Clear Sky Science · en
Substituent-induced oxidation-reduction molecular organic junction for interfacial hydrogen peroxide photosynthesis
A Smarter Way to Make a Familiar Disinfectant
Hydrogen peroxide is a common household disinfectant, yet making it in factories usually requires energy-intensive processes and hazardous chemicals. This study presents a new solid material that can make hydrogen peroxide directly from air and water using sunlight and gentle vibrations, while also helping clean up toxic metal pollution in water. By carefully arranging the reactive spots within the material at the scale of individual molecules, the researchers show how we might copy the elegance of natural photosynthesis to power greener chemistry and wastewater treatment.
Why Regular Catalysts Fall Short
Most industrial catalysts rely on one kind of active site—a spot on the surface where molecules stick, react, and leave. That works for simple reactions, but many real-world processes, such as splitting water or turning oxygen into useful chemicals, involve several steps that are easier if different tasks happen at different places. Nature already uses this trick: in photosynthesis and in enzymes, multiple specialized sites cooperate to shuffle electrons and protons in a precise order. Conventional engineered catalysts, by contrast, often pack their active sites together in a disorganized way, causing wasted energy and unwanted side reactions that lower efficiency.
Designing a Two-Sided Molecular Workbench
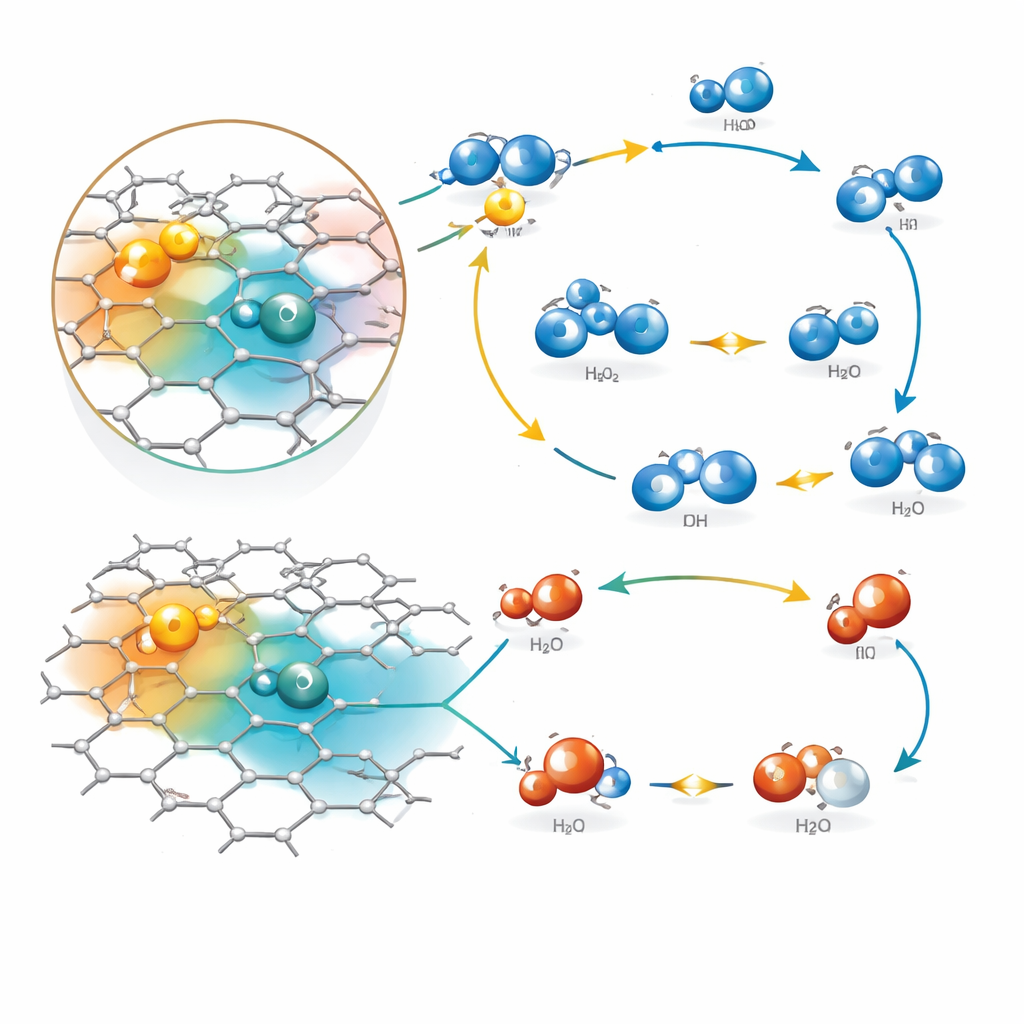
The team tackled this problem using a family of porous organic solids called covalent triazine frameworks. These are rigid networks built from rings of carbon and nitrogen, linked by benzene units, forming sheet-like structures with many internal channels. By swapping some of the benzene linkers for fluorine-decorated versions, they were able to fine-tune how electrons are distributed within the framework. Detailed computer simulations showed that with a specific amount of fluorine—creating a material dubbed CTF-TF-0.5—the electronic structure naturally divides into two distinct regions. One region tends to hold onto positively charged “holes,” acting as an oxidation zone, while the other concentrates extra electrons, serving as a reduction zone. In effect, the material becomes a built-in molecular junction, with separate “sides” dedicated to taking electrons away from molecules or giving electrons to them.
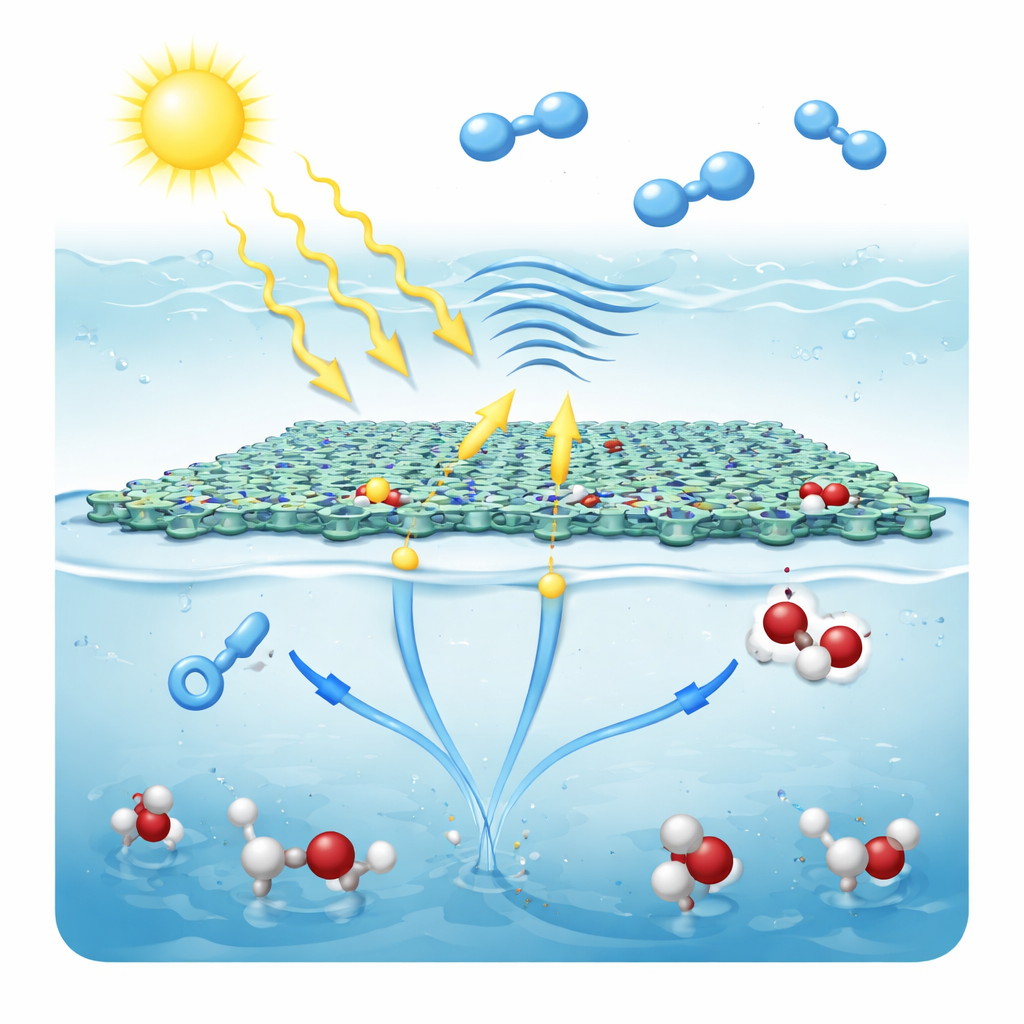
Turning Air and Water into Peroxide
In operation, thin flakes of CTF-TF-0.5 float at the boundary between air and water, forming a three-phase interface of gas, liquid, and solid. Sunlight excites electrons in the framework, and simultaneous ultrasound vibrations enhance its piezoelectric response, helping separate charges even more efficiently. Electrons travel through the structure toward the reduction zones, where they react with oxygen from the air just above the water’s surface. This stepwise process converts oxygen into hydrogen peroxide through reactive intermediates. At the oxidation zones, the positively charged holes pull electrons out of water molecules, generating short-lived radicals that also pair up to form hydrogen peroxide. Because oxidation and reduction happen at distinct but connected sites, unwanted charge recombination is suppressed and both half-reactions are driven along pathways that favor hydrogen peroxide rather than fully reducing oxygen to water.
Boosting Performance with Structure and Force
The researchers used a battery of techniques—spectroscopy, microscopy, and high-pressure measurements—to show how the special arrangement of sites affects behavior. Compared with related materials lacking the clean separation of functions, CTF-TF-0.5 shows stronger charge separation, higher surface potentials under light, and a more pronounced mechanical response when squeezed or vibrated, all of which promote faster electron migration. Under combined light and ultrasound at room temperature, the floating catalyst reaches a hydrogen peroxide production rate of about 4.7 millimoles per gram per hour, outperforming many previously reported organic photocatalysts and piezoelectric materials. The setup works not only in pure water but also in tap water, seawater, river water, rainwater, and hospital wastewater, maintaining substantial activity despite impurities.
Cleaning Toxic Metals from Real Wastewater
Beyond simply generating hydrogen peroxide, the team demonstrated a practical environmental use: removing arsenic from acidic mining wastewater. In this form of pollution, arsenic appears mainly as As(III), which is highly toxic and hard to capture. During treatment with CTF-TF-0.5 under light and ultrasound, the in situ–generated hydrogen peroxide oxidizes As(III) to As(V), a less toxic form that binds more readily to the framework and can be filtered out. In lab tests, the material converted over 95% of As(III) to As(V) within several hours and adsorbed the resulting As(V) efficiently, even in authentic mine effluent with a low pH similar to real-world conditions.
What This Means for Everyday Life
By building a catalyst whose molecular architecture explicitly separates where electrons are taken and given, this work shows a route to more efficient, selective chemical processes powered only by light and gentle mechanical energy. The new material can float on water, draw in oxygen from the air, and steadily produce hydrogen peroxide without added chemicals, all while helping to trap and remove dangerous metals like arsenic. For a layperson, the takeaway is that careful control of structure at the tiniest scales can translate into cleaner, safer ways to make familiar compounds and to treat polluted water, bringing industrial chemistry a step closer to the elegance of biological systems.
Citation: Li, Z., An, L., Guan, L. et al. Substituent-induced oxidation-reduction molecular organic junction for interfacial hydrogen peroxide photosynthesis. Nat Commun 17, 2794 (2026). https://doi.org/10.1038/s41467-026-70959-2
Keywords: hydrogen peroxide, photocatalyst, covalent triazine framework, water purification, arsenic removal