Clear Sky Science · en
A bioinspired microdevice unifying energy storage and actuation through hydration control
Why tiny machines need tiny muscles and batteries
Imagine a dust-sized robot that could swim through your bloodstream or inspect the tiniest cracks in a jet engine. To work on its own, such a micro-robot would need both a power source and moving parts that fit on a speck of dust. Today, batteries and motors are usually separate, bulky components. This paper describes a new, bioinspired approach where energy storage and motion are woven together in one microscopic device, much like how a real muscle stores and uses energy in the same place.
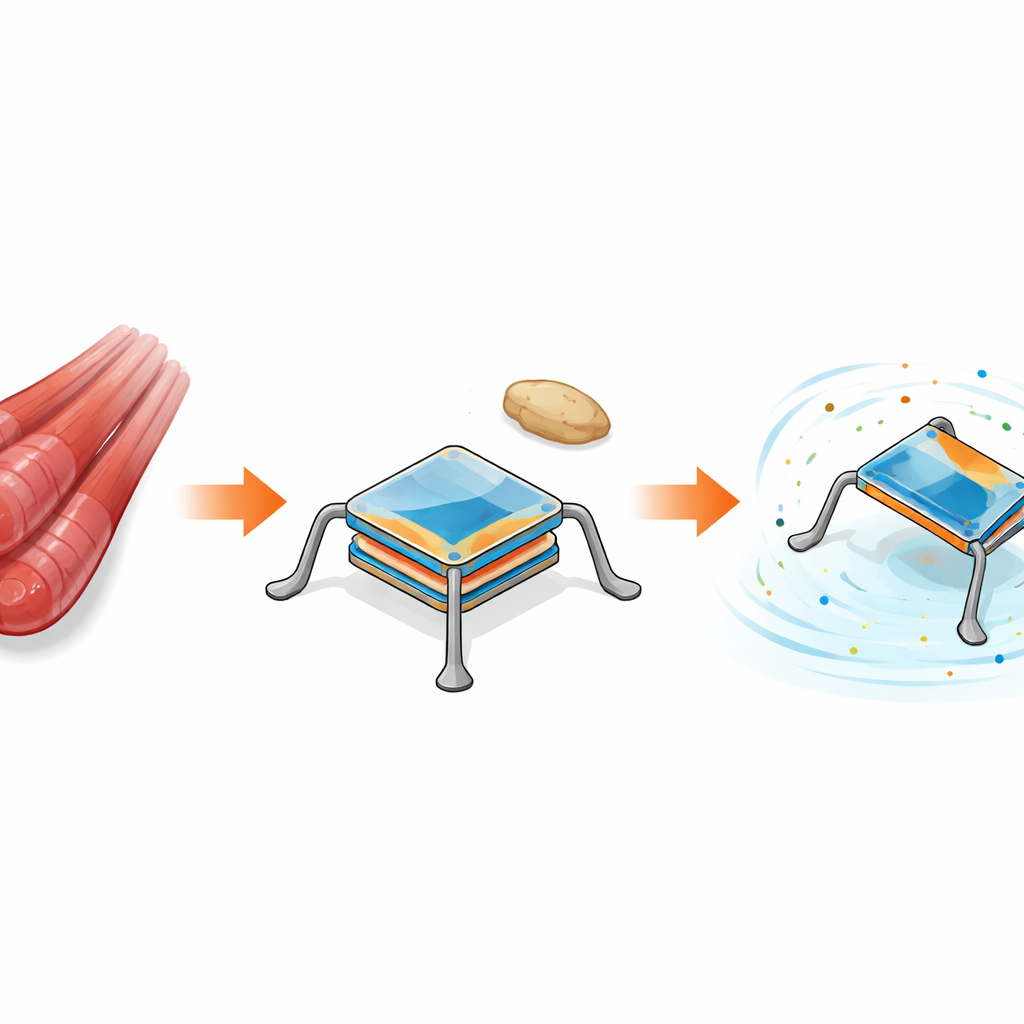
Learning from living muscles
In your body, muscles both store chemical energy and convert it directly into movement. The authors take this idea and apply it to very small machines. They use special plastics called conjugated polymers that swell and shrink when ions and water move in and out of them. The same movements that allow these plastics to store electrical energy can also make them expand and contract, turning them into tiny artificial muscles. By folding thin films into three-dimensional shapes using a micro-origami process, the team builds a sub-millimeter device that combines a rechargeable battery at its center with four flexible “legs” that act as micro-actuators around it.
Water: helper, troublemaker, and control knob
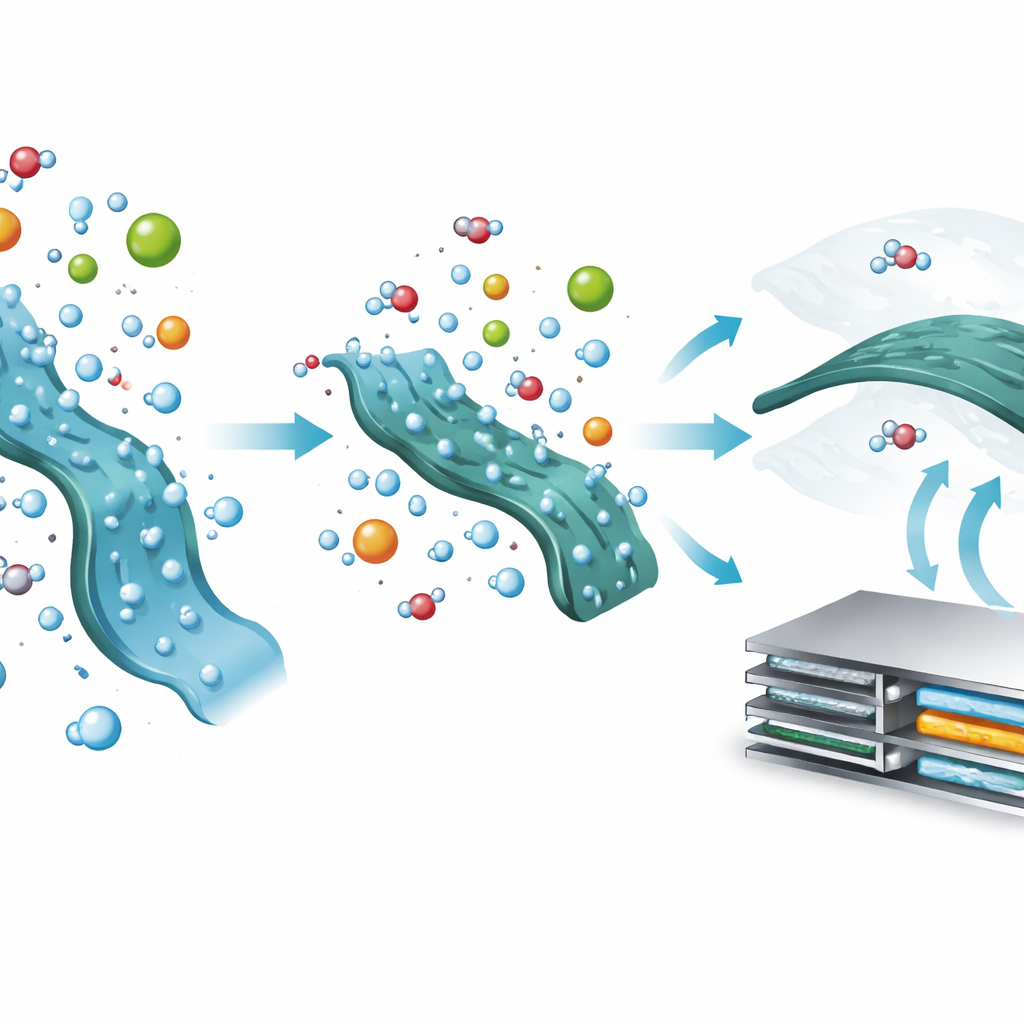
Water is both essential and dangerous for these plastics. When water tags along with charged particles entering the polymer, it helps the material swell strongly, which is good for motion. But too much water seeps into the polymer’s backbone and breaks its chemical structure over time, ruining its ability to store energy. Using sensitive techniques that track vibrations in the material and tiny changes in mass during charging and discharging, the researchers show that the way negative ions hold onto water—their “hydration”—controls this trade-off between powerful motion and long-term stability. Strongly hydrated ions drag dense shells of water into the polymer, causing large swelling, slow relaxation, and chemical damage. Weakly hydrated ions, by contrast, can shed their water and sit closer to the polymer, pushing excess water away.
Taming water with the right ions
To tip the balance in their favor, the team replaces common sulfate ions in the electrolyte with triflate ions, which naturally disrupt water shells. In the older sulfate-based liquid, the plastic electrode rapidly falls apart: water floods into its structure, triggers side reactions, and the battery capacity crashes within a few dozen cycles. With triflate, the onset of strong water involvement shifts to higher voltages, water uptake into the polymer is sharply reduced, and the material keeps its electrical activity over many cycles. Measurements show that, during operation, the polymer actually expels more water molecules when triflate is present, limiting harmful reactions and preserving the delicate conductive backbone that makes energy storage possible.
A grain-of-salt-sized power and motion unit
Building on this hydration control, the researchers create a dual-cell zinc–polymer microbattery with a footprint of just 0.56 square millimeters—smaller than a grain of salt. Folded into a stacked 3D shape, it delivers high areal capacity and can run for more than 2200 charge–discharge cycles while maintaining nearly perfect efficiency. Around this central power unit, they attach polypyrrole-based legs that bend when ions and a small amount of water move in and out. Compared with a traditional, strongly hydrated electrolyte used in such actuators, the triflate-based liquid lets the legs relax much faster and cuts their energy consumption by about a factor of four. The legs can flap repeatedly, stir tiny beads in water, and generate cilia-like flows, all while powered solely by the local microbattery. The same battery can also run simple electronics such as light-emitting diodes and a low-power watch.
Toward smarter, untethered micro-robots
The work shows that simply tuning how water clings to ions can unlock both durable energy storage and efficient motion in the same microscopic structure. By choosing weakly hydrated anions, the authors protect the polymer electrodes from water-driven decay and speed up the mechanical response of polymer actuators, all in an aqueous, biocompatible environment. This strategy extends beyond the specific plastics used here and could be applied to other conductive polymers and electrolytes. In the long run, controlling hydration in this way may enable tiny, untethered machines—such as implantable medical devices and micro-robots—where the “battery” and the “muscle” are no longer separate parts but two faces of the same smart material.
Citation: Zhang, W., Merces, L., Ma, J. et al. A bioinspired microdevice unifying energy storage and actuation through hydration control. Nat Commun 17, 2650 (2026). https://doi.org/10.1038/s41467-026-70831-3
Keywords: microbattery, conjugated polymer, hydration control, microactuator, microrobotics