Clear Sky Science · en
Interplay of SLC33A1-dependent and -independent Golgi sialic acid O-acetylation in CASD1 catalysis
How tiny sugar tweaks can shape health and disease
Our cells are coated with complex sugar chains that act like molecular ID cards, guiding immunity, development, and even how viruses latch on. This study explores a subtle but powerful tweak to those sugars—adding small chemical “caps” called acetyl groups to a terminal sugar named sialic acid—and uncovers a surprising two-part molecular machine that controls this process inside the cell’s Golgi apparatus. Understanding this hidden system helps explain certain severe neurological disorders and offers clues to how cells fine‑tune their surfaces in health and disease. 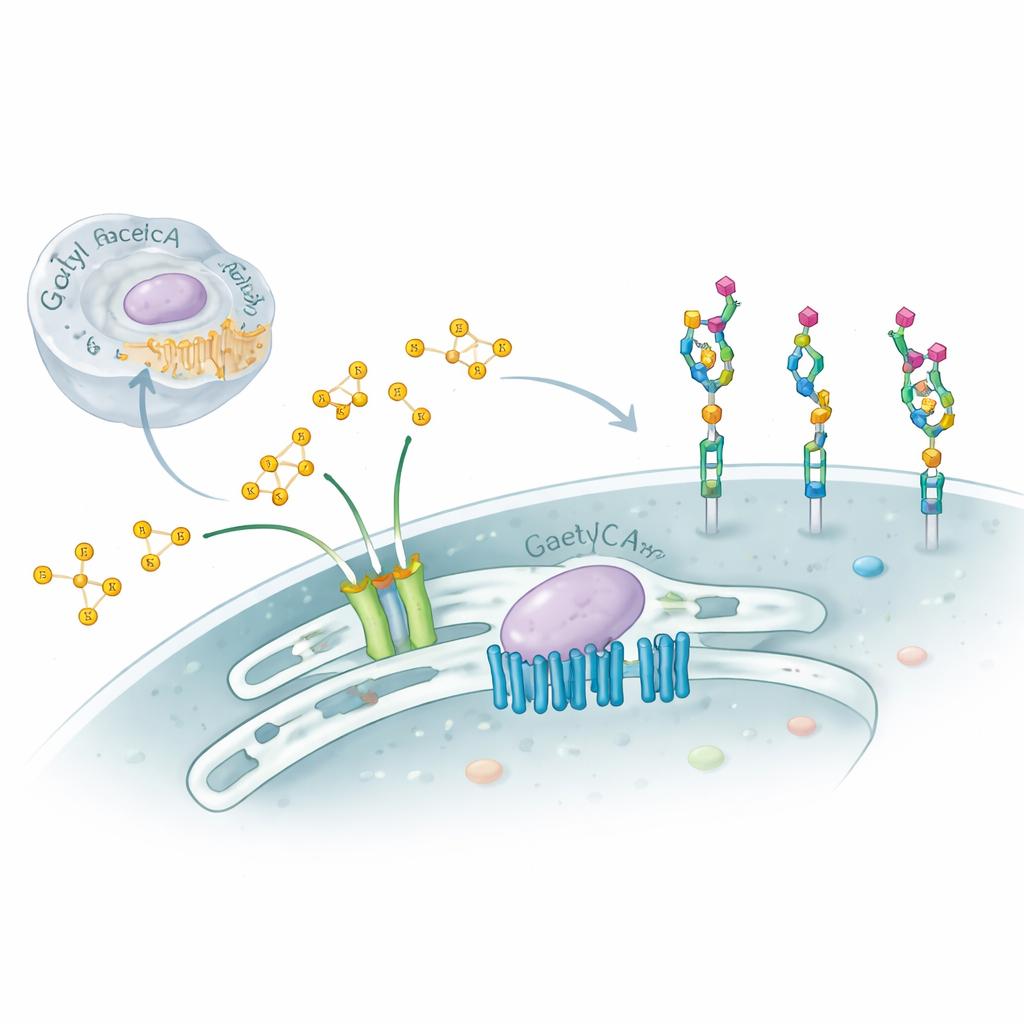
The special sugars at the cell’s surface
Sialic acids sit at the outer tips of many cell-surface sugars on proteins and lipids, particularly on molecules called gangliosides in the brain. By slightly changing sialic acids—such as adding one or two acetyl groups at specific positions—cells can mask existing recognition sites or create new ones. Viruses like influenza C and some coronaviruses specifically seek out these modified sugars, and acetylated gangliosides can influence whether nerve cells survive or die, as well as how cancers grow. Yet, despite their importance, the inner workings of the cell machinery that places these acetyl groups in the Golgi have remained poorly understood.
A transporter and an enzyme work hand in hand
The researchers focused on two proteins that reside in or around the Golgi: SLC33A1, a transporter that moves acetyl‑CoA (the cell’s main acetyl donor) across membranes, and CASD1, an enzyme known to transfer acetyl groups onto sialic acids. Using gene editing in human and hamster cell lines, they removed SLC33A1 and watched what happened to specific gangliosides. Cells without SLC33A1 could still make the basic gangliosides GD3 and GD2, but completely lost their 9‑O‑acetylated forms, a hallmark modification linked to cancer and neural function. Re‑introducing human SLC33A1 restored these acetylated gangliosides, showing that SLC33A1 is a crucial supplier of acetyl‑CoA for this pathway. 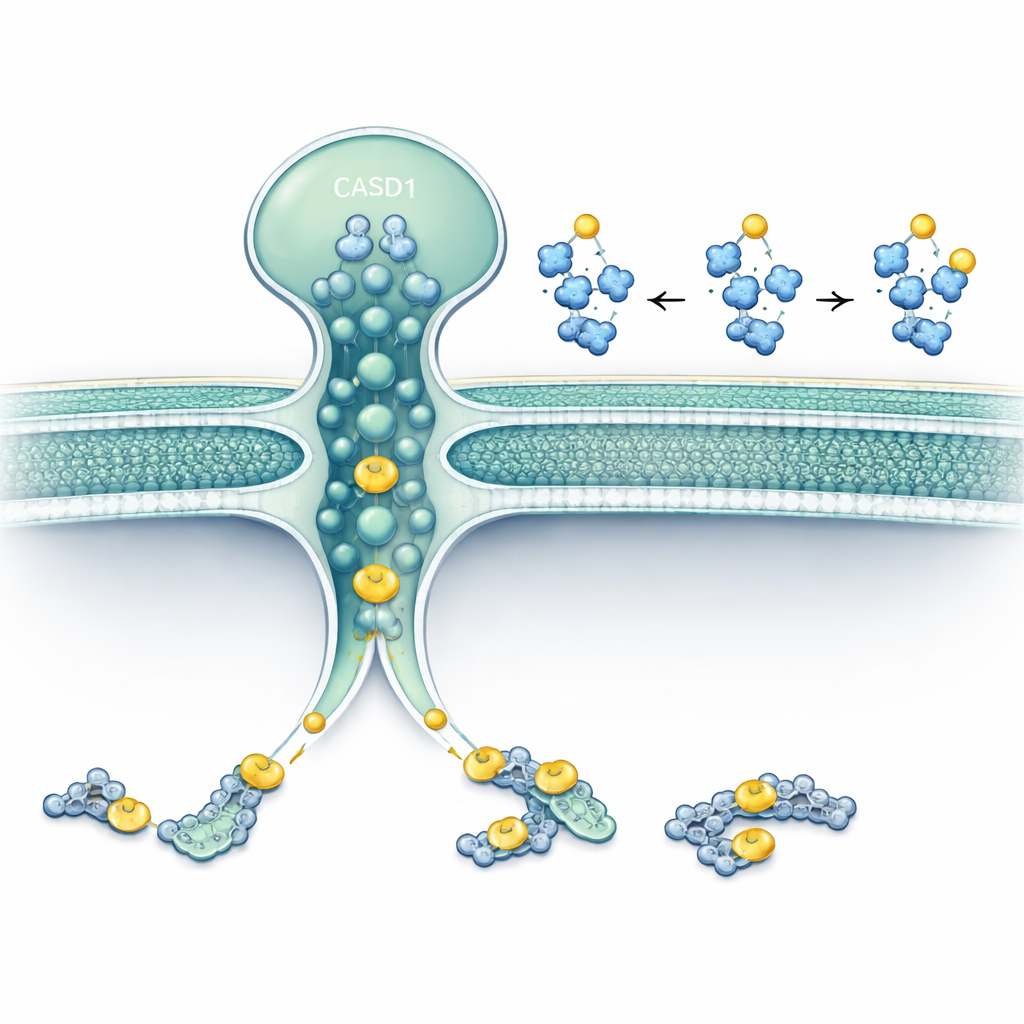
Patient mutations reveal medical relevance
SLC33A1 has been implicated in several rare neurological conditions, including a severe childhood disorder (Huppke–Brendel syndrome), a form of hereditary spastic paraplegia, and late‑onset cerebellar ataxia. The team recreated four patient‑derived SLC33A1 variants in cells lacking the normal transporter and tested whether these altered proteins could rescue ganglioside acetylation. All four showed reduced function, and the most severe childhood‑onset mutations barely restored any acetylation at all. Guided by a recently solved three‑dimensional structure of SLC33A1, the authors mapped these mutations onto the protein and showed how they likely disrupt its architecture or its ability to bind acetyl‑CoA. This strongly suggests that impaired ganglioside acetylation contributes to the neurological symptoms seen in patients.
A hidden second active site in CASD1
The work also uncovered an unexpected twist: CASD1 itself appears to be a dual‑purpose enzyme. Its known portion in the Golgi lumen uses acetyl‑CoA delivered by SLC33A1. But when SLC33A1 was knocked out, cells still managed to place a single acetyl group on certain sialic acids under some conditions, although they failed to add the second acetyl group needed for more complex patterns. Using advanced structure prediction and molecular simulations, the authors showed that the membrane‑spanning part of CASD1 closely resembles a known transmembrane acetyltransferase from lysosomes. They pinpointed key amino acids in this region that bind acetyl‑CoA on the cytosolic side and pass its acetyl group through the membrane directly to sialic acid. When these residues were mutated, CASD1 lost this second activity in both test‑tube assays and living cells.
Two routes, one finely tuned sugar code
Together, the data support a “two‑catalytic‑site” model. One route depends on SLC33A1 bringing acetyl‑CoA into the Golgi lumen, where the luminal domain of CASD1 adds an acetyl group to sialic acid, enabling both simple and double acetylation patterns. The other route is built into the membrane-spanning core of CASD1, which taps directly into cytosolic acetyl‑CoA and can still perform a basic level of acetylation even when SLC33A1 is missing. For a layperson, this means that a single enzyme, fed by two different acetyl supplies, writes subtle “punctuation marks” onto cell-surface sugars. When the transporter or either part of the enzyme is faulty, these marks are misplaced or missing, which may help explain certain neurodevelopmental and neurodegenerative diseases and highlights a new layer of regulation in how our cells shape their outer identity.
Citation: Albers, M., Bosse, L., Schröter, L. et al. Interplay of SLC33A1-dependent and -independent Golgi sialic acid O-acetylation in CASD1 catalysis. Nat Commun 17, 3156 (2026). https://doi.org/10.1038/s41467-026-71333-y
Keywords: sialic acid, Golgi acetylation, gangliosides, SLC33A1, CASD1