Clear Sky Science · en
Direct observations of atmospheric oxidized mercury speciation in polar areas
Why mercury at the poles matters to people
Mercury pollution may sound like a problem confined to smokestacks and old thermometers, but it quietly accumulates in fish and marine mammals that many northern communities rely on for food. What happens to mercury in the cold, sunlit air above Arctic sea ice or Antarctic snow helps determine how much of this toxic metal ends up in the ocean and, ultimately, on our plates. This study reports the first direct, real-time measurements of specific oxidized forms of mercury in polar air, forcing scientists to rethink how this global pollutant moves, transforms, and settles back to Earth.

How mercury travels and transforms
Mercury released from power plants, industry, and other sources usually enters the atmosphere as a neutral gas that can drift around the globe for many months. On its own, this form does not dissolve easily in water or stick quickly to surfaces. In polar regions, however, sunlight shining on salty snow and sea ice wakes up highly reactive halogen atoms such as bromine and iodine. These atoms act like chemical hooks, grabbing on to the otherwise sluggish mercury gas and turning it into oxidized forms that are more likely to dissolve in water, stick to particles, or wash out in snow and rain.
New eyes on invisible pollutants
Until now, scientists could rarely see these oxidized mercury molecules individually in the real atmosphere. Most instruments had to collect them on filters or coatings for many hours or days, blending all forms together and losing important details. In this research, scientists used an ultra-sensitive mass spectrometer that gently charges molecules in air and then weighs them with exquisite precision. They deployed this instrument at a Finnish Antarctic station and aboard an icebreaker drifting with the Arctic sea ice, capturing snapshots of polar air every few minutes. This setup allowed them to distinguish different oxidized mercury molecules by their exact mass and natural isotopic fingerprints.
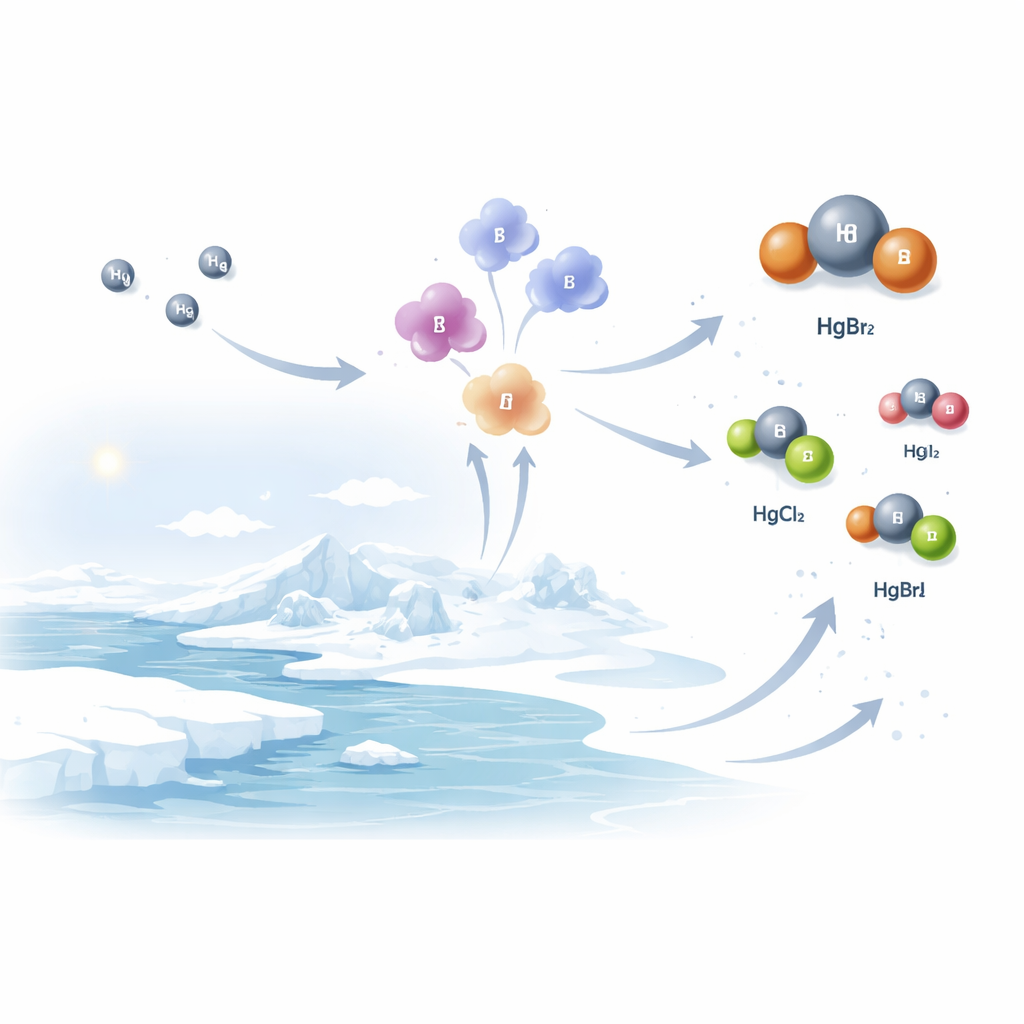
What the air above the ice revealed
The measurements showed that, in both the Arctic and Antarctica, one compound—mercury dibromide—was the dominant oxidized mercury species in springtime air close to the surface. In Antarctica, the instruments also picked up mercury dichloride and several iodide-containing forms, revealing a richer chemical mix than expected. The levels of these species matched, in total mass, what more traditional methods had reported for oxidized mercury, indicating that the newly resolved molecules account for much of what previous instruments had measured as a single lumped quantity. Importantly, the peaks in mercury dibromide lined up with drops in neutral mercury and changes in ozone, underscoring the central role of sunlight-driven bromine chemistry in shaping mercury pollution near the poles.
Why current models miss the mark
Computer models that simulate global mercury pollution had predicted a very different picture. They typically assume that mercury dichloride and certain hydroxyl-containing forms dominate the oxidized pool, with mercury dibromide playing only a minor role. The new field data directly contradict these assumptions: mercury dibromide appears to carry much more of the oxidized mercury load than models allow, and iodine chemistry shows up as a previously underappreciated player. Because each oxidized mercury species breaks apart in sunlight, sticks to particles, and dissolves in water at its own characteristic rate, getting the mix wrong can shift where, and how fast, mercury returns to the surface.
What this means for oceans, food, and policy
For non-specialists, the takeaway is that not all mercury in the air behaves the same way. If fast-breaking forms like mercury dibromide are more common than expected, more oxidized mercury may revert to its long-lived neutral form and travel farther before entering the ocean or land. That could alter which regions receive the heaviest mercury fallout and how much ultimately builds up in fish. By directly identifying individual oxidized mercury molecules in polar air, this study provides the missing chemical detail needed to refine global models and better judge the impact of pollution controls and international agreements aimed at protecting ecosystems and human health from mercury.
Citation: Jokinen, T., Gómez Martín, J.C., Feinberg, A. et al. Direct observations of atmospheric oxidized mercury speciation in polar areas. Nat Commun 17, 3160 (2026). https://doi.org/10.1038/s41467-026-71146-z
Keywords: atmospheric mercury, polar regions, halogen chemistry, air pollution, mass spectrometry