Clear Sky Science · en
Alternating-sequence polymer chain facilitating Li+ transport in covalent organic frameworks
Safer, Faster-Charging Batteries
Modern gadgets and electric cars all rely on lithium-ion batteries, but the flammable liquid inside today’s batteries can catch fire if damaged or overheated. Solid batteries that replace this liquid with a solid material promise much better safety and faster charging, yet many prototypes still move lithium ions too slowly. This article describes a new kind of solid material that lets lithium ions flow quickly and cleanly, opening a path toward safer, long‑lasting, fast‑charging batteries.
Building a Better Pathway for Ions
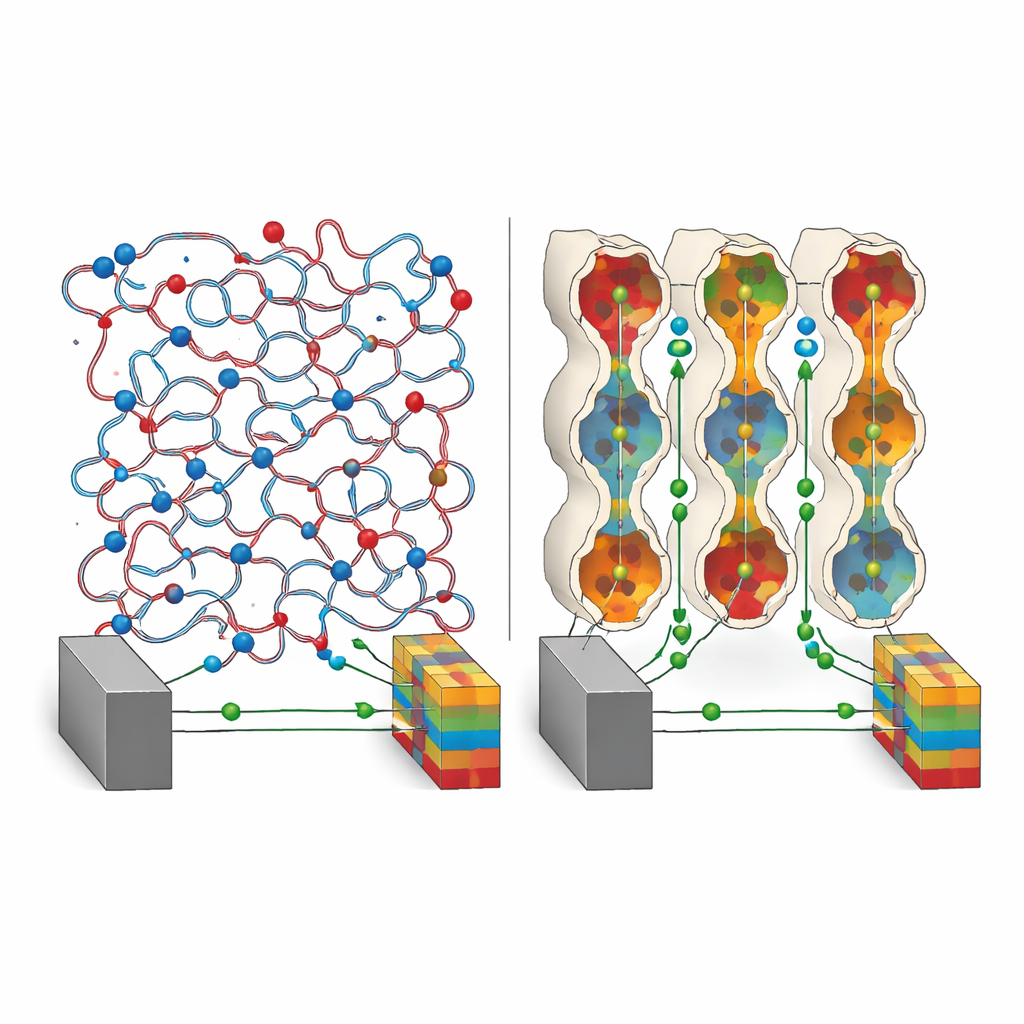
The heart of this work is a family of solids called covalent organic frameworks, or COFs. These are rigid, sponge‑like crystals made from light elements such as carbon, nitrogen, and oxygen, full of tiny, regularly arranged pores. COFs are attractive as battery electrolytes because their structure can be precisely designed. However, in earlier versions, the pores were essentially empty tunnels: they did not guide lithium ions very well, anions wandered freely, and overall ion flow was modest. The authors set out to redesign the inner lining of these pores so that lithium ions would see a continuous, well‑marked highway instead of a rough mountain trail.
An Alternating Chain Inside Tiny Pores
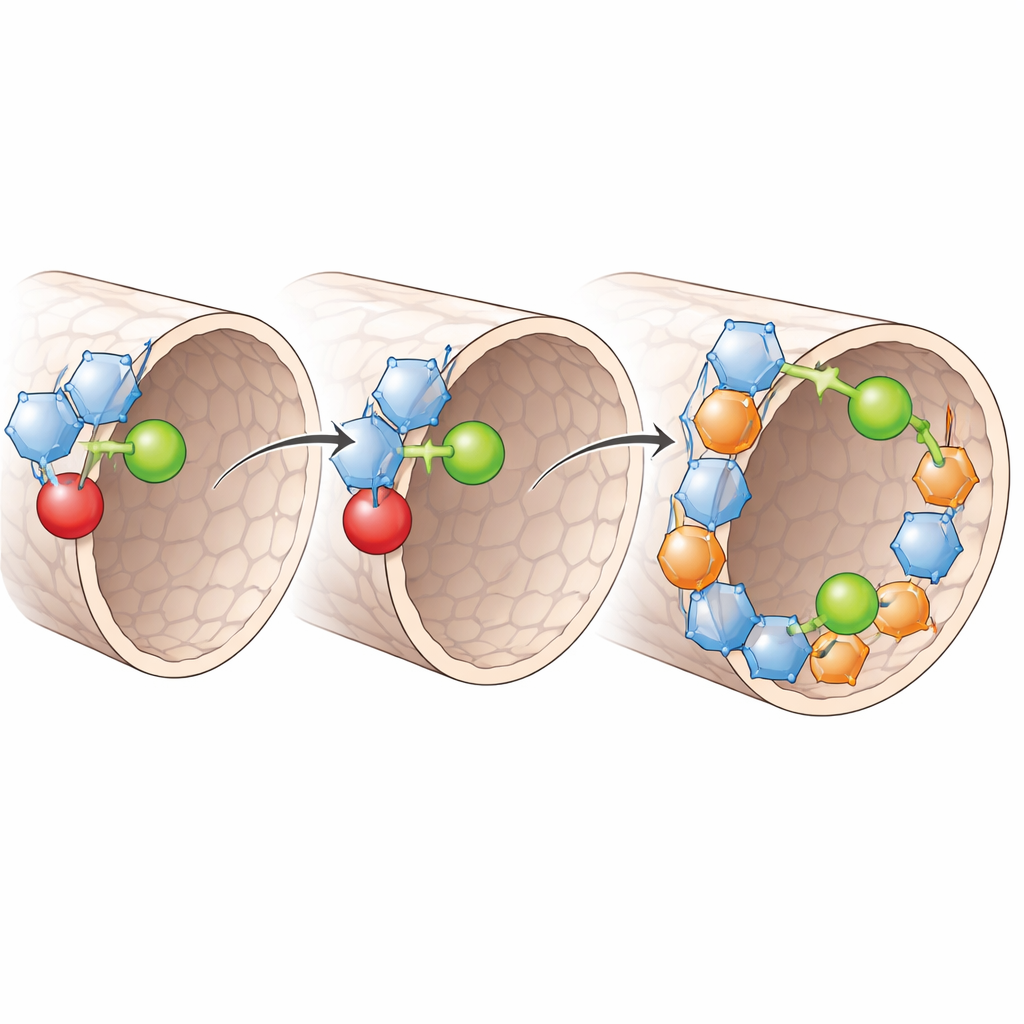
The researchers created a new COF, called PF–COF, by threading two types of short polymer segments into the pores in an alternating sequence. One segment resembles a familiar plastic (polyethylene oxide) that readily binds lithium ions and helps them hop from site to site. The other is a fluorine‑rich segment that strongly pulls electrons and stabilizes the material at high voltages. By alternating these two along the pore walls, the team engineered a repeating pattern of lithium‑friendly spots and electron‑withdrawing spots that reshape how charge is distributed inside the pores. Computer simulations and spectroscopy show that this pattern breaks up clumps of lithium salt, spreads lithium ions more evenly, and reduces their tendency to pair tightly with their negative partners.
Letting Lithium Through While Holding Anions Back
Measurements reveal that PF–COF conducts lithium ions unusually well for a solid, with a conductivity above 10−3 siemens per centimeter at room temperature. Just as important, almost all of the current is carried by lithium ions rather than by the accompanying anions: the lithium “transference number” reaches 0.9, a value typically seen only in specialized single‑ion conductors. This happens because the fluorinated segments give the pore walls an overall positive character that pins the negatively charged anions in place. Lithium ions, attracted and guided by the oxygen‑rich segments, then move along a continuous chain of sites from one end of the pore to the other. The result is a solid electrolyte that both speeds up lithium motion and cuts down on energy‑wasting traffic from other ions.
Stable Interfaces and Long Battery Life
Beyond ion flow inside the pores, the new material also improves what happens where the solid meets the metal lithium electrode. When used in a simple lithium‑on‑lithium test cell, the PF–COF electrolyte supports smooth plating and stripping of lithium for more than 7,500 hours with very small voltage changes, and microscopic images show a flat metal surface with few needle‑like “dendrites.” Detailed analysis reveals that the electrolyte helps form a thin, robust protective layer rich in lithium fluoride and lithium oxide, which stabilizes the interface and blocks dangerous growths. In full cells paired with a high‑energy nickel‑rich cathode (NCM811), the solid electrolyte delivers high capacity, excellent stability over hundreds of cycles, and unusually strong performance even at very high charge and discharge rates, where many other solid systems quickly fade.
What This Means for Future Batteries
By carefully decorating the inner walls of a porous crystal with an alternating sequence of short chains, the authors turn COFs into highly selective highways for lithium ions. This design both accelerates ion movement and protects the battery’s internal surfaces, enabling fast charging, long life, and compatibility with powerful cathode materials. For non‑specialists, the key message is that smart nano‑architecture – not just new chemicals – can make solid batteries safer and more practical, bringing next‑generation energy storage for electronics and electric vehicles a significant step closer.
Citation: Zhao, G., Yang, M., Zhang, Z. et al. Alternating-sequence polymer chain facilitating Li+ transport in covalent organic frameworks. Nat Commun 17, 2442 (2026). https://doi.org/10.1038/s41467-026-70591-0
Keywords: solid-state lithium batteries, covalent organic frameworks, lithium-ion transport, fast charging, battery safety