Clear Sky Science · en
Molecular insight into transcriptome profiling of aerobic exercise induced changes in aged skeletal muscle
Why this matters for healthy aging
As we grow older, our muscles gradually lose size and strength, a process that can lead to frailty and loss of independence. Doctors know that exercise helps older adults stay stronger, but what actually changes inside aging muscles when we work out has been unclear. This study uses a powerful gene-reading technique to look deep inside the muscles of older mice that performed regular aerobic treadmill exercise, revealing how thousands of genes respond and how these changes may help preserve muscle function and even brain health.
Muscles in later life: what goes wrong
The researchers first compared muscles from "young" mice to those from much older mice that did not exercise. Older muscles showed large shifts in the activity of more than 40,000 RNA molecules, which are the working copies of genes inside cells. Many of the changes were linked to the machinery that makes muscles contract and to the structure that holds muscle fibers together. The pattern supported what is already known from human studies: with age, we lose more of the fast, powerful fibers than the slower, more endurance-focused ones, and the connections between nerves and muscles become less reliable. They also saw reduced activity in genes that help regulate electrical signals and energy use, hinting at declines in how efficiently aging muscles handle movement and metabolism.
Putting old muscles on a treadmill
Next, the team asked what happens when old mice spend eight weeks doing regular treadmill running, similar to a human starting a consistent walking or light jogging routine in their seventies. After this aerobic training, the older "intervention" muscles showed striking shifts in gene activity compared with untrained old muscles. Hundreds of genes became more active, including many involved in the cell surface, the material surrounding muscle fibers known as the extracellular matrix, and pathways that regulate inflammation and tissue repair. One key gene, called Slpi, which helps control destructive enzymes and calm damaging inflammation, was strongly boosted by exercise. At the same time, some genes tied to breaking down damaged protein complexes and to stress responses were turned down, suggesting that well-trained aging muscles may experience less internal stress.
Rewiring communication between muscle, nerves, and the body
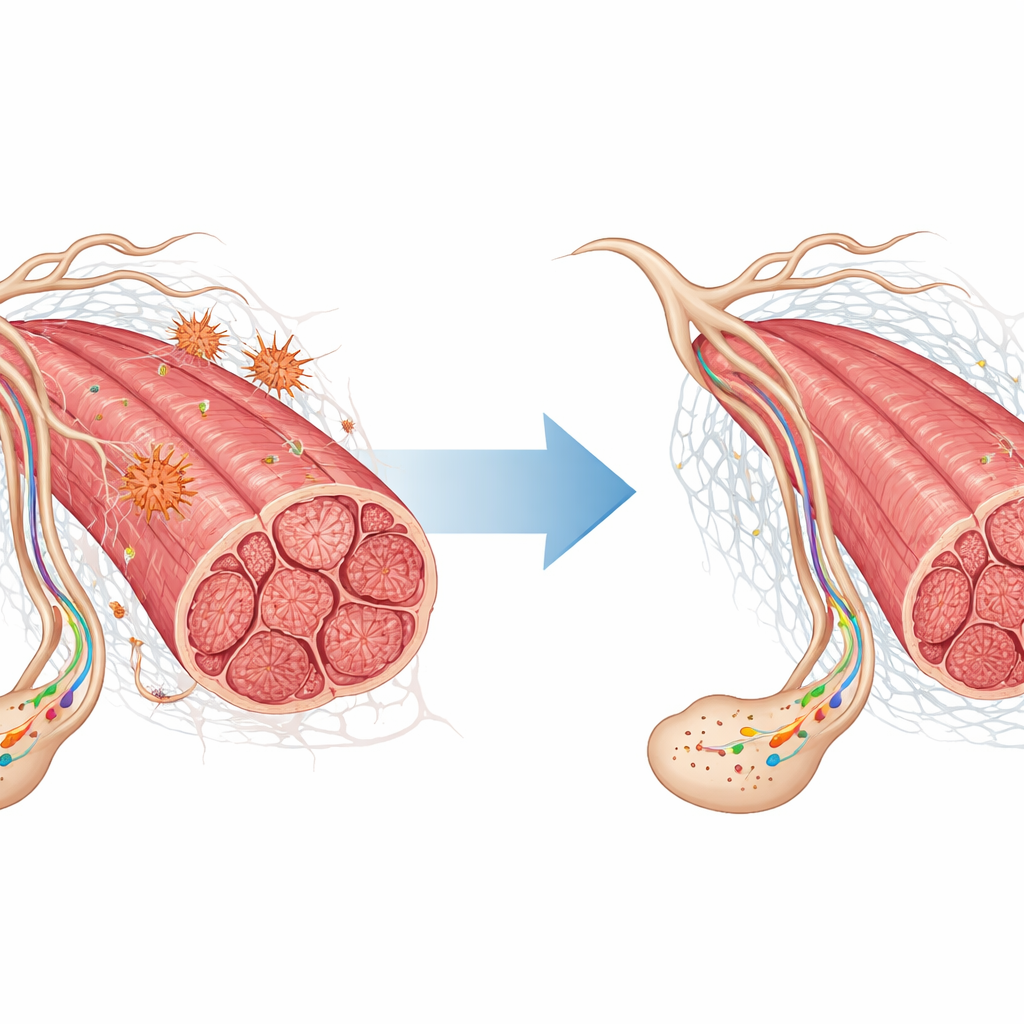
When the scientists compared exercised old muscles not just with old, but also with young muscles, another story emerged. Exercise in old age did not simply "reset" genes to a youthful pattern; instead, it created a distinct, adaptive profile. Many activated genes were linked to nerve signaling, including receptors involved in communication at synapses—the contact points where nerves talk to muscle fibers and to each other. Pathways related to neurotransmitter receptors and signal transmission became more active, which may help stabilize the aging neuromuscular junction and support better coordination and strength. At the same time, genes normally switched on when muscles are inactive or have lost nerve input were dialed down, consistent with the idea that regular movement helps prevent disuse and denervation signals from taking over.
Hidden players: non-coding RNAs and tissue scaffolding
Beyond classic protein-coding genes, the study highlighted several non-coding RNAs—small RNA molecules and long intergenic RNAs that do not make proteins but can subtly tune many other genes. Some of these appeared only in old muscles, while others rose or fell with exercise, marking them as potential switches that control how muscles adapt to both aging and training. Exercise also reshaped genes tied to the extracellular matrix, the supportive meshwork around muscle fibers. These changes may help maintain the physical integrity of muscle tissue, improve how forces are transmitted during movement, and influence signals traveling between muscle, bone, immune cells, and even the brain.
What this means for staying strong as we age
In everyday terms, this work shows that steady aerobic exercise in later life does more than just keep muscles looking fit—it rewrites the molecular script inside aging muscle cells. The gene activity patterns suggest that regular training helps old muscles damp down harmful inflammation, reinforce their structural scaffolding, fine-tune nerve–muscle communication, and recruit lesser-known RNA regulators to support adaptation. Although the study was done in male mice, and more work is needed in humans and in females, it strengthens the message that it is never too late to move: consistent aerobic exercise can drive deep, beneficial changes in aging muscles that may help combat conditions like sarcopenia and preserve function and quality of life.
Citation: Anwar, M., Kaur, P., Gupta, D. et al. Molecular insight into transcriptome profiling of aerobic exercise induced changes in aged skeletal muscle. npj Aging 12, 45 (2026). https://doi.org/10.1038/s41514-026-00336-2
Keywords: sarcopenia, aerobic exercise, skeletal muscle aging, gene expression, treadmill training