Clear Sky Science · en
Multi-layered molecular profiling informs the diagnosis and targeted therapy of desmoplastic small round cell tumor
A rare cancer in need of better answers
Desmoplastic small round cell tumor (DSRCT) is an extremely rare and aggressive cancer that mostly affects children, teenagers, and young adults. Because it is so uncommon and can mimic other tumors under the microscope, doctors often struggle to diagnose it correctly and to choose effective treatments. This study asks a simple but powerful question: if we deeply analyze DSRCT tumors at many molecular levels—not just DNA, but also RNA, protein patterns, and chemical tags on DNA—can we both sharpen the diagnosis and uncover new, more precise treatment options for patients who have already run out of standard therapies?
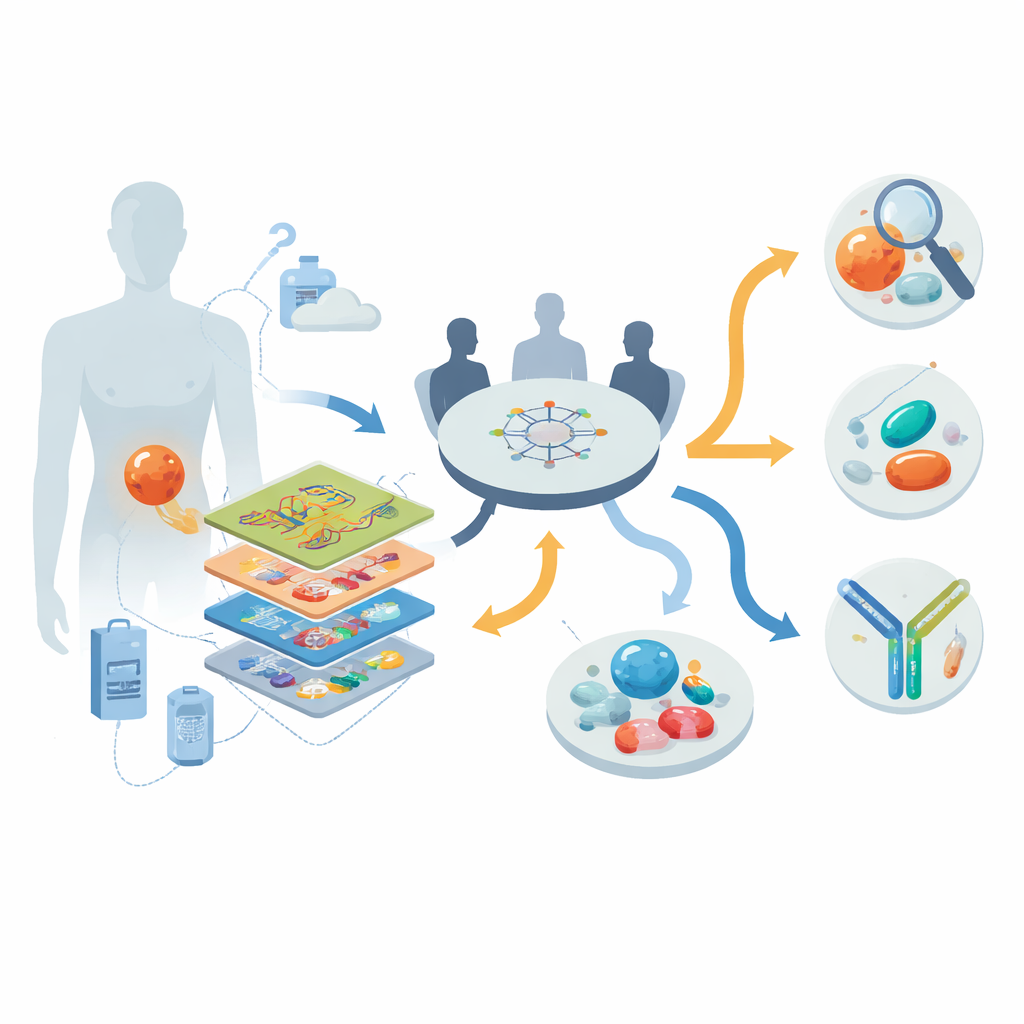
Looking inside tumors layer by layer
The researchers worked within a nationwide precision oncology program in Germany, enrolling 30 people with advanced, mostly treatment-resistant DSRCT between 2013 and 2022. For each patient they applied a “multi-omics” toolkit: whole-genome or exome sequencing to study DNA; RNA sequencing to see which genes were actively switched on; DNA methylation profiling to capture epigenetic fingerprints; and, in nine patients, detailed protein and phosphoprotein measurements that reveal which signaling pathways are actually active. A multidisciplinary tumor board then reviewed these data together with each patient’s clinical history to generate individualized recommendations for diagnosis clarification, targeted drugs, and clinical trials.
Fixing mistaken diagnoses and revealing hidden targets
One striking finding was how often deep profiling corrected the original diagnosis. In eight out of 30 patients, tumors initially labeled as other entities—such as cancers of unknown origin or vaguely defined sarcomas—were reclassified as DSRCT once a hallmark gene fusion (EWSR1::WT1) and a characteristic methylation pattern were identified. At the same time, the DNA of these tumors turned out to be relatively quiet: there were few mutations and only occasional changes in copy number, so classic “mutated gene = drug target” approaches yielded little. Instead, the richest clues came from RNA and protein layers. By comparing gene activity in DSRCT with hundreds of other sarcomas, the team found repeated overexpression of several surface molecules and signaling enzymes that could, in principle, be attacked with existing or emerging drugs.
From molecular signals to tailored treatments
Based on these patterns, the tumor board issued 107 molecularly informed management suggestions for 28 of the 30 patients. Most recommendations involved drugs that block tyrosine kinases—key enzymes that drive cell growth and blood vessel formation—or newer strategies that latch onto highly expressed surface proteins. Examples included small-molecule kinase inhibitors such as pazopanib; peptide-based radiation aimed at somatostatin receptors; experimental CAR T cells targeting the adhesion molecule CLDN6; and antibody–drug conjugates that deliver toxins to cells bearing the ERBB2 (HER2) receptor. Seventeen patients were considered eligible for biomarker-matched clinical trials, underscoring how detailed molecular data can open doors to studies that would otherwise be hard to access for people with an ultra-rare disease.
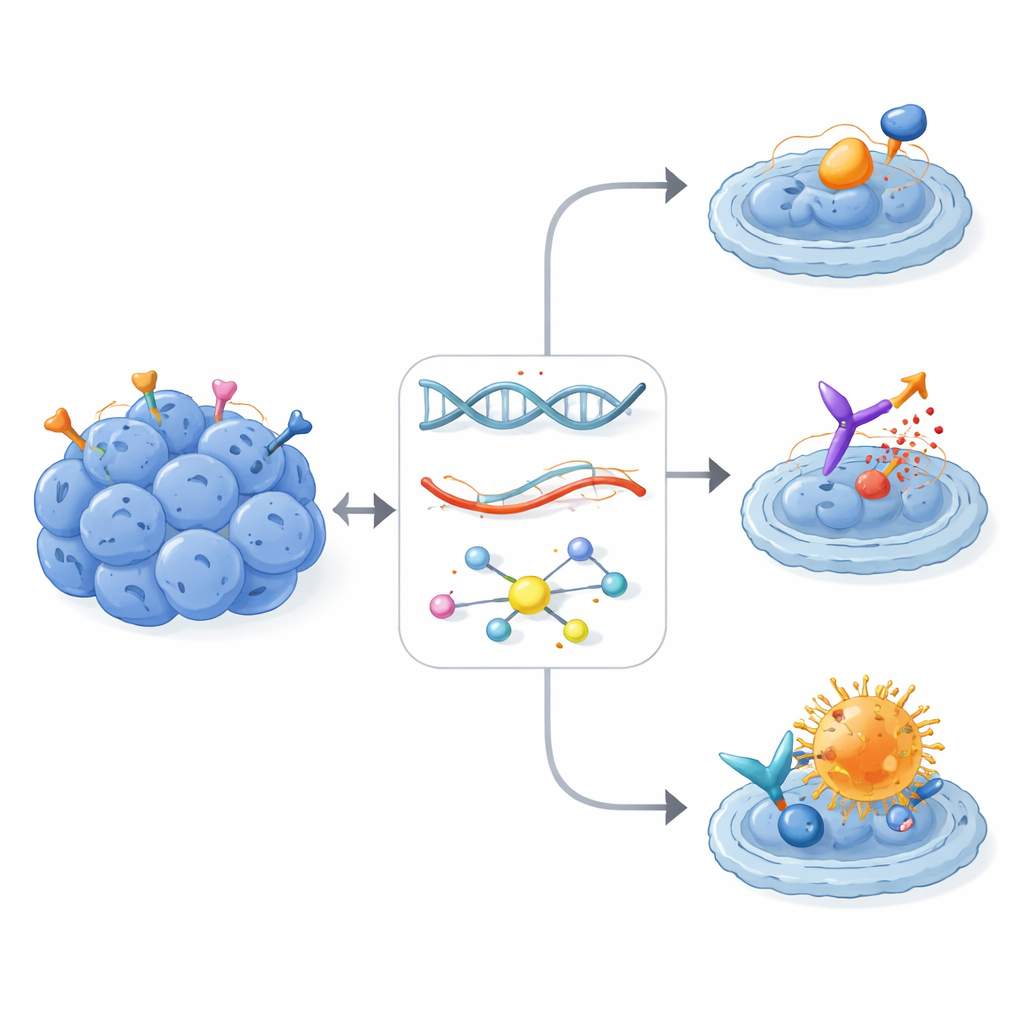
Real-world impact in heavily pretreated patients
Of all the recommended personalized therapies, 16 were actually given to 13 patients, all guided by RNA-level signals and sometimes refined by phosphoprotein data. Even though these individuals had already been through multiple rounds of chemotherapy and local treatments, eight of the 13 achieved disease control—five with partial tumor shrinkage and three with stable disease. Multi-target tyrosine kinase inhibitors, most often pazopanib, produced meaningful and sometimes long-lasting benefit in several patients, especially when the drug’s known targets were clearly overactive in the tumor. Perhaps most notable were two patients with high ERBB2 expression who received the antibody–drug conjugate trastuzumab deruxtecan (T-DXd). Both experienced prolonged responses lasting up to two years or more, despite having exhausted many previous therapy lines and despite the ERBB2 receptor not showing the strong activation pattern typically required for classic ERBB2-blocking drugs.
What this means for patients and future care
For people with DSRCT, the study delivers a cautiously optimistic message. Although this cancer has few obvious DNA mutations to exploit, looking across multiple molecular layers can both correct misdiagnoses and reveal actionable weaknesses that are invisible to standard tests. The work shows that, even in a rare and aggressive disease, a biology-guided approach can produce real clinical benefit, including durable responses to drugs like pazopanib and trastuzumab deruxtecan. More broadly, it argues that patients with DSRCT—and by extension other ultra-rare cancers—stand to gain from routine access to comprehensive molecular profiling and expert review, paving the way for future trials in which treatments are chosen not by tumor type alone, but by the unique molecular fingerprints of each person’s cancer.
Citation: Renner, M., Oleś, M., Paramasivam, N. et al. Multi-layered molecular profiling informs the diagnosis and targeted therapy of desmoplastic small round cell tumor. Nat Commun 17, 3397 (2026). https://doi.org/10.1038/s41467-026-71636-0
Keywords: desmoplastic small round cell tumor, precision oncology, multi-omics profiling, targeted therapy, antibody drug conjugates