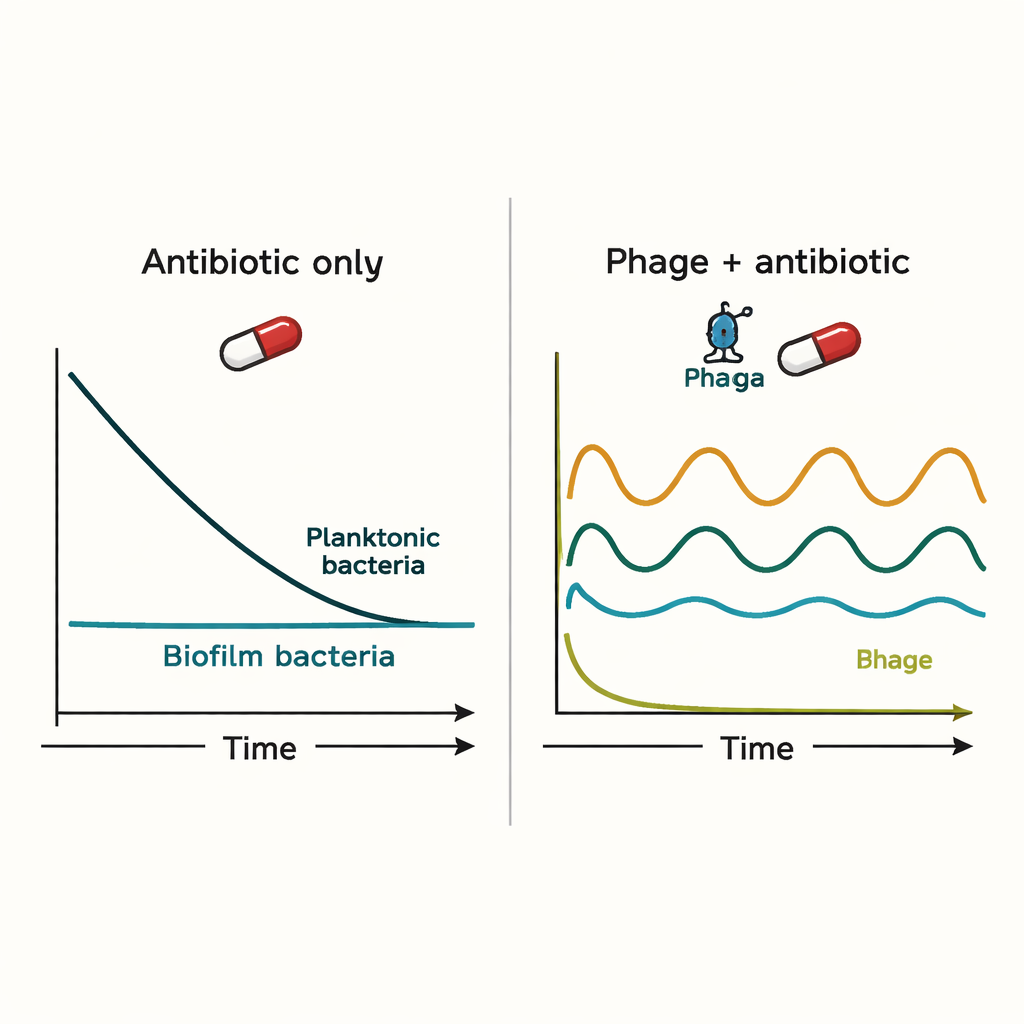
The special role of timing and resistance
The model became more hopeful when antibiotic-resistant bacteria were included. Phages were able to attack these resistant cells, sometimes removing them entirely. If phage treatment was started first and antibiotics were added several hours later, the combined strategy reduced free-floating bacteria by many orders of magnitude and eliminated resistant subpopulations in the simulations. Even so, the biofilm-associated bacteria were not fully cleared; instead, their numbers were held at lower levels that might be easier for the immune system to manage. This suggests that phages may be most useful not as magic bullets, but as helpers that prevent resistance and keep infections in check.
What this means for patients and doctors
For people facing an infected joint implant, this work offers both caution and promise. The model suggests that simply adding phages to standard antibiotics is unlikely to cure deep biofilm infections by itself. However, phages could still be valuable tools: they may help wipe out antibiotic-resistant bacteria and reduce the total number of germs enough for the body to cope, especially if used before or carefully timed with antibiotics. The authors emphasize that these are theoretical predictions, not clinical proof. Their main message is that thoughtful experiments—guided by models like this one—are needed to discover when phage therapy can genuinely improve outcomes for patients with prosthetic joint infections.
Citation: Levin, B.R., Gil-Gil, T., Berryhill, B.A. et al. A theoretical exploration of protocols for treating prosthetic joint infections with combinations of antibiotics and bacteriophage. npj Biofilms Microbiomes 12, 51 (2026). https://doi.org/10.1038/s41522-025-00908-2
Keywords: prosthetic joint infection, biofilm, bacteriophage therapy, antibiotic resistance, mathematical modeling