Clear Sky Science · en
Proton channels govern vesicular carbonate chemistry in mineralizing cells of a marine calcifier
How Tiny Builders Shape Our Planet
Much of Earth’s limestone cliffs and coral reefs is made by microscopic builders that pull dissolved ingredients from seawater and turn them into solid rock. This study looks inside one such builder—the larval stage of a sea urchin—to ask a surprisingly basic question: how do its cells control acidity while they grow a skeleton from calcium carbonate? The answer reveals an elegant electrical trick that may also explain why marine shell‑forming creatures are so vulnerable to a more acidic ocean.
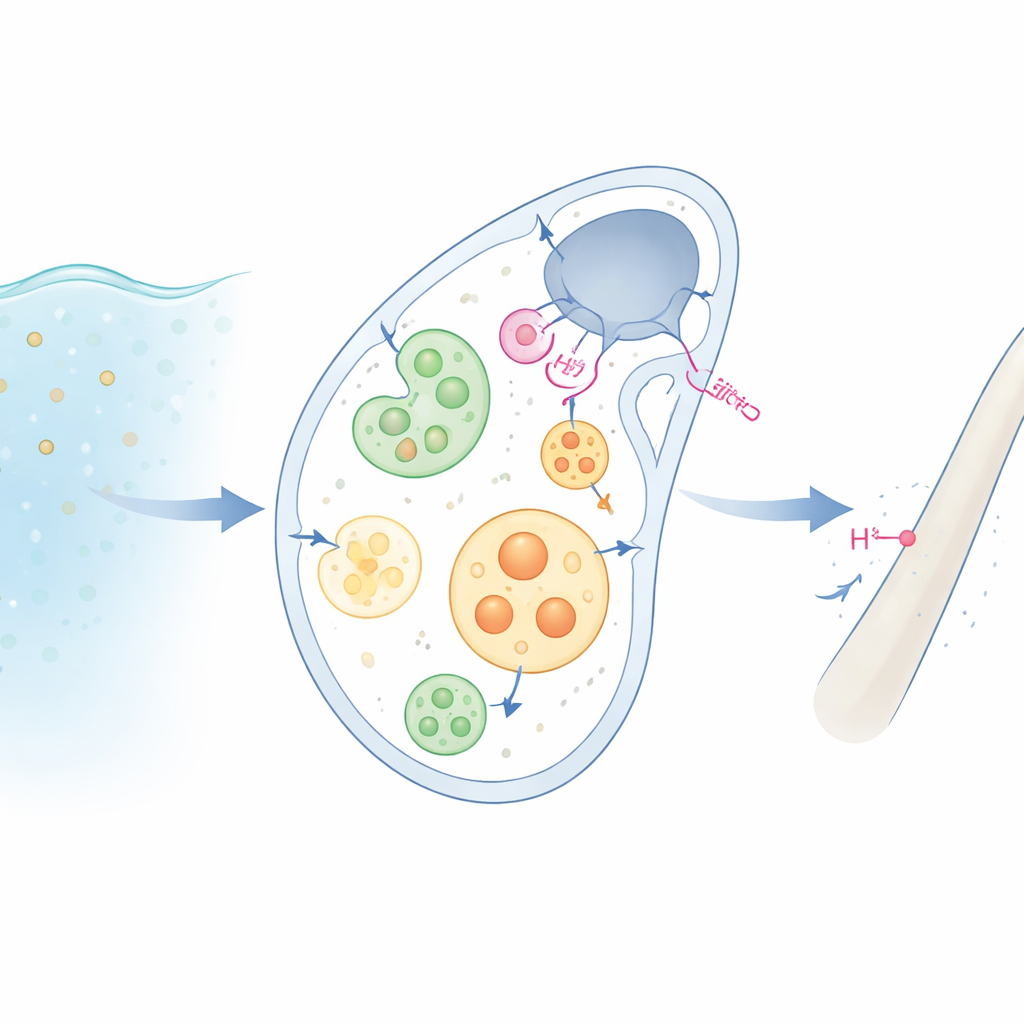
From Seawater to Skeleton
Sea urchin larvae harden their bodies with tiny calcite rods made of calcium carbonate. To do this, specialized “mineralizing” cells first sip in seawater, packaging it into small internal droplets, or vesicles. Inside these droplets, the ingredients for the mineral—calcium, carbonate, and other ions—are concentrated into an amorphous, glassy precursor that later hardens into crystal. But every time calcium carbonate forms, extra acid (protons) is produced. If that acid were allowed to build up, it would dissolve the very mineral the cells are trying to make. How the cells prevent this self‑destruction has been a long‑standing mystery.
Measuring Acidity Inside Moving Droplets
The researchers used live imaging and pH‑sensitive fluorescent dyes to watch the chemistry inside these vesicles in real time. They found that almost half of the vesicles were strongly alkaline—more basic than normal seawater—while the rest were neutral or slightly acidic. When they briefly changed the acidity of the surrounding seawater, both the interior of the cells and the vesicles shifted as well, showing that protons can move relatively freely across these membranes. However, vesicles were partly shielded compared with the cell interior: they responded more slowly and less dramatically, suggesting a controlled “leakiness” that helps keep conditions favorable for mineral formation.
Electric Leaks That Remove Acid
To uncover what creates this leakiness, the team focused on a proton channel protein called Otop2l, previously linked to sea urchin skeleton growth. Using antibodies, they saw that Otop2l sits not only in the outer membrane of the mineralizing cells but also on the membranes of large internal vesicles. When they reduced Otop2l levels with a targeted genetic blocker, both the cells and their vesicles became less responsive to changes in external pH, indicating that this channel is a main route for protons to leave. Electrical recordings in frog eggs and human cells engineered to produce Otop2l showed that the channel opens wide under alkaline conditions and in the presence of calcium and magnesium—exactly the situation in calcifying vesicles. In essence, the cells use their natural voltage difference across the membrane, together with Otop2l, to drive acid out without spending much metabolic energy.
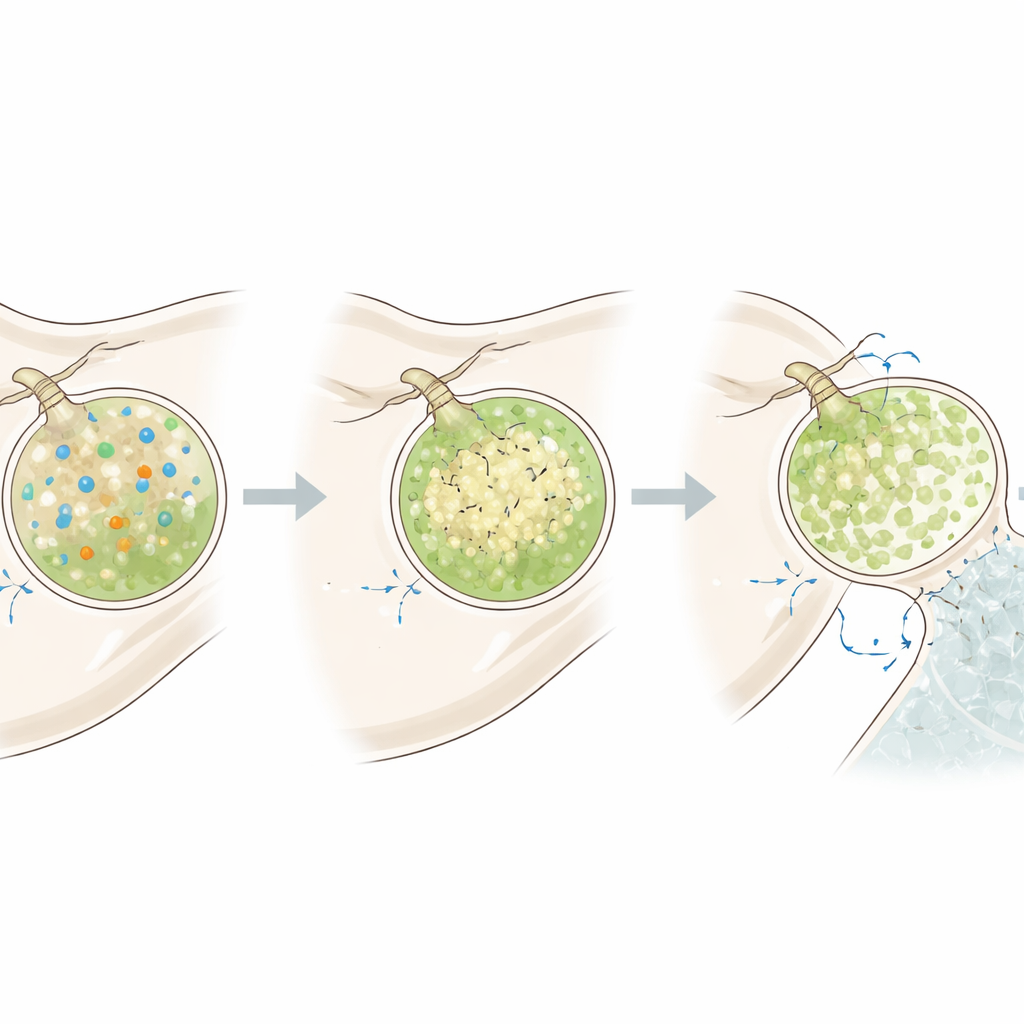
Vesicles Ramping Up for Repair
The team then asked what happens when the larvae need to rebuild skeleton after damage. They dissolved the larval skeletons by briefly exposing them to low‑pH seawater, then returned them to normal water and tracked vesicles during regrowth. During this high‑demand phase, mineralizing cells produced more vesicles overall, with a strong rise in alkaline, calcium‑rich ones. These vesicles could recover their high pH more quickly after an acid pulse, and the cell interiors also became more alkaline. Yet the cells’ electrical potential remained similar, implying that they adjust mainly by boosting carbonate supply and proton conductance rather than overhauling their electrical state.
Why Ocean Acidification Matters
Put together, the findings support a model in which sea urchin mineralizing cells run an “open” system: they continuously endocytose seawater, pump carbon into vesicles, and rely on proton channels and membrane voltage to bleed excess acid back into the surrounding ocean. This setup is efficient as long as external pH stays within the narrow, stable range that has characterized much of Earth’s recent history. But if seawater becomes more acidic—as is happening today with rising carbon dioxide—this outward flow of protons can weaken or even reverse. The work therefore offers mechanistic insight into why many marine calcifiers struggle under ocean acidification: the very channels that once made biomineralization cheap and robust may become liabilities in a rapidly changing sea.
Citation: Jonusaite, S., Przibylla-Diop, C., Musinszki, M. et al. Proton channels govern vesicular carbonate chemistry in mineralizing cells of a marine calcifier. Nat Commun 17, 2578 (2026). https://doi.org/10.1038/s41467-026-70837-x
Keywords: biomineralization, sea urchin larvae, proton channels, ocean acidification, calcium carbonate