Clear Sky Science · en
Implantable soft bladder-machine interface for neurogenic bladder dysfunction
Why this matters for everyday life
For many people with spinal cord injuries or other nerve problems, the bladder stops working properly: they cannot feel when it is full and it may squeeze at the wrong time. This can lead not only to embarrassing leaks but also to serious kidney damage. The article describes a new soft, implantable “bladder–machine” link that gently coats the bladder, senses how it moves, and delivers small pulses of electricity to calm misbehaving contractions. It points toward smarter, less intrusive care for people living with long‑term bladder problems.
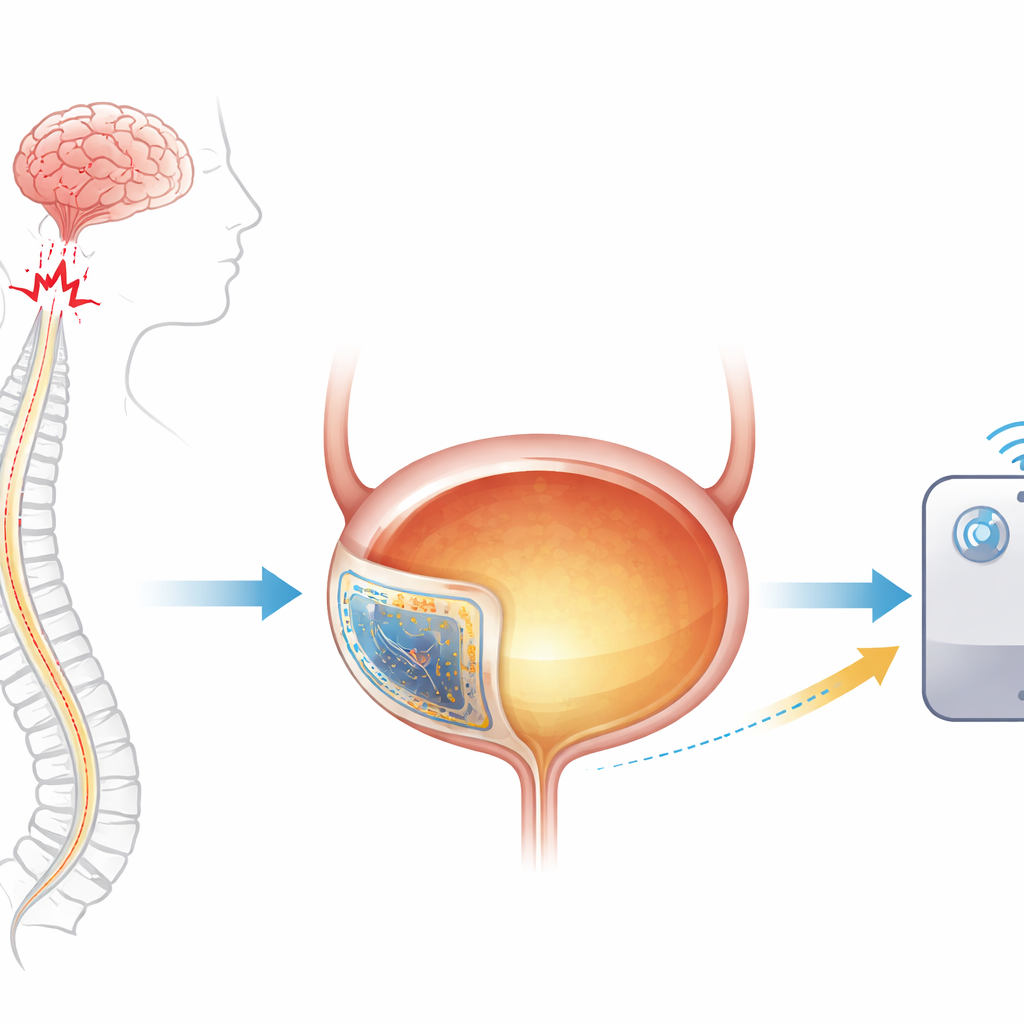
A common but hidden health problem
Conditions such as neurogenic bladder—often caused by spinal cord injury or diseases like multiple sclerosis—affect hundreds of millions of people worldwide. When nerves between the brain and the bladder are damaged, the bladder may overfill without warning, fail to empty, or contract uncontrollably. This raises pressure in the urinary tract and can ultimately injure the kidneys. Existing treatments rely on catheters, medicines with side effects, or bulky electrical devices that do not move well with the soft, balloon‑like bladder. Because the bladder expands in all directions when it fills, most traditional electronics either break, lose contact, or must be kept outside the body.
A soft electronic patch that stretches like a balloon
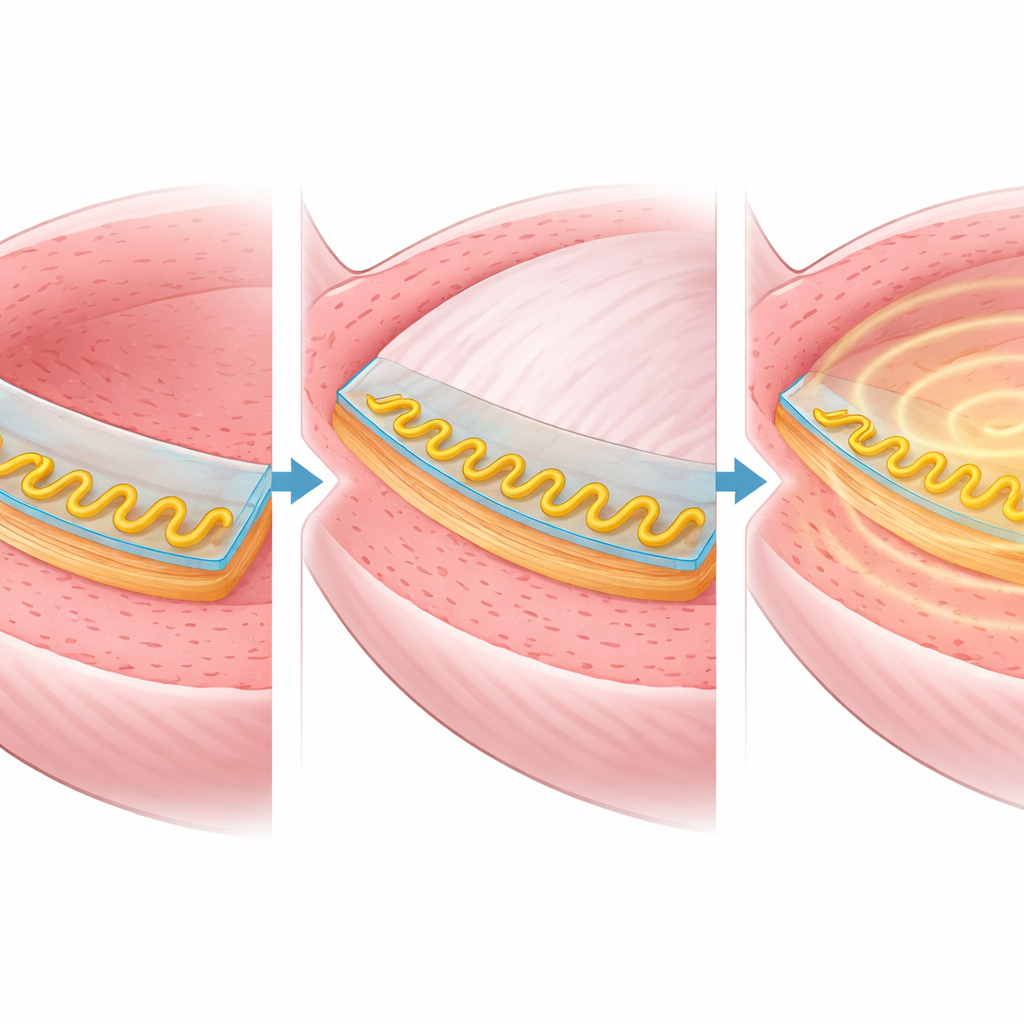
The researchers designed an implantable bladder–machine interface (BdMI) that behaves more like skin than metal. It is a thin, rubbery patch that can be stitched onto the outside of the bladder. Inside this patch lies a special gold film that conducts electricity even when stretched to many times its original area. Instead of cracking, the gold forms tiny, maze‑like grooves and “nano‑needles” that unfold smoothly as the bladder swells. In laboratory tests, the film survived being stretched more than eightfold in all directions and pulled back and forth tens of thousands of times while still carrying electrical signals, far beyond the demands of normal bladder filling and emptying.
How the patch senses and tames the bladder
Built on top of this stretchable film, the BdMI combines two main jobs. First, it works as a motion and pressure sensor: as the bladder wall expands, the electrical resistance of the patch changes, allowing the system to infer how full the bladder is and how strongly it is contracting. Second, small exposed areas on the patch act as electrodes that can deliver controlled electrical pulses to the bladder muscle. In rat experiments, the device was stitched onto the bladder and connected by thin wires to a small connector on the skull, which linked to standard recording and stimulation equipment. Over days of normal activity, the patch flexed and moved with the organ without tearing or peeling off.
Putting the system to the test in healthy and injured bladders
In healthy rats, the BdMI tracked bladder filling and emptying in real time. The resistance of the sensor rose gradually as the bladder filled, then changed sharply when the animal urinated, matching pressure readings from a traditional catheter. Electrical pulses through the electrodes could trigger bladder contractions on demand, showing that the device could both read and influence bladder behavior. The team then turned to a more demanding test: rats with spinal cord injury that develop an overactive, poorly controlled bladder. In these animals, the patch’s motion signal could distinguish meaningful contractions—those that cause or nearly cause urine release—from minor, clinically unimportant twitches. Statistical analysis showed that the size of the resistance change could serve as a biomarker to sort different types of contractions without relying on an internal pressure tube.
Early signs of therapy, not just monitoring
To explore treatment, some injured rats received daily electrical stimulation through the BdMI for a week, while others had the device implanted but received no active pulses. All groups showed similar overall bladder pressures and tissue health, indicating that the device and stimulation protocol were safe. Crucially, however, animals that received real stimulation had fewer unwanted bladder contractions than untreated injured rats, and their contraction frequency shifted toward that of uninjured animals. The strength of each contraction did not change, but the episodes became less frequent, suggesting that regular stimulation helped quiet the overactive bladder circuits without harming the tissue.
What this could mean for future patients
The study shows that a thin, ultrastretchy electronic patch can live on the moving surface of the bladder, sense its behavior continuously, and deliver gentle electrical therapy that reduces abnormal contractions in an animal model. For a layperson, the take‑home message is that engineers are getting closer to “smart” internal bandages that both listen to and talk to troubled organs. If similar devices can be adapted and proven safe in humans, people with neurogenic bladder might one day rely less on catheters and guesswork, and more on an implanted guardian that monitors their bladder and intervenes before damage or accidents occur.
Citation: Li, H., Wang, S., Yu, Q. et al. Implantable soft bladder-machine interface for neurogenic bladder dysfunction. Nat Commun 17, 2458 (2026). https://doi.org/10.1038/s41467-026-70680-0
Keywords: neurogenic bladder, implantable bioelectronics, soft sensors, electrical stimulation therapy, spinal cord injury