Clear Sky Science · en
FGFR signaling establishes spatial gradients of secretory cell identities along the airway proximal-distal axis
How Our Airways Quietly Sort and Defend the Air We Breathe
Every breath sweeps air over millions of cells lining our windpipe and lungs. These cells do far more than provide a passive surface: they detoxify pollutants, fight germs, and help keep tiny air sacs from collapsing. This study reveals that secretory cells in the mouse airway are not all the same. Instead, they are organized in smooth gradients from the throatward (proximal) to the deep lung (distal) regions, and a key signaling pathway, called FGFR2b, helps set up and maintain these gradual shifts in cell identity and function. Understanding this hidden order gives insight into how lungs stay healthy and what may go wrong in chronic lung disease.
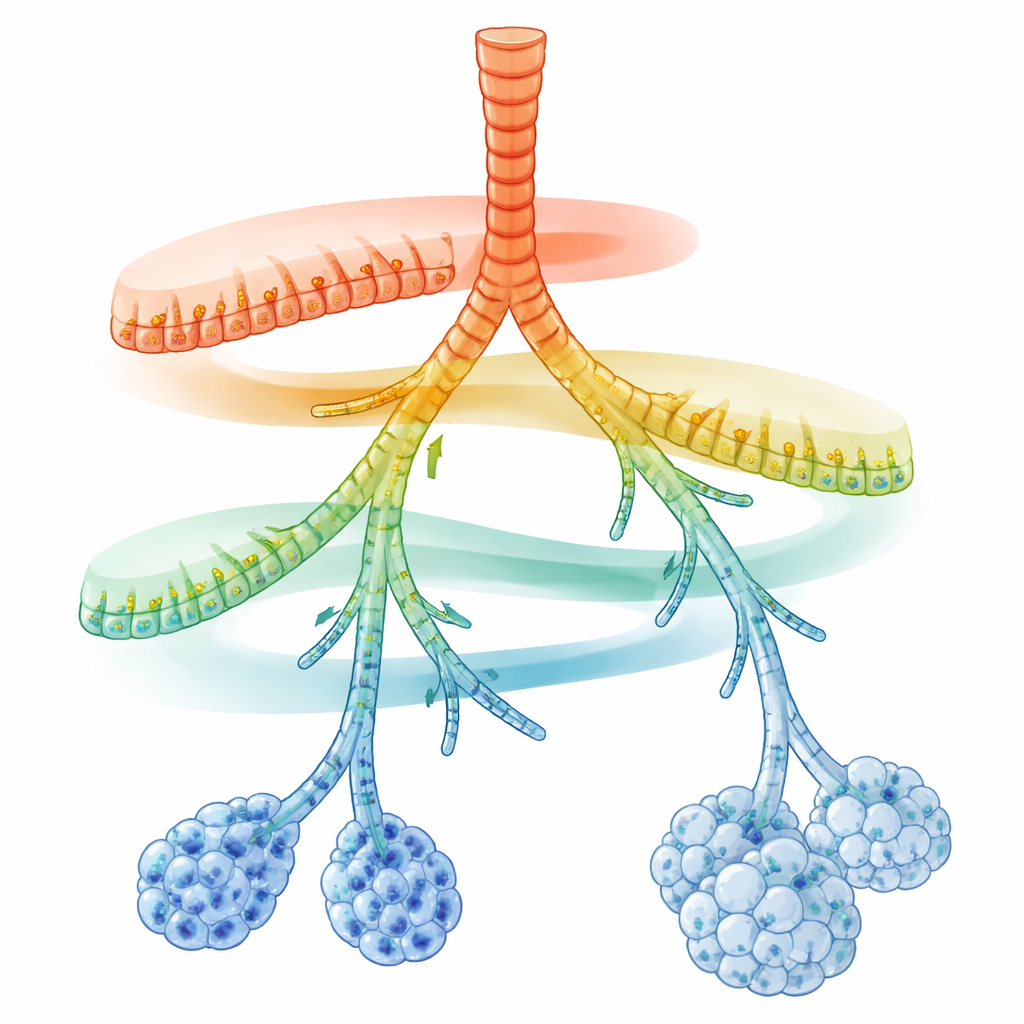
A Hidden Map Along the Breathing Tubes
The researchers focused on secretory cells, which line the airway tubes and secrete mucus, antimicrobial peptides, and surfactant. Using single-cell RNA sequencing, they read out the active genes in thousands of individual cells from adult mouse lungs. When they projected these cells into a map of molecular similarity, secretory cells did not fall into a few rigid types. Instead, they formed a continuous chain of states connecting large airways to tiny air sacs. On top of a shared core program for detoxifying inhaled chemicals, two opposing gene activity gradients appeared: near the larger airways, genes involved in rapid, innate immune defense were highest; toward the farthest branches and alveoli, genes for lipid handling, surfactant production, and antigen presentation dominated.
From One Starting Point to Many Specialized Cell States
To see how this pattern emerges, the team labeled secretory precursors during late fetal life and followed their descendants across early life using lineage tracing and single-cell analysis. These labeled cells initially formed an immature pool with strong protein-making machinery and genes linked to tissue building. After birth, as breathing and exposure to the outside world began, these cells gradually diversified into several mature groups: proximal secretory cells rich in innate-defense genes, more central cells with developmental and regulatory signatures, double-positive cells bridging airway and alveolar identities, and alveolar type 2 cells specialized for surfactant. The detoxification program turned on early and broadly, whereas innate immunity and antigen presentation programs came on later and in distinct regions, indicating that secretory cell features are acquired in a defined sequence over time.
How a Growth Signal Shapes Distal Airway Identity
A major clue to what drives these gradients came from a receptor called FGFR2b, part of a growth-factor signaling pathway already known to be essential for alveolar type 2 cells. FGFR2b and its target genes were more active in distal airway and alveolar cells than in proximal ones. When the researchers switched off FGFR2b specifically in secretory cells just after birth, the normal distal program faltered. Genes required for surfactant production and vesicle trafficking declined, while genes typical of flat alveolar type 1 cells, mitochondrial components, ion channels, and stress-survival factors rose. Some intermediate airway cells now coexpressed moderate levels of proximal innate-defense markers and distal markers, effectively blurring the normal spatial pattern. In organoid cultures grown from airway cells, adding FGFR ligands suppressed proximal innate-defense markers and boosted surfactant-related genes in a way that matched the in vivo findings, confirming that activating FGFR2b is sufficient to push cells toward a distal secretory identity.
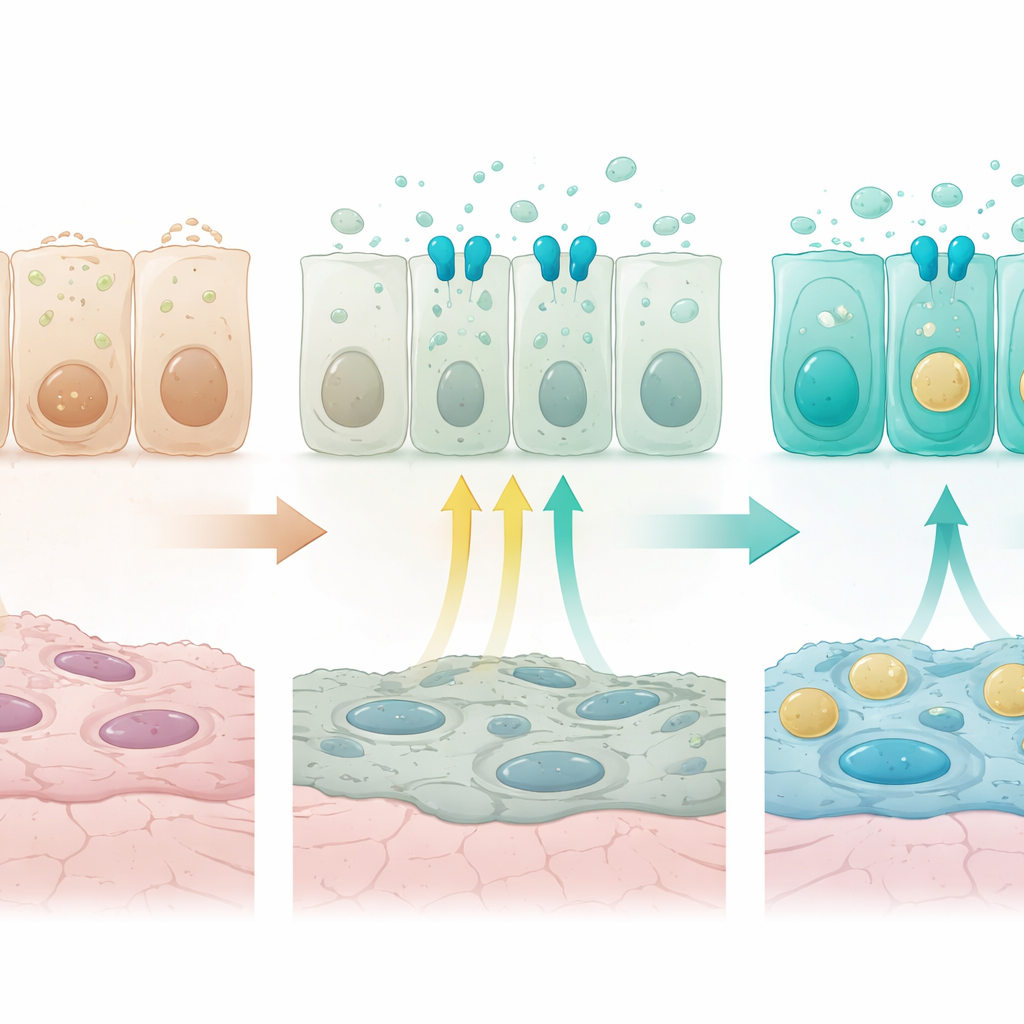
Fine-Tuning by Interaction with Other Signals
The team also probed how FGFR2b signaling interacts with another pathway driven by VEGFA, best known for shaping blood vessels. In organoids, VEGFA alone could dampen some innate-defense genes, and blocking its receptor altered certain distal markers, but it could not fully substitute for FGFR2b. In fact, loss of FGFR2b in vivo reduced Vegfa expression, and combined stimulation of both pathways fed back to lower FGFR2b levels, pointing to a complex cross-talk between these signaling systems. In adult mice, turning off FGFR2b no longer dramatically reshaped innate-immunity gradients, but it was still required to maintain expression of key distal markers, such as the surfactant gene Sftpb and the classic airway secretory marker Scgb1a1, and to preserve hybrid airway–alveolar cells.
Why These Gradients Matter for Lung Health
The picture that emerges is of the airway lining as a finely graded landscape rather than a patchwork of isolated zones. Proximal regions are tuned for rapid, mucous-based defense, while distal regions combine surfactant production with more specialized immune communication. FGFR2b signaling helps establish and sustain this layout, especially in the distal compartments, by promoting lipid and vesicle-related programs and restraining inappropriate proximal-like features. Disrupting these gradients, whether by genetics, environment, or disease, could shift the balance between defense, detoxification, and gas-exchange support, contributing to conditions where distal airways take on more proximal traits. This framework offers a new way to think about lung disorders that “repattern” the airway tree and may guide future strategies to restore proper cellular organization.
Citation: Sountoulidis, A., Theelke, J., Liontos, A. et al. FGFR signaling establishes spatial gradients of secretory cell identities along the airway proximal-distal axis. Nat Commun 17, 2651 (2026). https://doi.org/10.1038/s41467-026-70842-0
Keywords: airway epithelium, secretory cells, FGFR2b signaling, lung development, spatial gradients