Clear Sky Science · en
The ribosome-associated N-terminal acetyltransferase B coordinates global proteostasis and autophagy in plants by creating Ac/N-degrons
How Plants Keep Their Proteins in Balance
Every cell in a plant is packed with proteins that must be constantly built, repaired, and removed. When this balancing act falters, growth slows and stress can become deadly. This study uncovers how a tiny chemical tag placed on the very beginning of many proteins helps plants decide which proteins to recycle quickly and how strongly to activate an internal clean‑up process called autophagy. Understanding this control system reveals how plants survive long periods without light or nutrients, and may one day help us breed crops that better withstand harsh conditions.
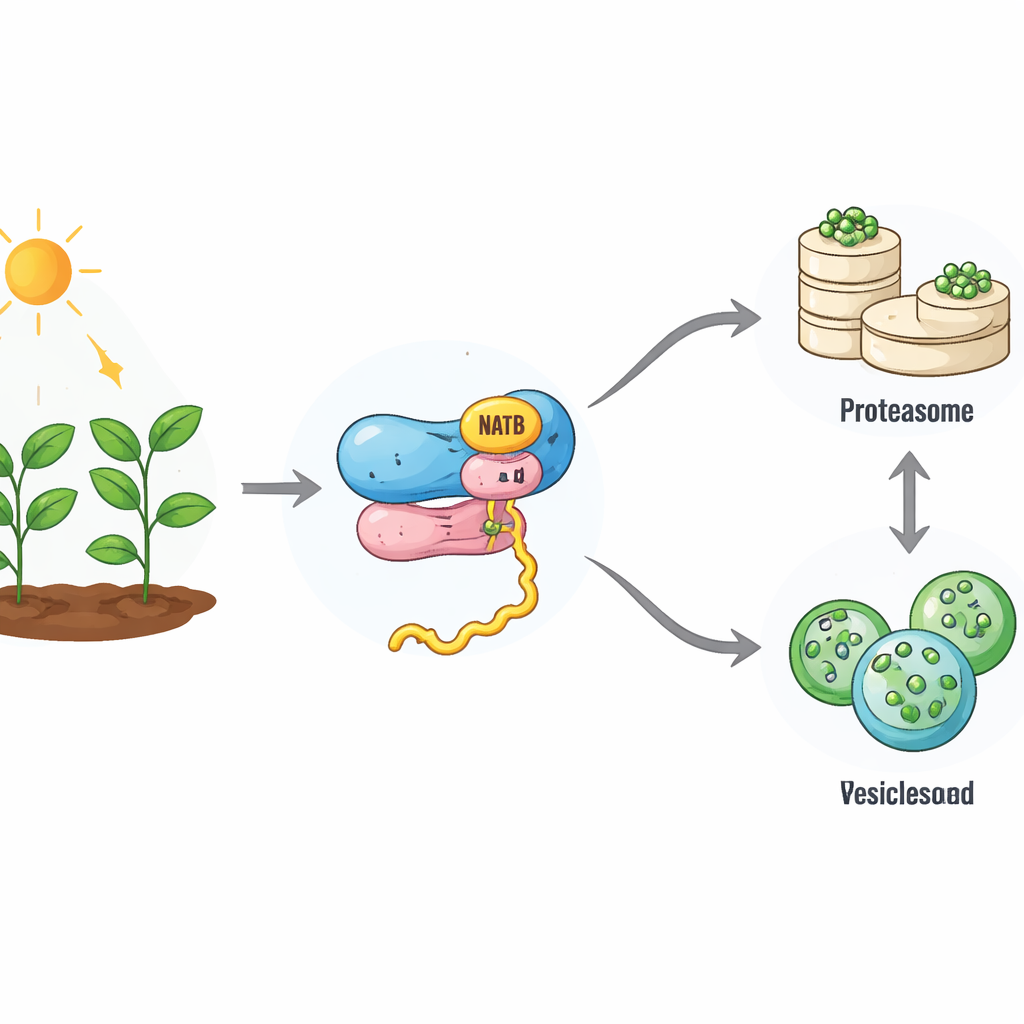
A Small Tag with Big Consequences
As new proteins are made on ribosomes, they can be “capped” by a chemical group at their starting end. In plants, a ribosome‑linked enzyme complex called NatB adds this cap to about one‑fifth of all proteins. Until now, scientists did not fully understand what this widespread tagging means for the fate of those proteins. By using CRISPR gene editing to switch off the catalytic part of NatB in Arabidopsis, the authors created plants that largely lack this modification on NatB’s usual targets. Surprisingly, these plants were stunted but survived, unlike NatB‑deficient animals, suggesting that plant cells can partly compensate for the loss. Still, many proteins that normally carry the NatB tag became only partially or not at all modified, offering a window into how this system works.
Slower Protein Turnover and a Shift in the Cellular Recycling System
When the team measured how quickly proteins are broken down, they found that NatB‑deficient plants have a sluggish cellular recycling machinery. The main proteindestruction route, the ubiquitin‑proteasome system, worked less efficiently: its activity dropped, and proteins earmarked for this pathway carried fewer of the usual “destroy me” marks. At the same time, the overall rate of new protein production also declined. Detailed protein surveys showed that many NatB‑dependent proteins were now more stable and accumulated in the cell, indicating that the NatB tag normally helps turn some proteins into short‑lived components. Yet not all NatB targets behaved this way, pointing to a more selective effect shaped by each protein’s sequence and context.
Autophagy Kicks In as a Backup Plan
The study revealed that, as the proteasome route slows down, another recycling pathway ramps up. This second system, autophagy, encloses portions of the cell into membrane bubbles that are delivered to a compartment where contents are broken down and reused. NatB‑deficient plants showed higher levels of core autophagy proteins and a stronger flow of material through this pathway, especially during darkness when energy is scarce. Plants lacking NatB survived much longer in extended dark or under nitrogen and sulfur starvation than normal plants, but this advantage disappeared when autophagy genes were disabled. This indicates that boosted autophagy helps compensate for the weakened proteasome system, keeping the cell’s protein economy from collapsing.
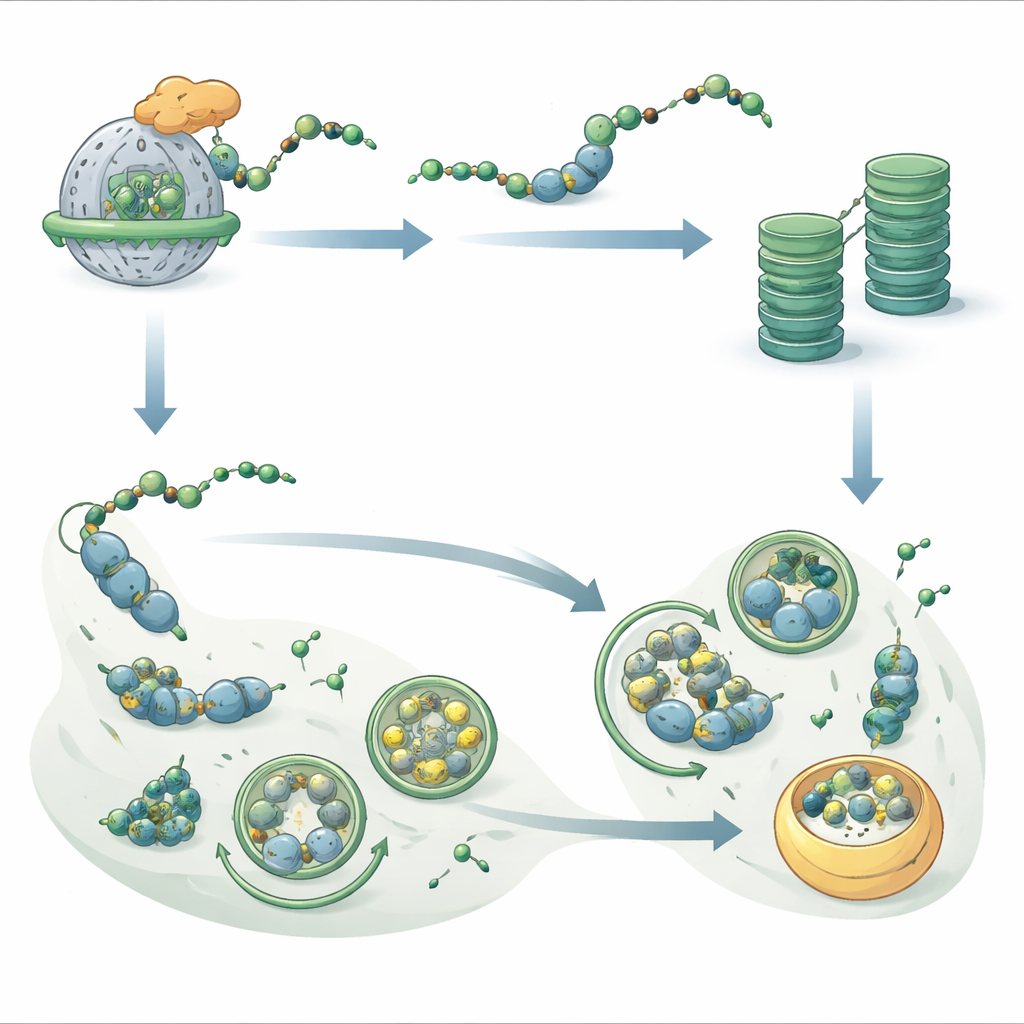
A Key Energy Sensor at the Heart of the Switch
To understand what flips the balance from proteasome use toward autophagy, the authors focused on an energy‑sensing protein complex called SnRK1. Two closely related subunits, KIN10 and KIN11, start with sequences that make them likely NatB clients. The researchers showed that NatB can directly tag these proteins in test‑tube assays. In plants lacking NatB, only KIN11 strongly accumulated, and its active, phosphorylated form was more abundant. Careful tracking of protein decay revealed that when KIN11 carries the NatB tag, it is more readily marked for destruction by the proteasome, whereas untagged KIN11 persists. Plants that lacked both NatB and KIN11 lost their extra dark‑stress resistance, while plants engineered to overproduce KIN11 alone became more tolerant to prolonged darkness. These findings pinpoint KIN11 as a crucial messenger that, when stabilized, pushes the cell to favor autophagy and energy conservation.
What This Means for Plant Survival
Put simply, NatB writes a removable “use‑and‑discard” mark on specific proteins, including the KIN11 energy sensor. When NatB is active, KIN11 is kept in check, protein turnover via the proteasome is brisk, and plants grow rapidly under good conditions. When NatB activity is lost or reduced, KIN11 is spared from fast destruction, autophagy is turned up, and plants shift into a survival mode that conserves resources and copes better with long periods of darkness or poor nutrition. This work reveals NatB as a central coordinator between two major recycling systems in plant cells and explains how a subtle chemical tag at the very start of a protein can tip the balance between growth and endurance.
Citation: Gong, X., Pożoga, M., Boyer, JB. et al. The ribosome-associated N-terminal acetyltransferase B coordinates global proteostasis and autophagy in plants by creating Ac/N-degrons. Nat Commun 17, 3116 (2026). https://doi.org/10.1038/s41467-026-71208-2
Keywords: protein quality control, autophagy, plant stress tolerance, post-translational modification, protein degradation