Clear Sky Science · en
Spatial heterogeneity of MDSCs mediated by ANXA1-FPRs signaling drives immune suppression in OSCC progression
Why the Body’s Defenses Struggle Against Some Mouth Cancers
Oral squamous cell carcinoma, a common form of mouth cancer, often resists today’s most exciting cancer treatments: immunotherapies that unleash the body’s own defenses. This study asks a basic but crucial question: why do powerful immune cells, which should recognize and destroy cancer, so often fail in these tumors? By mapping where different cells sit inside the tumor and how they talk to each other, the researchers uncover a shifting “cat‑and‑mouse” game between cancer cells, immune suppressor cells, and killer T cells that helps explain treatment failure—and suggests a way to fix it. 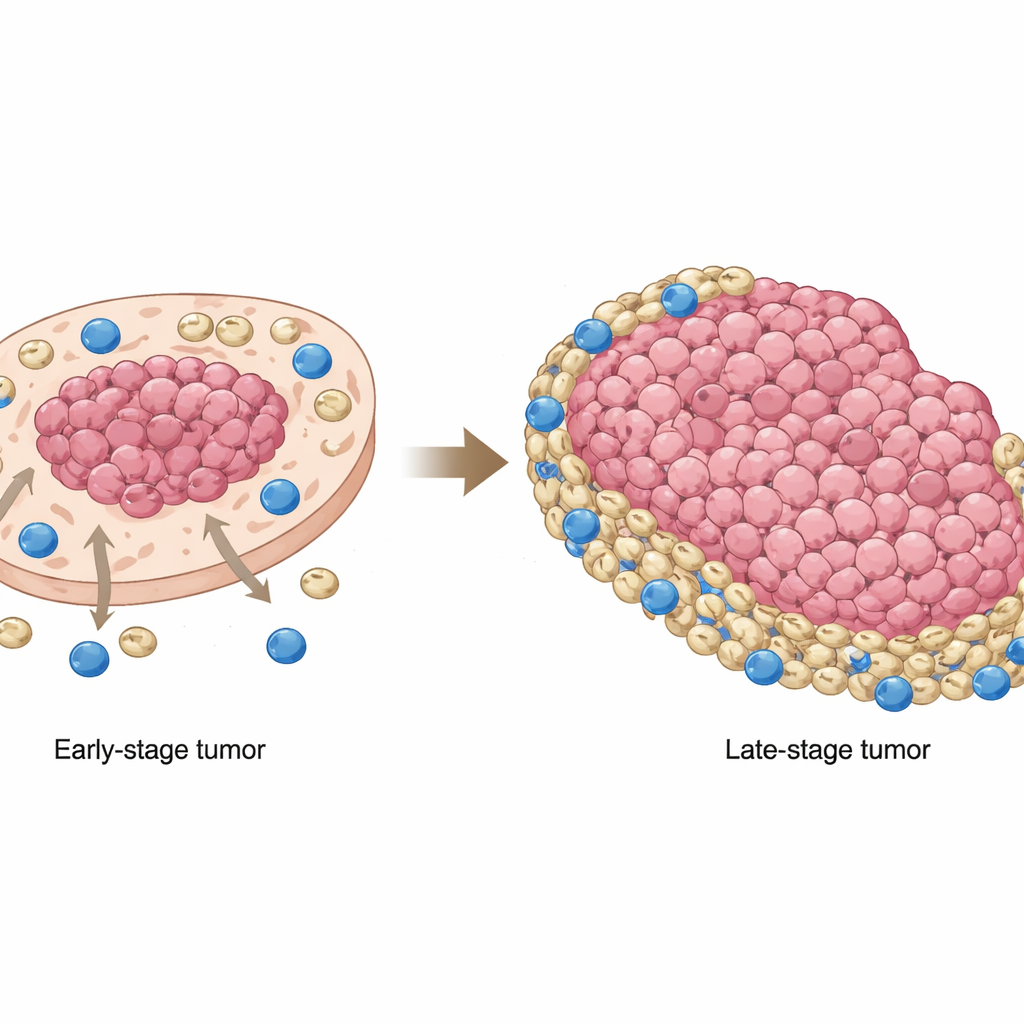
The Hidden Landscape Inside a Tumor
The authors combined two cutting-edge approaches: single-cell RNA sequencing, which reads out the activity of individual cells, and spatial transcriptomics, which shows where those cells are located within actual tumor tissue slices. Analyzing tumors from patients with early and late stages of oral cancer, they identified major cell players: cancerous epithelial cells, CD8 “killer” T cells, other T and B cells, fibroblasts that form structural support, and specialized immune cells called myeloid-derived suppressor cells (MDSCs). While early tumors contained more active CD8 T cells, late-stage tumors showed weaker T cell signaling and stronger immune-dampening signals from myeloid cells, pointing to a progressively more hostile environment for anti-cancer immunity.
Suppressor Cells on the Move
One of the most striking findings was how the position of MDSCs changed as tumors advanced. In early-stage cancers, these suppressor cells tended to cluster within tumor-cell–rich regions, close to the cancer core. In later stages, they shifted outward, accumulating in tumor-poor areas and at the margins, where CD8 T cells were more common. This meant that in advanced tumors, killer T cells were largely pushed to the edges and then met by a belt of MDSCs, which dampened their ability to attack cancer cells. Patient samples and mouse models confirmed that this redistribution was not just a curiosity: when MDSCs overlapped less with cancer cells and instead camped in non-tumor regions, patients had worse overall survival. 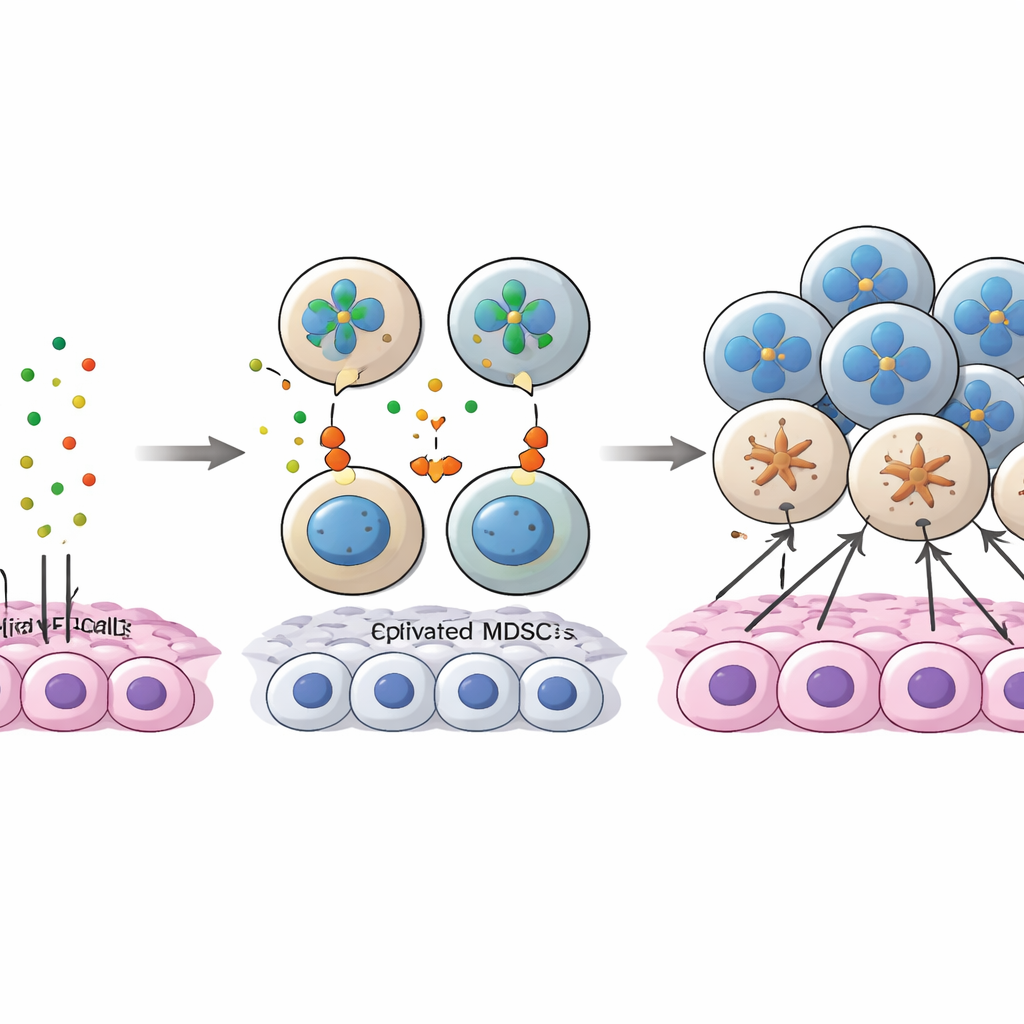
A Signaling Switch Between Cancer and Suppressor Cells
The study zoomed in on a molecular “conversation” between cancer cells and MDSCs involving a protein called annexin A1 (ANXA1) on tumor cells and a family of receptors called FPRs on myeloid cells. In early disease, ANXA1 on cancer cells strongly engaged FPR1 and FPR3, especially on MDSCs and tumor-associated macrophages, helping recruit and maintain these suppressor cells in the tumor core. As cancer progressed, ANXA1 levels on tumor cells dropped, weakening these original connections. At the same time, more MDSCs began expressing another receptor, FPR2, and ANXA1–FPR2 signaling emerged as a compensating route to keep MDSCs engaged. A specific subset of tumor cells with high ANXA1, which also showed stem-like features, appeared to act as “bait,” drawing in MDSCs early on; when ANXA1 fell, MDSCs were no longer anchored in the core and instead shifted toward regions rich in CD8 T cells.
Turning Off the Shield to Help Immunotherapy Work
To test whether breaking this communication could improve treatment, the researchers used a mouse model of oral cancer and blocked FPR2 with a small-molecule inhibitor called WRW4. On its own, FPR2 inhibition reduced the presence of MDSCs and increased CD8 T cells in the tumor environment but did not sufficiently slow tumor growth. Likewise, treatment with an anti–PD-1 immune checkpoint antibody alone produced modest benefits, because MDSCs still formed a suppressive barrier. When both drugs were combined, however, tumor growth was strongly curtailed and mice lived longer. In these animals, MDSC infiltration dropped sharply while CD8 T cells infiltrated deeper into tumor-cell–rich regions, suggesting that blocking FPR2 removes a key shield that normally protects cancer cells from immune attack.
What This Means for Future Cancer Care
Taken together, the work shows that not only the types of cells but also their spatial arrangement and signaling partnerships determine whether immunotherapy can succeed in oral cancer. As tumors advance, MDSCs migrate from the center to the edges, where they intercept killer T cells, and ANXA1–FPR2 signaling helps maintain this suppressive arrangement. By disrupting this pathway—especially in combination with existing checkpoint drugs—doctors may be able to rewire the tumor environment so that the body’s own immune cells can once again recognize and destroy cancer. This suggests a concrete strategy: pairing FPR2 blockers with immunotherapy could improve outcomes for patients whose oral cancers currently resist the best available treatments.
Citation: Li, F., Han, Y., Ou, F. et al. Spatial heterogeneity of MDSCs mediated by ANXA1-FPRs signaling drives immune suppression in OSCC progression. Nat Commun 17, 2535 (2026). https://doi.org/10.1038/s41467-026-70861-x
Keywords: oral squamous cell carcinoma, tumor microenvironment, myeloid-derived suppressor cells, spatial transcriptomics, cancer immunotherapy