Clear Sky Science · en
Frazzled/DCC directs spatial progenitor integration ensuring steady-state intestinal turnover
How the Gut Quietly Renews Itself
Every day, the cells lining our intestines wear out and must be replaced, yet the organ keeps its shape and size with remarkable precision. This study, using the fruit fly as a model, uncovers a hidden guidance system that tells newborn gut cells exactly where to go to plug tiny gaps in the lining. Understanding this “cell traffic control” not only clarifies how healthy organs maintain themselves, but also sheds light on how similar guidance signals might be hijacked during cancer spread.
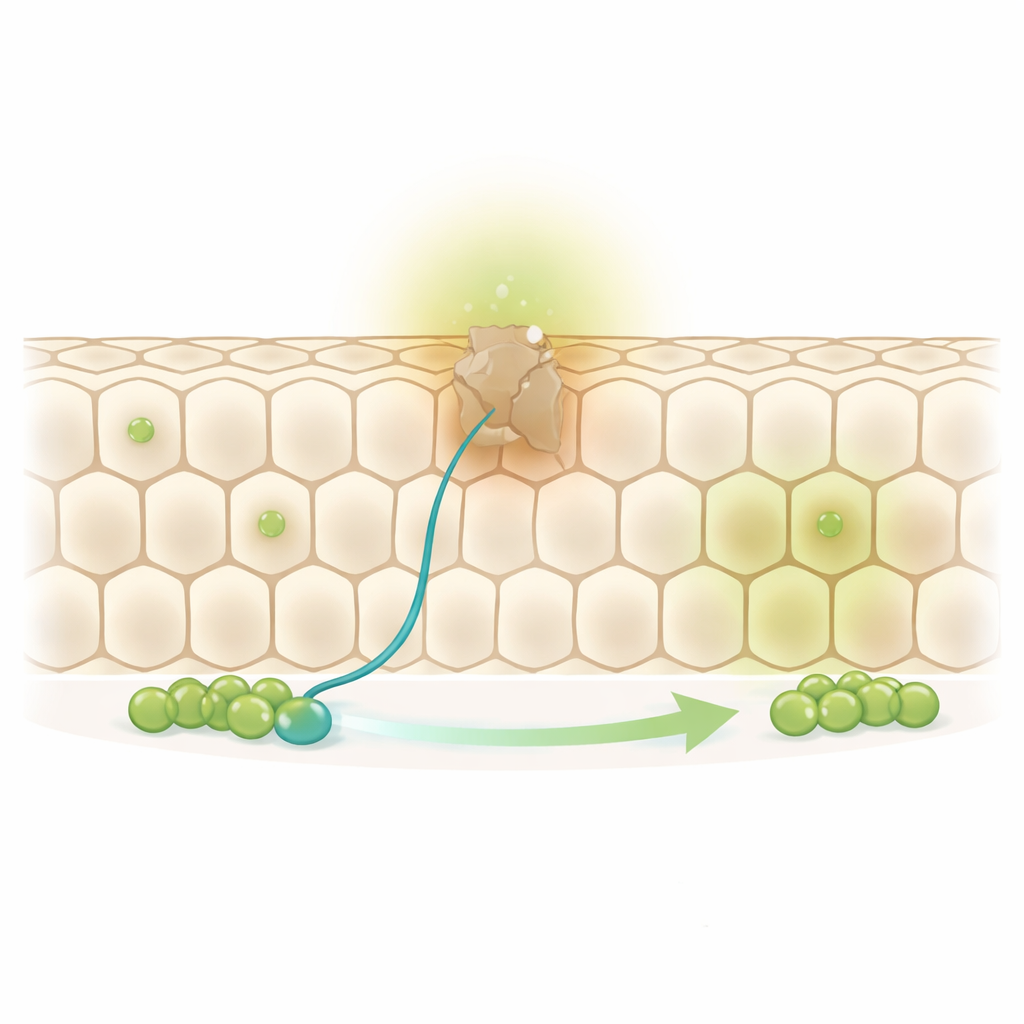
A Honeycomb in Constant Motion
The fruit fly midgut is covered by a single layer of large absorptive cells arranged like a honeycomb. At the base of this sheet sit scattered stem cells and their immediate daughters, called progenitors. When an old absorptive cell reaches the end of its life, a stem cell divides and its progenitor daughter eventually replaces the worn-out neighbor. However, the authors noticed that roughly one-third of the cells in this honeycomb have no stem cell or progenitor directly next to them. That raised a puzzle: how are these “out-of-reach” cells renewed without leaving holes in the barrier?
New Cells on the Move
By tracking individual replacement events over a week, the researchers found that these remote cells are renewed just as often as those sitting right next to a stem cell. That implies that progenitor cells must move. Indeed, the team observed that progenitors extend long, thin protrusions—cellular feelers—farther and more frequently than stem cells themselves. These protrusions are not random: under normal conditions they point mainly toward older, unreplaced neighbors rather than freshly renewed ones, hinting at an active search-and-rescue behavior aimed at the cells most in need of replacement.
A Nerve Guidance Signal Repurposed for the Gut
To understand how these protrusions are steered, the authors turned to a family of molecules best known from brain wiring: Netrins and their receptors Frazzled/DCC and Unc-5. In the nervous system, Netrins act like long-range beacons that attract or repel growing nerve fibers. In the fly gut, the team showed that the Frazzled/DCC and Unc-5 receptors are found specifically on progenitor cells, concentrated in their protrusions. Worn-out absorptive cells, in turn, begin to produce and release a Netrin called Netrin-B. When the researchers increased Netrin-B in selected cells, nearby progenitors grew longer protrusions that oriented toward the source, then migrated to occupy that spot. When they blocked Netrin-B or disabled Frazzled, protrusions shortened, remote cells were no longer efficiently replaced, and the flies died earlier, underscoring how vital this guidance is for gut health.
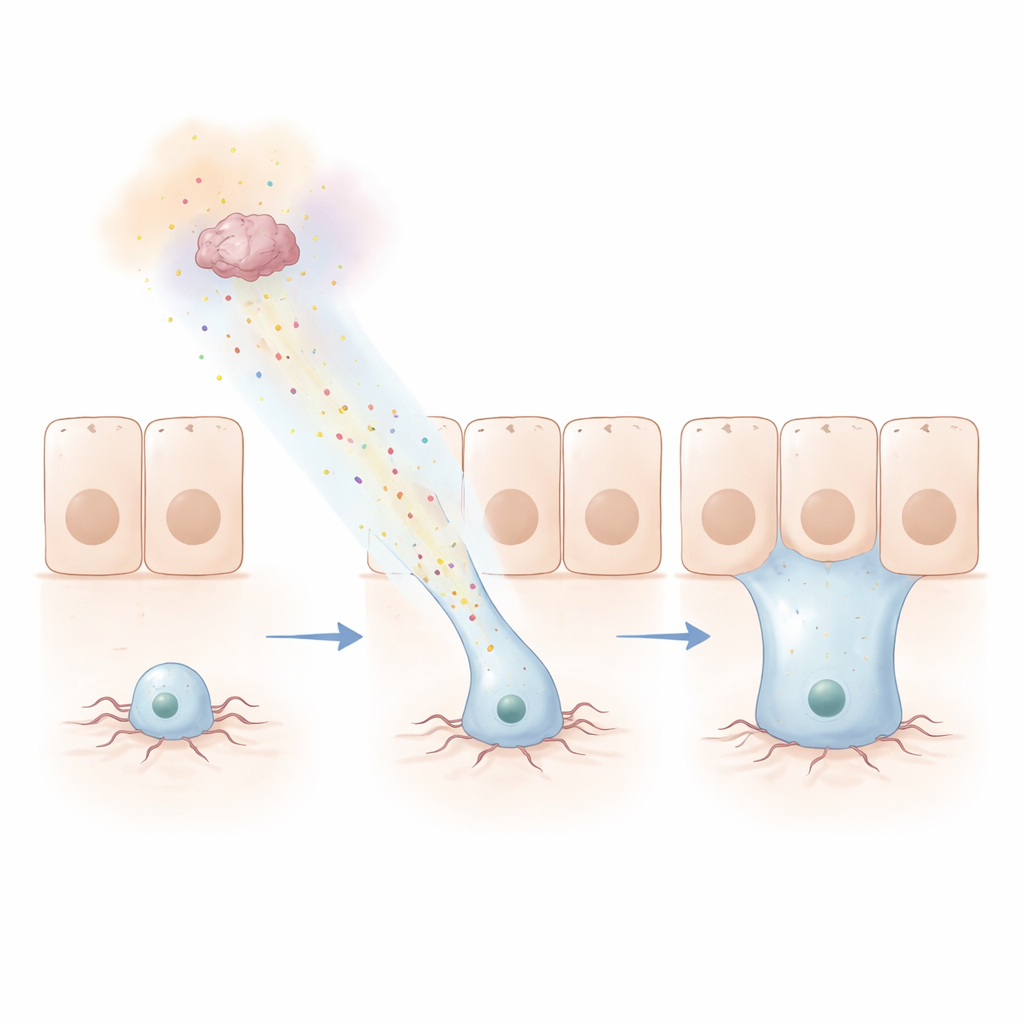
Following a Chemical Trail
To probe how far this signal can reach, the team built a clever “Hamelin” assay, named for the Pied Piper. They made a ring of cells at the junction between gut regions secrete Netrins, while fluorescently labeling progenitors at a distance. Over days, progenitors migrated up to tens of micrometers toward the source, even crossing a sharp boundary into a different tissue layer and integrating there. Human versions of Netrins and the DCC receptor could swap in for their fly counterparts and still guide these movements, showing that the mechanism is deeply conserved. The same actin machinery that powers cell movement elsewhere in the body was required: when key components were removed, protrusions and long-range renewal failed.
Why This Matters for Health and Disease
In simple terms, this work reveals that the gut does not rely only on local cell division pressure to renew its lining. Instead, dying cells send out a chemical “help” signal, Netrin-B, which attracts specific progenitors equipped with Frazzled/DCC receptors. These progenitors extend feelers along the gradient, crawl toward the aging cell, and slot into place to keep the barrier intact. Because the same Netrin–DCC system is active in mammals and has been linked to cancer invasion and metastasis, the fly findings provide concrete mechanistic support for viewing these molecules as double-edged: essential for orderly repair in healthy tissue, but potentially dangerous when misregulated in tumors that learn to move and colonize new organs.
Citation: Zipper, L., Ramon-Cañellas, P., Akkas-Gazzoni, F. et al. Frazzled/DCC directs spatial progenitor integration ensuring steady-state intestinal turnover. Nat Commun 17, 2491 (2026). https://doi.org/10.1038/s41467-026-70704-9
Keywords: intestinal stem cells, cell migration, netrin signaling, tissue homeostasis, cancer metastasis