Clear Sky Science · en
A myoneural actuator with engineered biophysics for implantable biohybrid systems
Rebuilding Muscles as Smart Living Machines
Imagine if doctors could turn a person’s own muscle into a living motor that never tires, can be steered by a computer, and can safely sit next to delicate organs for years. This study describes a way to do just that. The researchers engineer a “myoneural actuator” in rats—a muscle whose nerve wiring is deliberately rearranged so it can be driven by electronics, resist fatigue, and still behave like natural tissue. Such living actuators could one day restore movement, support failing organs, or deliver realistic sensations from advanced prosthetic limbs.
Why Living Muscles Make Better Motors
Artificial motors that run on electricity, air, or fluids are powerful, but they are heavy, hard to shrink to body-friendly sizes, and made from materials that do not blend well with living tissue. In contrast, skeletal muscle is light, energy-efficient, self-repairing, and already designed to work inside the body. It can grow, adapt, and respond to tiny nerve signals. However, when muscles are controlled directly by electrical pulses from an implanted device, they quickly tire. This fatigue happens because standard electrical stimulation tends to over-recruit the biggest, fastest, but least durable fibers first. To harness muscle as a dependable, implantable motor, the team needed to change how its nerve fibers are organized and recruited—without giving up the muscle’s natural health and sustainability.
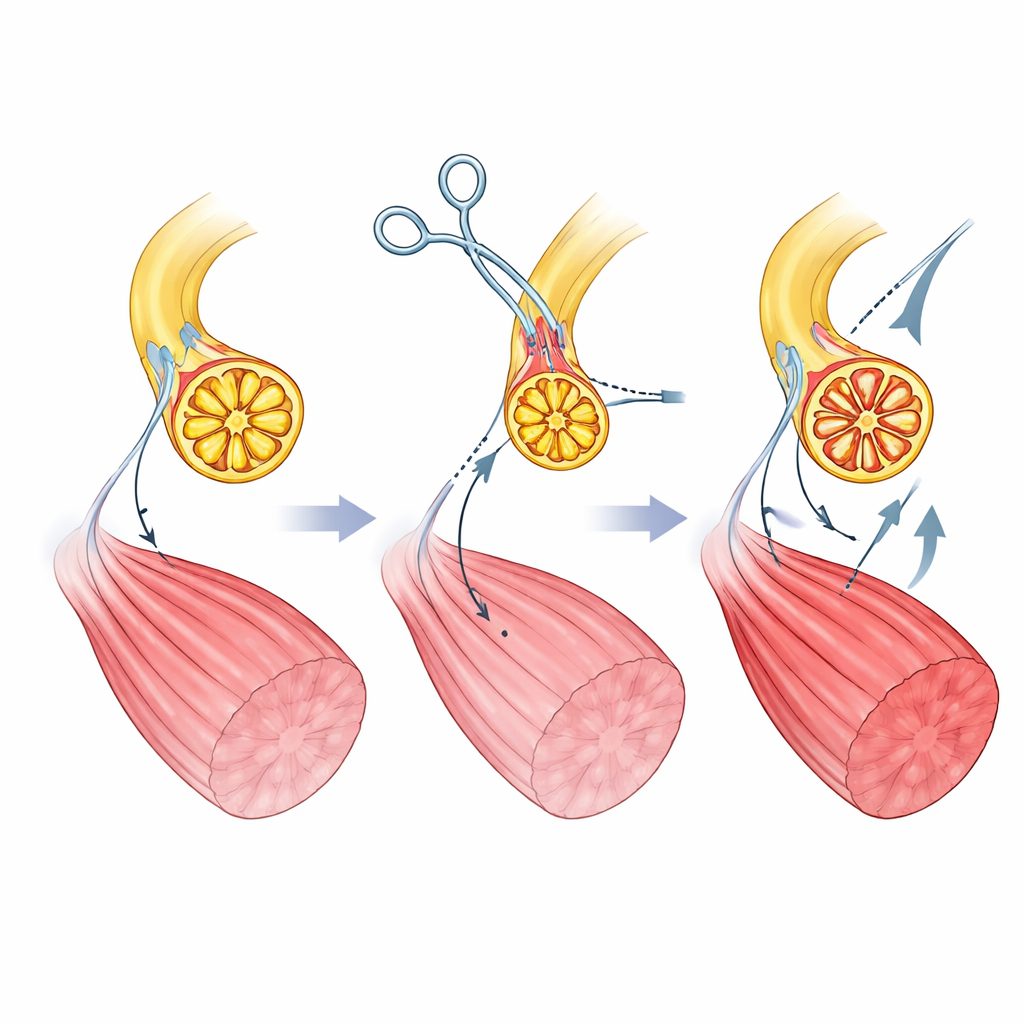
Swapping Nerve Pathways to Rewrite Control
The authors create their myoneural actuator in rats by surgically rewiring a leg muscle. They first cut the normal motor nerve that carries commands from the spinal cord and prevent it from growing back. Then they route a different nerve—one that usually carries touch and stretch signals from the skin and tissues—directly into the muscle. Over time, this “sensory” nerve regenerates inside the muscle and forms new contact points with muscle fibers. Microscopy shows that these new junctions look and behave like normal motor connections, and the muscle regains its ability to contract when the sensory nerve is electrically stimulated. Importantly, the axons in this sensory nerve are more uniform and smaller in size than those in the original motor nerve, setting the stage for more balanced muscle fiber recruitment under computer control.
Muscles That Keep Working When Pushed Hard
Once the rewired muscles had healed, the team put them through demanding test routines. They compared native muscles with the new actuators during repeated single twitches and during long stretches of continuous contraction. The myoneural actuators lost force much more slowly and showed steadier output over time, with a 260 percent improvement in fatigue resistance under continuous work. Their behavior over minutes looked fundamentally different from unmodified muscle: instead of a rapid drop in strength, force declined more gradually and then stabilized. Despite losing some mass during the rewiring process, the actuators preserved healthy fiber structure and produced similar force per unit mass as native muscle, and they maintained their condition for at least 15 weeks even without exercise or additional stimulation.
Computer Control Without Disturbing the Brain
Because the new nerve wiring disconnects the muscle from the brain’s normal motor signals, all contractions now come from an external stimulator. The researchers embed the muscle in a feedback loop: a sensor measures force, a controller adjusts the nerve stimulation, and the system tracks a chosen target force over many cycles. Native muscles quickly fail in this setup, but the myoneural actuators keep following the command signals. To make the system safer and more flexible, the team also introduces a reversible “nerve block.” By applying a high-frequency electrical signal on the nerve closer to the spinal cord, they prevent stimulation pulses from sending unwanted signals back to the central nervous system while still allowing the muscle side to respond. In tests, the actuator continued to generate controlled forces while the brain remained effectively insulated from the artificial stimulation.
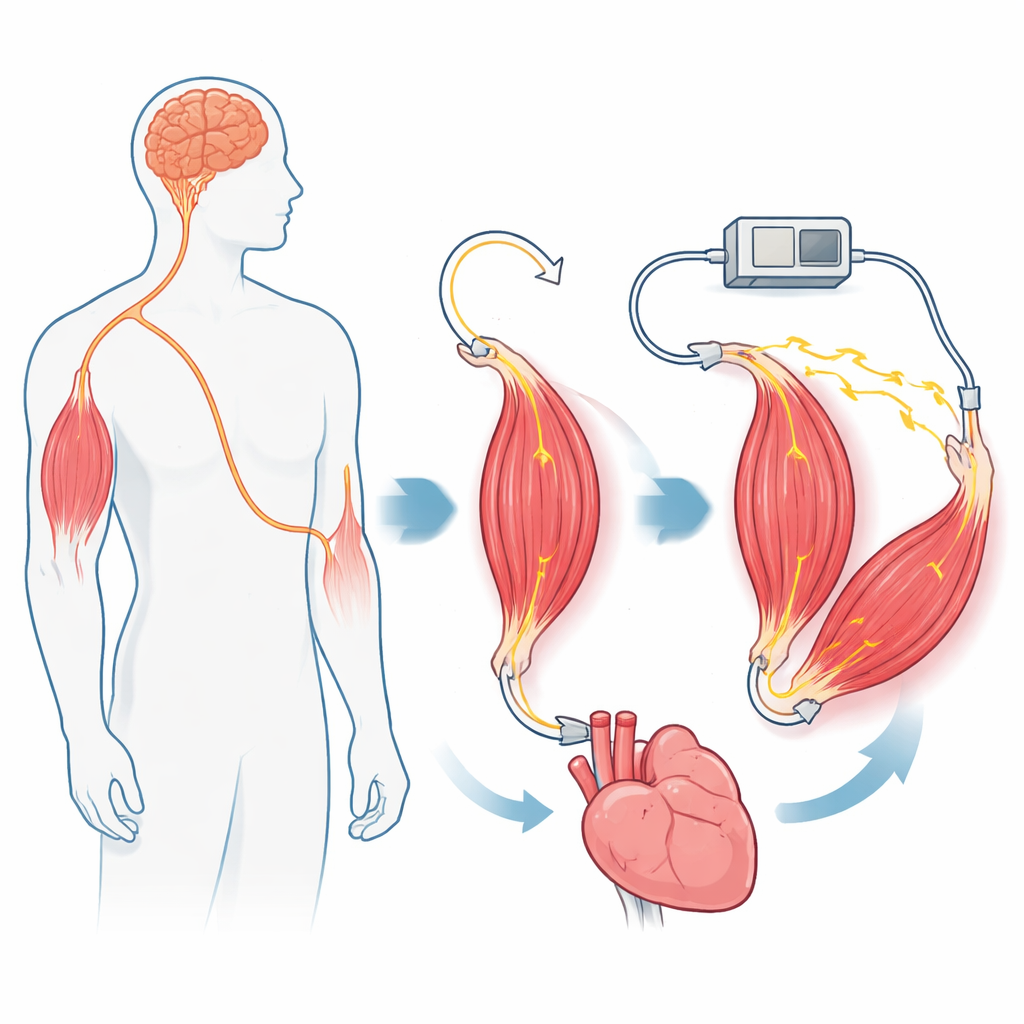
From Bionic Limbs to Helping Failing Organs
To show how this living motor could be used, the authors build two demonstration systems in rats. In the first, they couple the actuator in series with another muscle that stands in for the leftover muscle at an amputation site. By tightening or relaxing this pair, they change the stretch of the residual muscle and, in turn, the activity of its sensory nerve fibers—essentially dialing up or down the limb’s sense of position and force. This “proprioceptive mechanoneural interface” could eventually provide natural-feeling feedback from robotic limbs or virtual avatars. In the second system, they wrap the actuator around a loop of small intestine filled with fluid. When driven by nerve stimulation, the muscle squeezes and releases the gut segment, and motion tracking shows that the organ moves in step with the actuator. This hints at future devices that could lend mechanical strength to weakened organs like the intestine, bladder, or even heart.
What This Could Mean for Future Medicine
Overall, this work shows that by carefully rerouting nerves, a person’s own muscle can be turned into a fatigue-resistant, computer-driven actuator that still behaves like native tissue and can be electrically isolated from the brain when needed. Because the approach relies on surgical techniques and electrodes that already resemble existing clinical tools, it may be easier to translate than fully synthetic implants or lab-grown tissues. If similar myoneural actuators can be built and controlled safely in humans, they could form the core of new biohybrid systems that restore limb sensation, assist failing organs, and provide precise mechanical cues to the body without the bulk and stiffness of traditional machines.
Citation: Song, H., Herrera-Arcos, G., Friedman, G.N. et al. A myoneural actuator with engineered biophysics for implantable biohybrid systems. Nat Commun 17, 2584 (2026). https://doi.org/10.1038/s41467-026-70626-6
Keywords: biohybrid actuator, neuroprosthetics, muscle fatigue, nerve regeneration, organ support