Clear Sky Science · en
Bidirectional catalysts with dual-atom dynamic d-band centre modulation and support self-reconstruction for de/hydrogenation in MgH2/Mg
Safer fuel for a clean-energy future
Hydrogen is often hailed as a clean fuel of the future, but storing it safely and compactly remains a major challenge. This study tackles that problem by improving how a promising storage material, magnesium hydride, takes up and releases hydrogen. The researchers created a tiny, precisely built catalyst that speeds both directions of the reaction—loading hydrogen in and pushing it back out—while working at lower temperatures and staying stable over many cycles. Their approach could help make hydrogen storage safer, more efficient, and more practical for large-scale use.
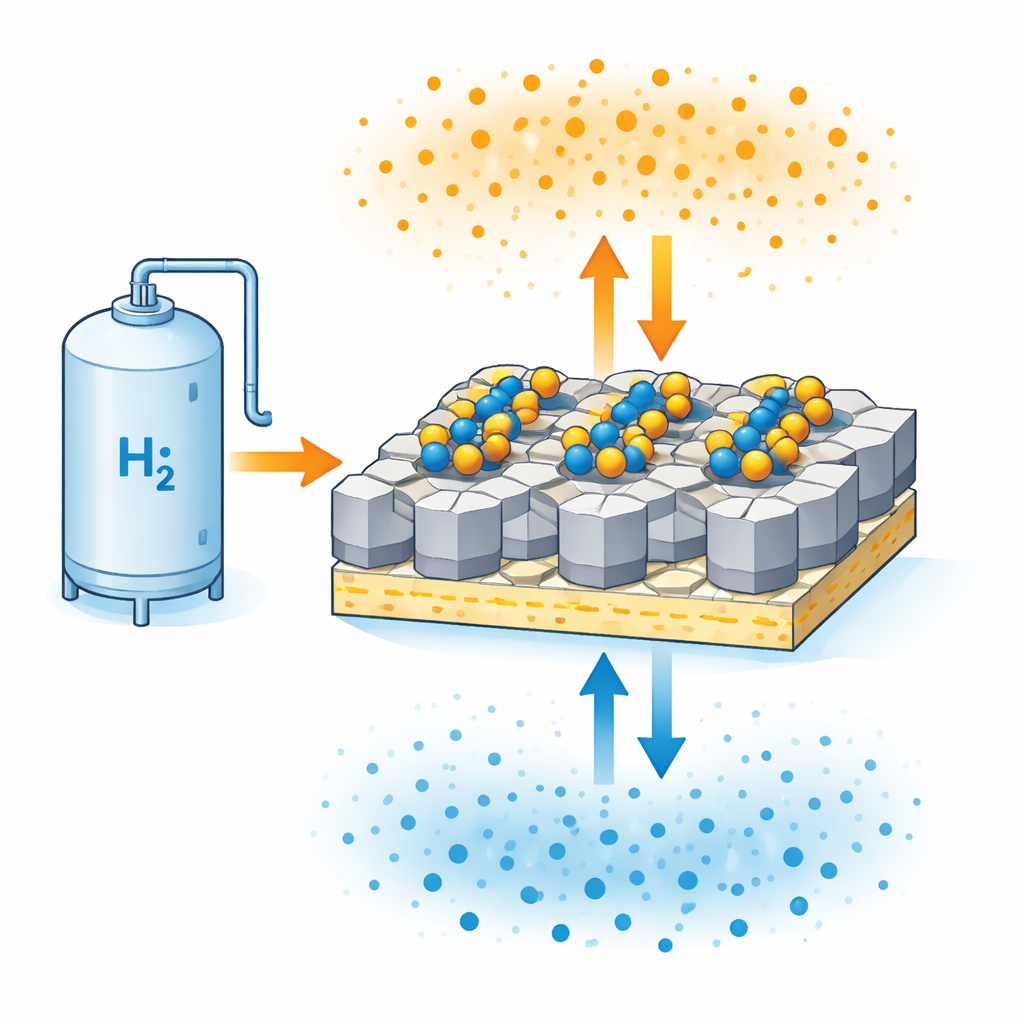
Why storing hydrogen in solids is hard
Instead of compressing hydrogen gas into heavy tanks, one attractive option is to store it inside solids, where hydrogen atoms tuck into the material’s structure. Magnesium hydride is especially appealing because it can hold a large amount of hydrogen by weight and is relatively safe. The drawback is that it only absorbs and releases hydrogen quickly at high temperatures, and the reaction itself is sluggish. Earlier attempts to fix this relied on adding simple metal particles or single-atom catalysts that worked well in one direction, usually helping hydrogen come out more easily but not doing as well when it needed to go back in. That imbalance limits how useful the material can be in real devices that must charge and discharge repeatedly.
A tiny team of atoms with shared duties
The authors designed a new type of catalyst built from pairs of different metal atoms—nickel and cobalt—anchored on the surface of titanium oxide. These dual-atom pairs are spread out one by one across the support, rather than clumping into bigger particles. Computer simulations showed that when nickel and cobalt sit next to each other, they subtly reshape each other’s electronic structure. As a result, nickel becomes especially good at breaking the bond between magnesium and hydrogen when the material is releasing gas, while cobalt becomes especially good at splitting incoming hydrogen molecules when the material is taking up gas. The titanium oxide support also plays an active role: it can form defects and change its own oxidation state, which helps shuttle electrons and keep the metal atoms from wandering and clumping together.
How the new material behaves in practice
To test the concept, the team mixed a small amount of the dual-atom catalyst into magnesium hydride using ball milling, a process that grinds the materials together down to very fine scales. Microscopy and spectroscopy confirmed that nickel and cobalt remained isolated or paired on the titanium oxide, and that the catalyst particles coated the magnesium hydride evenly. When the researchers heated the material and monitored hydrogen release, they found that the starting temperature for gas coming out dropped dramatically, by more than 200 degrees Celsius compared to untreated magnesium hydride. The rate of hydrogen release increased sharply as well, and the overall energy barrier for the reaction fell to about one-third of its original value.
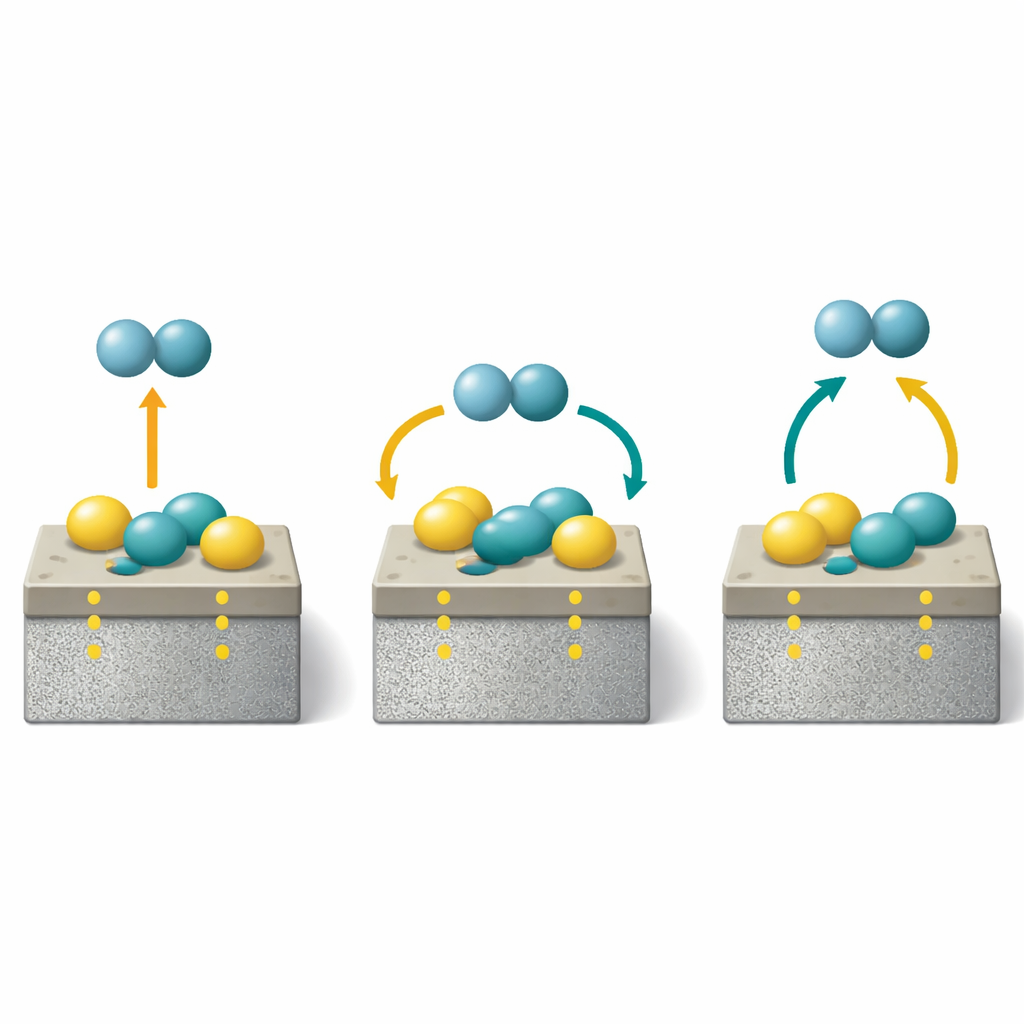
Fast in, fast out, and built to last
The benefits were just as striking when the material absorbed hydrogen. Under moderate pressure, the catalyst-treated magnesium alloy could take up several percent by weight of hydrogen even at room temperature, and it did so quickly. At somewhat higher temperatures, it reached near-full capacity in seconds. Crucially, this rapid performance did not fade with use: after 100 cycles of hydrogen loading and unloading, the material still kept almost all of its storage capacity. Detailed measurements suggest that the titanium oxide support continuously rearranges its internal defects in a reversible way, while the strong bonding between the metals and the support prevents the nickel and cobalt atoms from aggregating, preserving the finely tuned catalytic structure.
What this means for hydrogen technology
In everyday terms, the researchers have taught a solid material to “breathe” hydrogen in and out more easily, using a carefully choreographed duo of metal atoms sitting on a smart support. By lowering the temperatures and energy penalties needed for both storing and releasing hydrogen, and by maintaining performance over many cycles, this approach brings magnesium-based storage closer to practical use in systems such as fuel-cell vehicles or backup power units. More broadly, the work offers a recipe for designing other reversible catalysts in which different atoms share and swap roles during charging and discharging, potentially benefiting many chemical processes that must run efficiently in both directions.
Citation: Jin, J., Zhang, J., Zhang, J. et al. Bidirectional catalysts with dual-atom dynamic d-band centre modulation and support self-reconstruction for de/hydrogenation in MgH2/Mg. Nat Commun 17, 2447 (2026). https://doi.org/10.1038/s41467-026-70604-y
Keywords: hydrogen storage, magnesium hydride, catalyst design, dual-atom catalysts, clean energy materials