Clear Sky Science · en
Single-pot mechanochemically-enabled fluorine atom closed-loop economy using PFASs as fluorinating agents
Turning “Forever Chemicals” into Useful Tools
Plastics and coatings that resist stains, heat, and chemicals often owe their powers to PFAS, a large family of compounds nicknamed “forever chemicals” because they refuse to break down in nature. This durability has created a global pollution problem, contaminating water, soil, wildlife, and people. The study summarized here describes a way not only to break apart these stubborn substances at room temperature, but also to capture and reuse their valuable fluorine atoms in new, useful molecules—offering a rare win–win for environmental cleanup and chemical manufacturing.
Why Forever Chemicals Are So Hard to Handle
PFAS are woven into modern life: they appear in nonstick pans, waterproof jackets, medical devices, electronics, and more. Their carbon–fluorine bonds are among the strongest in chemistry, giving them exceptional stability and making them hard to burn, dissolve, or chemically attack. Existing ways to destroy PFAS often require very high temperatures or strong electrical or chemical treatments, which consume energy, generate waste, and usually discard the fluorine instead of recovering it. Because fluorine is prized in medicines and advanced materials, finding a gentle method that both dismantles PFAS and recycles their fluorine has become an important scientific goal.
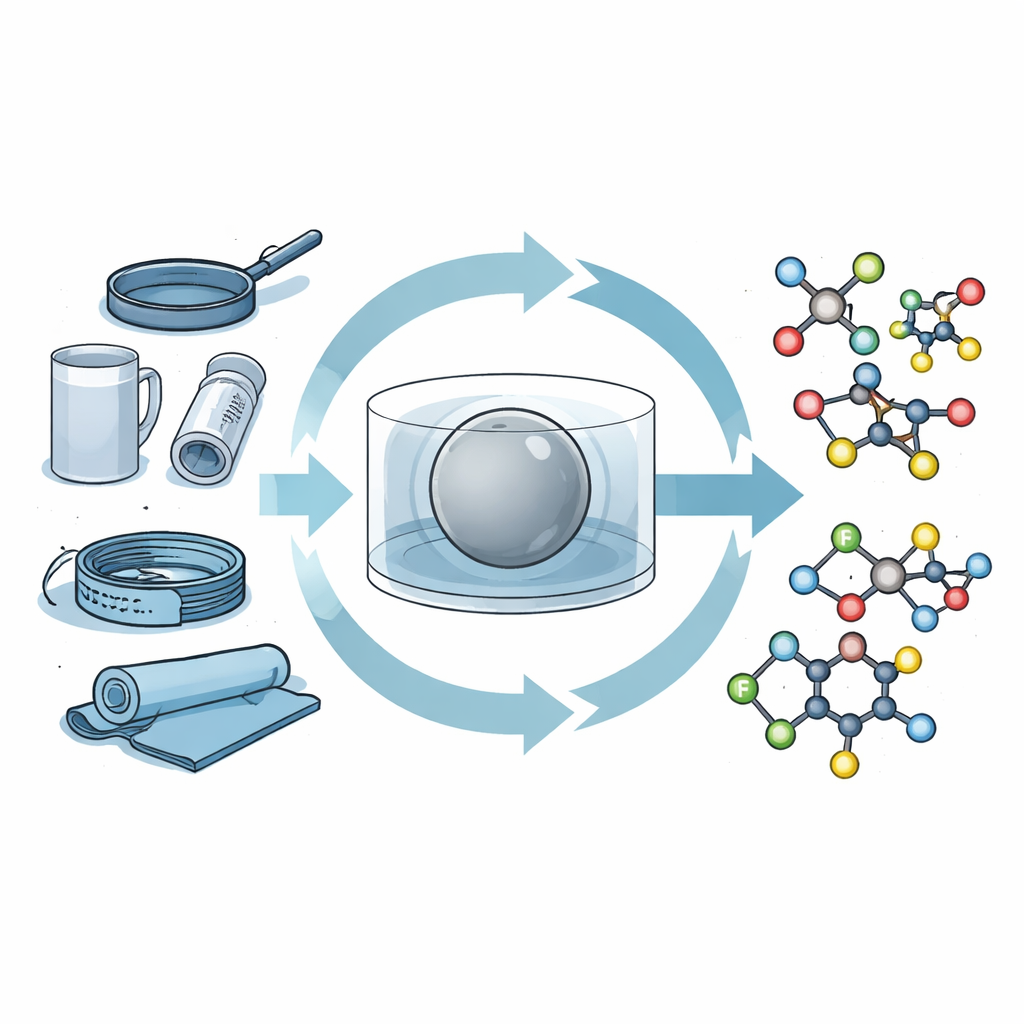
Smashing Plastics to Free Fluorine
The researchers use a technique called mechanochemistry, which replaces hot liquids and large reactors with solid powders shaken together in a sealed metal jar containing a heavy ball. As the ball rattles at high speed, it grinds and compresses the materials, triggering chemical changes through mechanical force rather than heat. In their “single-pot” setup, PFAS plastics such as common pipes, filters, and the well-known nonstick material PTFE are milled together with a simple solid base. This step pulls fluorine atoms off the plastic chains, generating fluoride in place and turning the original polymer into a degraded, carbon-rich residue.
From Waste Fluorine to High-Value Building Blocks
Without opening a new flask or purifying intermediates, the team then adds another solid ingredient—a sulfonyl chloride—to the same jar and keeps milling. The freed fluoride atoms react to form sulfonyl fluorides, a class of highly stable, selectively reactive molecules that serve as key building blocks for “click” chemistry, a modular way of snapping together complex structures. Under these conditions, many different PFAS materials, from finely powdered polymers to tough fluoroplastic parts, efficiently donate their fluorine to create sulfonyl fluorides in high yield. The products can be collected simply by rinsing and filtering, with almost no solvent and no column chromatography, even when the process is scaled up to tens of grams.
Probing How the Breaking and Making Happens
To understand what happens inside the mill, the scientists monitored the release of fluoride over time and analyzed the leftover solids. For one common PFAS plastic, PVDF, the base appears to strip hydrogen and fluorine pairs from the chain, leaving behind segments with carbon–carbon double bonds and forming fluoride salts. For fully fluorinated materials that lack hydrogen, the pathway is different: the team found signs of carbon turning into small mineral-like fragments and amorphous carbon, hinting at bond-breaking sequences that may involve reactive intermediates. In all cases, the mechanochemical treatment steadily liberates fluoride ions, which then become available for the second, fluorine-transfer step.
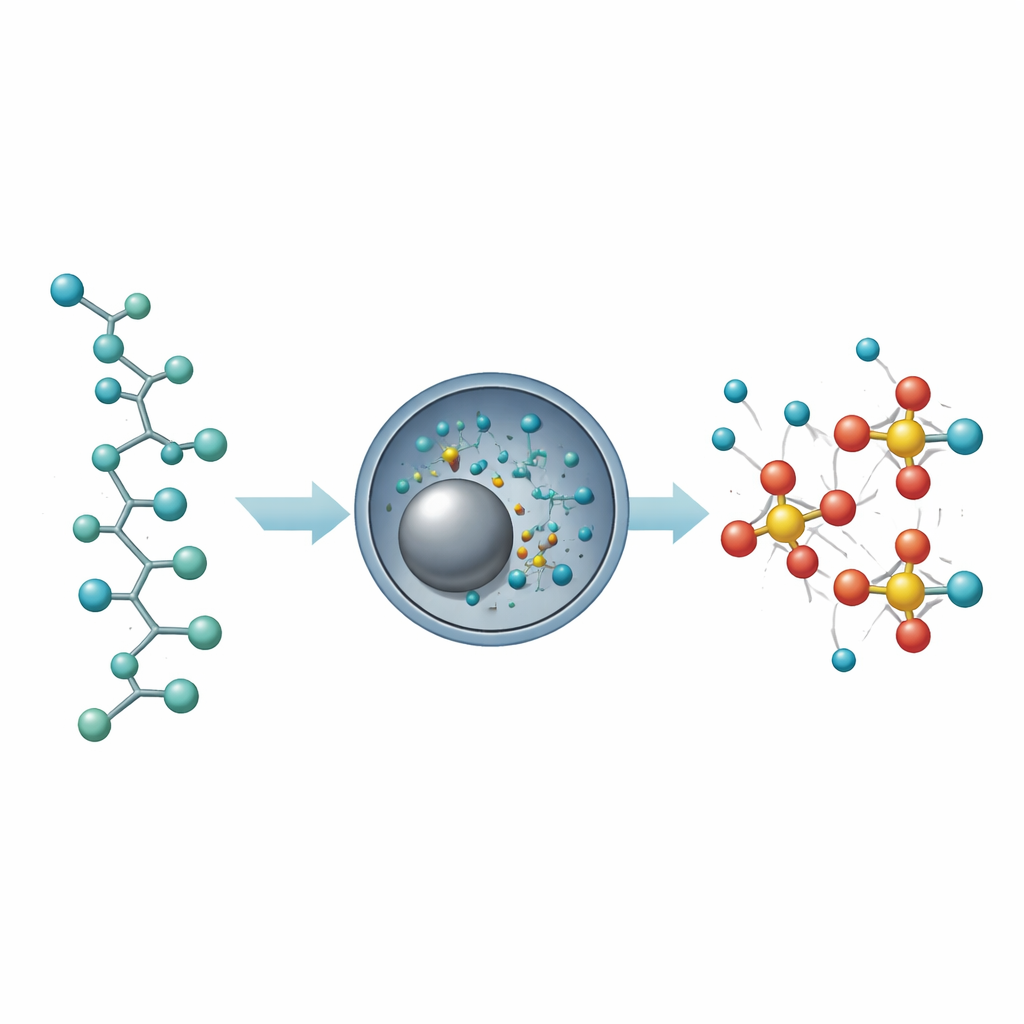
Closing the Loop on Fluorine Use
The sulfonyl fluorides made from PFAS-derived fluoride proved to be versatile. The authors used them to attach sulfonyl groups to a range of complex molecules, including natural products, vitamins, and drug-like compounds, demonstrating that fluorine reclaimed from discarded plastics can feed directly into sophisticated synthesis. Because the method works on mixed consumer waste, operates at room temperature with minimal solvent, and avoids harsh reagents, it points toward a “closed-loop” fluorine economy in which the element is repeatedly reused instead of mined and discarded. In practical terms, this approach suggests a future where problematic forever chemicals are not just destroyed, but upcycled into new materials and medicines, turning an environmental liability into a valuable resource.
Citation: Long, H., Kirby, G. & Ackermann, L. Single-pot mechanochemically-enabled fluorine atom closed-loop economy using PFASs as fluorinating agents. Nat Commun 17, 2696 (2026). https://doi.org/10.1038/s41467-026-70766-9
Keywords: PFAS recycling, mechanochemistry, fluorine economy, plastic waste upcycling, sulfonyl fluorides