Clear Sky Science · en
Dual-axis myelination covariance drives the functional connectivity emergence during infancy
How Newborn Brains Get Connected So Quickly
Newborn babies already show surprisingly adult-like patterns of brain activity, even though their brain wiring is still under construction. This paper explores a key puzzle: how can infant brains coordinate distant regions so early in life, before the main communication highways of white matter are fully built? The authors argue that a more subtle feature of the brain’s outer layer—how its insulation grows in sync across regions—helps explain how early brain networks emerge and begin to support later behavior.
Looking Beyond the Brain’s Big Cables
For years, scientists have assumed that the brain’s functional networks mainly arise from the gradual myelination of white matter, the deep bundles of nerve fibers that accelerate electrical signals. But in newborns, these tracts are far from mature, reaching only a fraction of adult myelination, while resting brain activity already falls into recognizable networks. This mismatch suggests that long-distance wiring alone cannot explain how early brain communication works. The authors instead focus on gray matter, the thin outer sheet of the brain where nerve cell bodies live and where myelination begins earlier and follows its own timetable.
Two Ways the Cortical Insulation Grows Together
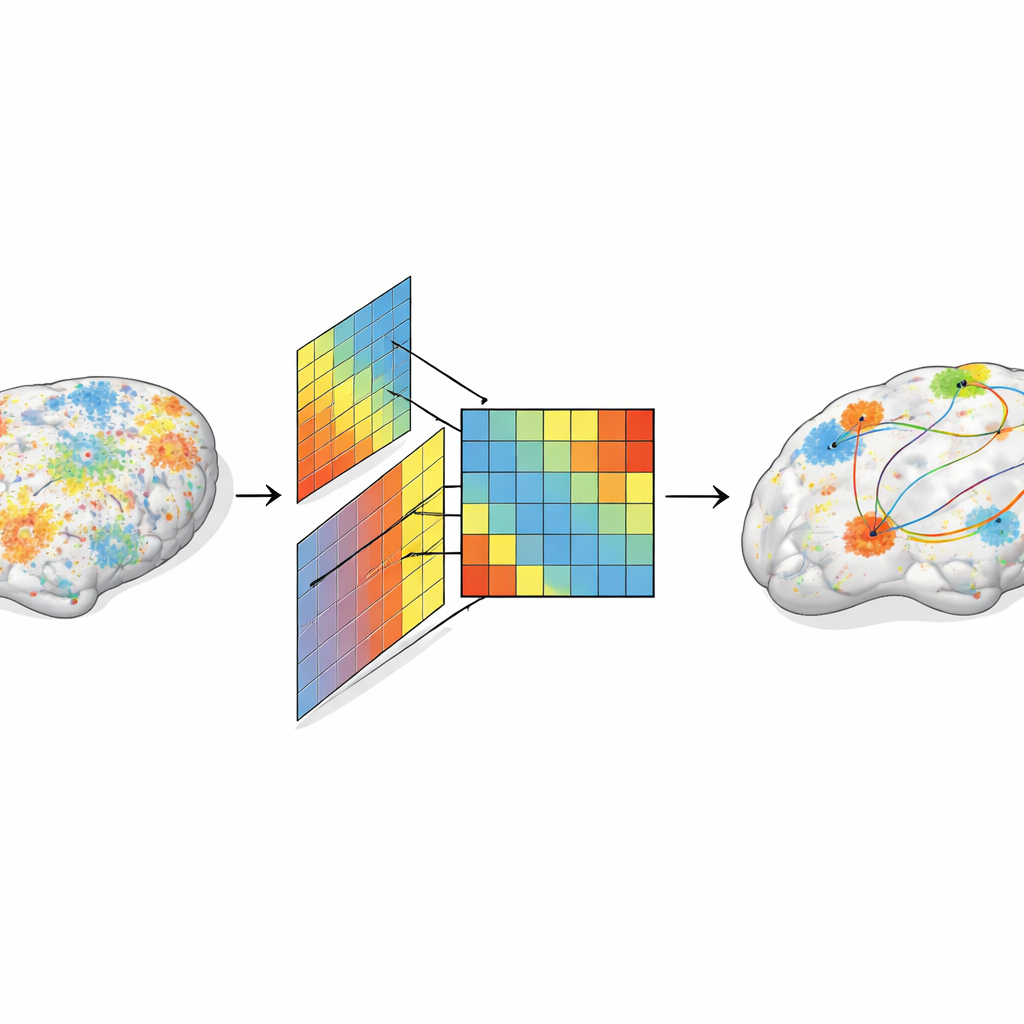
The team introduced a “dual-axis” framework to capture how myelination in the cortex changes in a coordinated way. One axis looks across babies: if the same pair of regions tends to be similarly myelinated in many infants, they share a group-level developmental pattern. The other axis looks within each baby’s brain: if two regions have similar myelin levels inside one infant, they share an individual-level pattern. From detailed MRI scans in hundreds of newborns, the researchers built maps of these covariances and then asked how well they could predict which regions show synchronized activity at rest, a standard measure of functional connectivity.
A New Measure Ties Structure to Activity
By combining both axes, the authors defined a myelination–function coupling index, or MFC, that reflects how strongly local myelination patterns line up with functional connections. They found that this index was highest in primary sensory and motor areas, as well as key regions like the insula and parts of the temporal lobe. MFC increased with age over the late fetal and early postnatal weeks, following a hierarchical progression: basic sensory and motor regions strengthened first, while higher-order networks grew more slowly. Importantly, this gray-matter-based coupling outperformed traditional measures based on white matter tracts, suggesting that early brain communication is strongly shaped by synchronized microstructural growth in the cortex itself.
Distance, Birth, and Genes All Play a Role
The study also shows that the strength and growth of this coupling depend on how far apart brain regions are, on whether development happens before or after birth, and on underlying gene activity. Nearby regions start out with stronger coupling, but it is the longer-range connections whose coupling grows most rapidly after birth, laying the groundwork for more complex coordination across the brain. When the authors separated time spent in the womb from time after birth, they found that gestational age had a stronger impact on MFC than time spent outside, underscoring the importance of the intrauterine environment. Yet extrauterine experience still mattered: full-term babies showed higher coupling in several associative regions than preterm infants scanned at the same postmenstrual age. Gene-expression data from fetal and neonatal brains revealed that regions with high MFC are enriched for genes involved in blood–brain barrier function, blood-vessel development, and the growth of myelin-producing glial cells, tying the observed patterns to specific biological processes.
Early Patterns That Forecast Later Abilities
Finally, the researchers connected these early brain patterns to behavior more than a year later. Infants whose brains showed stronger myelination–function coupling, especially in sensorimotor networks and in longer-range connections, tended to perform better on motor and other developmental measures at around 18 months of age. This suggests that how well cortical regions “grow together” structurally and functionally in the newborn period can foreshadow later skills. To a lay observer, the central message is that early brain function is not governed solely by the large, obvious nerve bundles. Instead, it also depends critically on the fine-tuned, synchronized maturation of the brain’s outer layers, guided by genetics, prenatal environment, and early experience. This view offers a richer picture of how healthy brain wiring emerges—and why disruptions before or shortly after birth might leave lasting marks on development.
Citation: Liu, W., Chen, Y., Wang, X. et al. Dual-axis myelination covariance drives the functional connectivity emergence during infancy. Nat Commun 17, 2624 (2026). https://doi.org/10.1038/s41467-026-70660-4
Keywords: infant brain development, myelination, functional connectivity, gray matter, neurodevelopment