Clear Sky Science · en
Timosaponin AIII enhances CAR-T cell potency and prevents relapse through impairing CAR-Tregs
Turning Down Immune Brakes to Boost Cancer Therapy
For people with aggressive blood cancers, CAR-T cell therapy can be life-changing: a patient’s own immune cells are reprogrammed to hunt down tumor cells. Yet many patients still relapse when those engineered cells lose their punch. This study explores a plant-derived molecule, Timosaponin AIII, that helps CAR-T cells stay active longer by selectively disabling a key brake inside the immune system, potentially making these therapies more durable and effective.
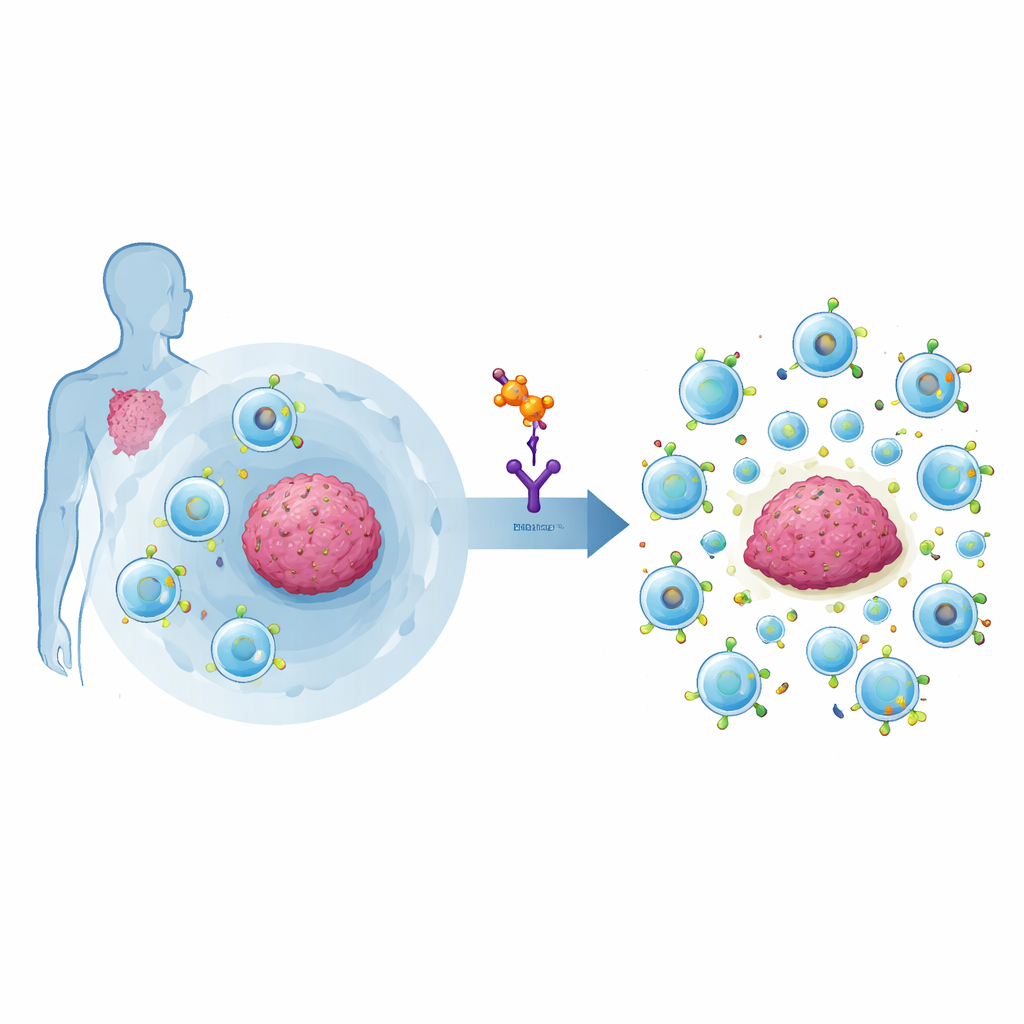
Why Powerful Cancer Fighters Sometimes Stall
CAR-T cells are custom-built immune cells that carry an artificial sensor to recognize cancer targets such as CD19 on malignant B cells. In real patients, however, only about half enjoy long-lasting remissions. One major culprit is a special subset of immune cells called regulatory T cells, or Tregs. These cells normally prevent the immune system from attacking healthy tissue, but in cancer they can smother anti-tumor responses, including those of CAR-T cells. Recent clinical studies showed that when CAR-T products contain too many Tregs, patients are far more likely to resist treatment or relapse. Unfortunately, existing drugs that deplete Tregs are often blunt instruments, harming helpful immune cells along with the harmful ones.
Finding a Gentle Helper in a Medicinal Plant
The researchers built a screening system that reports when a central Treg control switch, the protein FoxP3, turns off immune activation. They tested more than 3,000 natural and approved compounds and flagged those that could loosen FoxP3’s grip without killing cells. One candidate stood out: Timosaponin AIII (TAIII), a steroid-like molecule from the traditional Chinese herb Anemarrhena asphodeloides, already in clinical testing as a topical anti-inflammatory skin treatment. In human T cells grown under conditions that usually turn them into Tregs, TAIII sharply reduced the formation and activity of these suppressive cells, while leaving other T-cell types largely intact. This suggested TAIII could nudge the immune system away from suppression and toward attack.
How a Small Molecule Frees CAR-T Cells
Diving into the mechanism, the team used chemical probes, protein binding tests, and computer modeling to discover that TAIII attaches to the adenosine A2A receptor (A2AR), a well-known immune “off switch” that responds to high levels of adenosine in tumors. Rather than blocking the main binding site, TAIII wedges into a cholesterol-sensitive region of the receptor in the cell membrane, acting as an allosteric inhibitor. By displacing cholesterol-like lipids and dampening a signaling chain that ends in the protein CREB, TAIII lowers FoxP3 production and undermines the Treg program. When A2AR was removed from T cells or CAR-T cells, TAIII lost its effect, confirming that this receptor is its critical target.
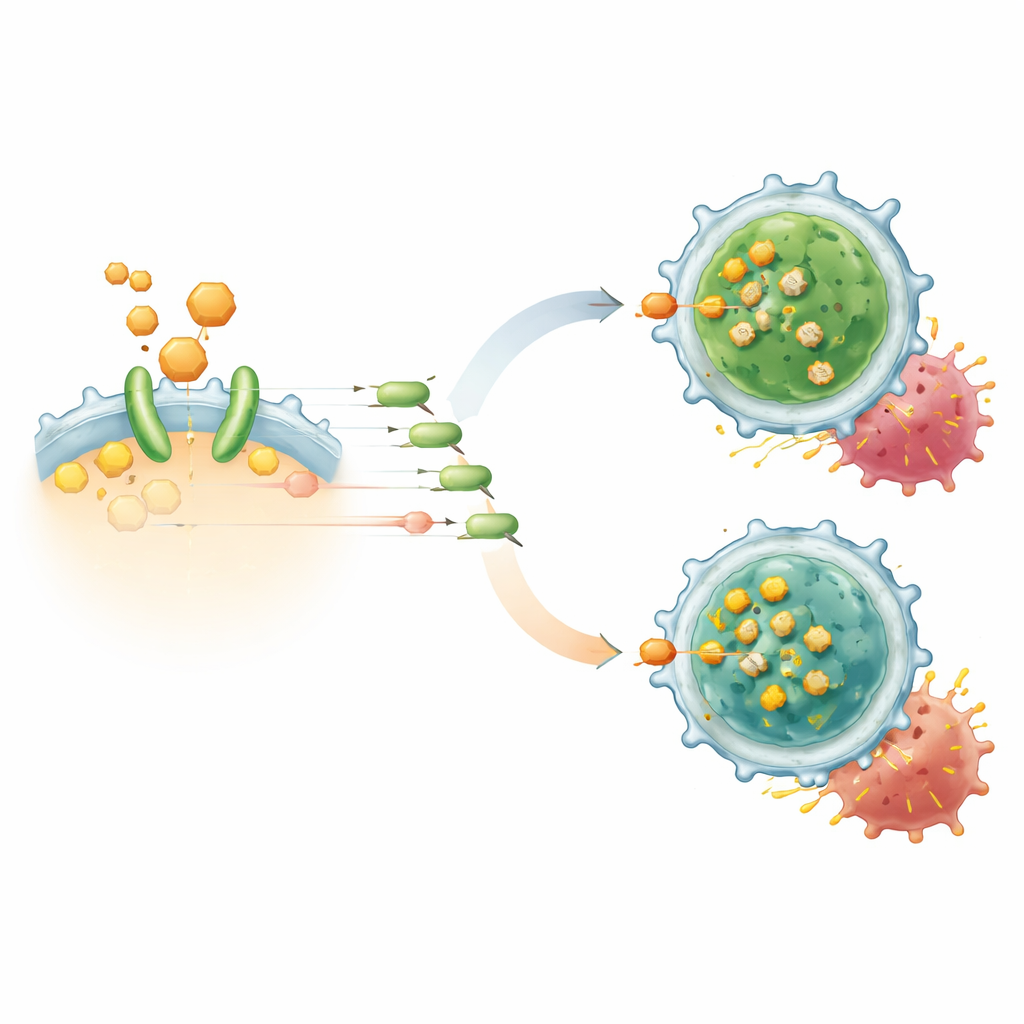
Stronger, Longer-Lasting CAR-T Responses in Models
In laboratory co-culture experiments, adding TAIII to human CD19 CAR-T cells increased their ability to kill lymphoma cells and boosted the release of attack molecules such as interferon-gamma and IL-2. At the same time, it shrank the fraction of CAR-T cells that bore Treg-like traits and promoted central memory T cells, a subset linked to lasting protection. Single-cell RNA sequencing revealed that TAIII-treated CAR-T populations contained fewer FoxP3-high regulatory cells and more vigorous killer T cells. In multiple mouse models of blood cancers and solid tumors, combining TAIII with CAR-T therapy slowed tumor growth, deepened responses, and dramatically reduced late relapses. When CAR-T products were experimentally stripped of Tregs, TAIII no longer added benefit, underscoring that its main role is to dismantle this suppressive subset rather than to over-stimulate all T cells.
Beyond CAR-T: Rewiring the Tumor Immune Neighborhood
TAIII’s effects were not limited to engineered cells. In immunocompetent mice bearing solid tumors, daily TAIII dosing reduced the number of Tregs inside tumors, increased infiltration by CD8 “killer” T cells, and raised levels of anti-tumor cytokines, all without obvious toxicity or weight loss. When combined with anti–PD-1 checkpoint therapy—another widely used form of immunotherapy—TAIII amplified tumor control and expanded beneficial memory-like T cells. Importantly, TAIII showed little effect in severely immune-deficient mice that lack functional T cells, indicating that its anti-cancer activity depends on reshaping the immune response rather than directly poisoning cancer cells.
What This Could Mean for Future Cancer Care
To a layperson, the message is that the authors have found a way to selectively ease off one of the immune system’s internal brakes without cutting the fuel line or flooring the gas pedal. By tuning down regulatory T cells through a precise interaction with the A2A receptor, Timosaponin AIII helps CAR-T cells stay active, form longer-lived memory populations, and avoid being silenced by the tumor environment. In preclinical studies and tests with patient-derived CAR-T cells, this translated into stronger tumor killing and fewer relapses. While clinical trials will be needed to confirm safety and effectiveness in people, TAIII emerges as a promising, plant-based add-on that could make cutting-edge immunotherapies both more durable and more broadly successful.
Citation: Hou, M., Zhang, W., Qi, Z. et al. Timosaponin AIII enhances CAR-T cell potency and prevents relapse through impairing CAR-Tregs. Nat Commun 17, 3045 (2026). https://doi.org/10.1038/s41467-026-70867-5
Keywords: CAR-T cell therapy, regulatory T cells, adenosine A2A receptor, Timosaponin AIII, cancer immunotherapy