Clear Sky Science · en
A single cluster of RNA Polymerase II molecules is stably associated with active genes
How cells turn genes on in powerful bursts
Every cell in your body must decide which genes to use and when, often switching them on in short, intense bursts. For decades, scientists have suspected that enzymes responsible for reading DNA gather into tiny “hot spots” inside the nucleus to boost this process, but how these clusters work has remained controversial. This study peeks inside living fruit fly embryos with advanced microscopes to show that each active gene is partnered with a single, stable cluster of the enzyme that reads DNA into RNA, and that these clusters behave more like crowded work sites than exotic droplets of cellular “phase-separated” matter.
Little machines that read the genome
The enzyme at the center of this story is RNA Polymerase II, a molecular machine that slides along DNA and copies genes into RNA, the first step toward making proteins. Earlier work painted conflicting pictures: some experiments suggested that polymerase molecules group into big, long-lived “factories” serving many genes at once, while others saw only fleeting gatherings of a few molecules. The authors focused on a dramatic moment in early fruit fly development called zygotic genome activation, when a quiet embryo suddenly switches on thousands of its own genes. This natural surge in activity provided a powerful test bed for watching how polymerase molecules move, gather, and engage with genes in real time.
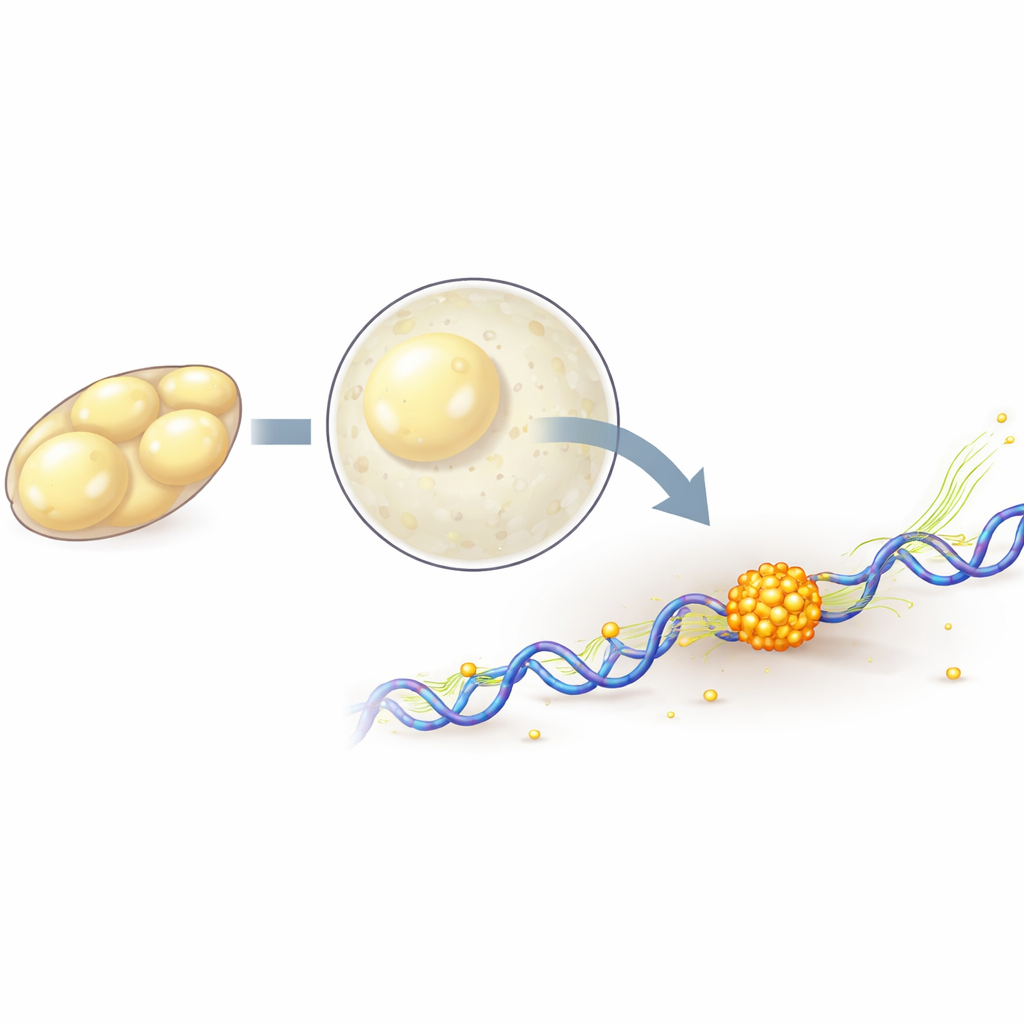
Watching single molecules in a living embryo
To follow individual polymerase molecules, the team genetically tagged one of their core components with fluorescent proteins, then used lattice light-sheet microscopy and single-molecule tracking to record their motion in 3D with high speed and gentle illumination. They found that as the embryo enters its major activation phase, more polymerase molecules become tightly attached to DNA, consistent with more genes being switched on. By briefly blocking different steps in the transcription cycle with drugs, they could separate molecules that are just starting at a gene from those that are actively moving along it. This analysis showed that cluster formation depends on the very first steps of turning a gene on, while active copying along the gene tends to weaken and shorten the clusters.
Clusters that change character as development unfolds
Imaging whole nuclei over time revealed dozens of small polymerase clusters long before the genome-wide activation wave, with their number and spacing changing as nuclear divisions slow down. Early in development, many clusters last nearly as long as the gap between cell divisions, suggesting they are dominated by polymerases in an early, “poised” state that often fails to produce full RNA molecules. Later, when transcription ramps up, clusters become more dynamic: their lifetimes no longer simply track the cell cycle, and their internal makeup shifts toward polymerases that are actually elongating along genes. Other measurements of how molecules move inside and outside clusters indicate that, near active genes, polymerase molecules are more confined and likely to bump back into the same sites, supporting the idea of a locally busy work zone rather than a loose, liquid droplet.
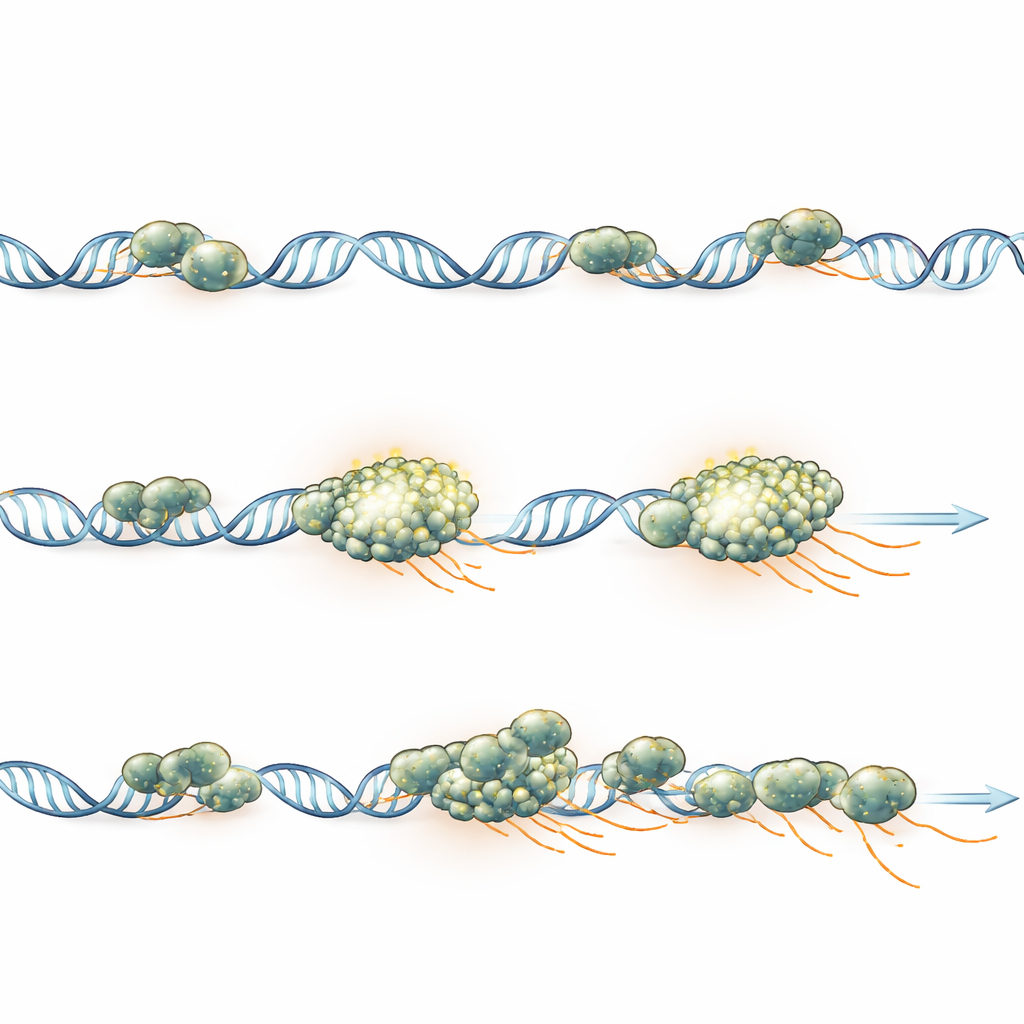
One cluster, one gene during a burst
To link clusters directly to gene output, the researchers watched specific reporter genes that glow where new RNA is being made, while simultaneously tracking polymerase. For several different genes, they consistently saw just one polymerase cluster sitting at each active gene copy during a transcription burst. The intensity of the cluster rose and fell in lockstep with the amount of nascent RNA, and when duplicated sister gene copies could be resolved, each carried its own distinct cluster rather than sharing one. Computer simulations, tuned to match the imaging conditions, showed that genes with strong polymerase loading form visible clusters, while weaker genes may still recruit polymerase but remain too dim to detect, explaining why only a minority of active genes show clear clusters in the microscope.
What this means for how genes are controlled
This work argues that, in these embryos, polymerase clusters mainly reflect how many enzymes are actively engaged at a single gene, not a separate “factory” structure or a special droplet that must form to allow transcription. A cluster appears when many polymerases are loaded in quick succession, stays stably associated with that one gene throughout a burst of activity, and gradually disperses as polymerases finish copying and depart. To a lay reader, the take-home message is that gene switching is organized through focused, gene-by-gene hubs of activity: each active gene temporarily gathers its own team of copying machines, and the size and lifetime of that team directly mirror how strongly the gene is turned on.
Citation: Mukherjee, A., Kapoor, M., Shankta, K. et al. A single cluster of RNA Polymerase II molecules is stably associated with active genes. Nat Commun 17, 2580 (2026). https://doi.org/10.1038/s41467-026-70775-8
Keywords: RNA polymerase II clustering, zygotic genome activation, transcription bursting, gene regulation in embryos, single-molecule imaging