Clear Sky Science · en
Decoupling electronic and geometric effects in Pd catalysts via thermal surface reconstruction for selective hydrogenation
Turning Heat into a Tool for Cleaner Chemistry
Chemists depend on solid catalysts to make everything from medicines to plastics, but the tiny metal particles that do the work are complicated and fickle. In this study, researchers show how a simple heat treatment can reshape and rewire palladium particles so they hydrogenate alkynes—an important class of chemical building blocks—much more efficiently and selectively. By learning how to tune both the shape and the electronic nature of these particles in a controlled way, they provide a roadmap for cleaner industrial reactions with fewer unwanted by‑products.
Why Catalyst Shape and Charge Matter
On the surface of a solid catalyst, atoms are arranged in a landscape of terraces, edges, and corners. Where a molecule lands in this landscape often determines what reaction it undergoes. At the same time, the electronic character of the surface—how electron rich or electron poor the metal atoms are—controls how strongly molecules stick and how easily bonds are broken or formed. In real catalysts, these geometric and electronic factors are usually tangled together, making it hard to say which one is actually responsible for better performance. Untangling them is essential if we want to design catalysts on purpose instead of by trial and error.
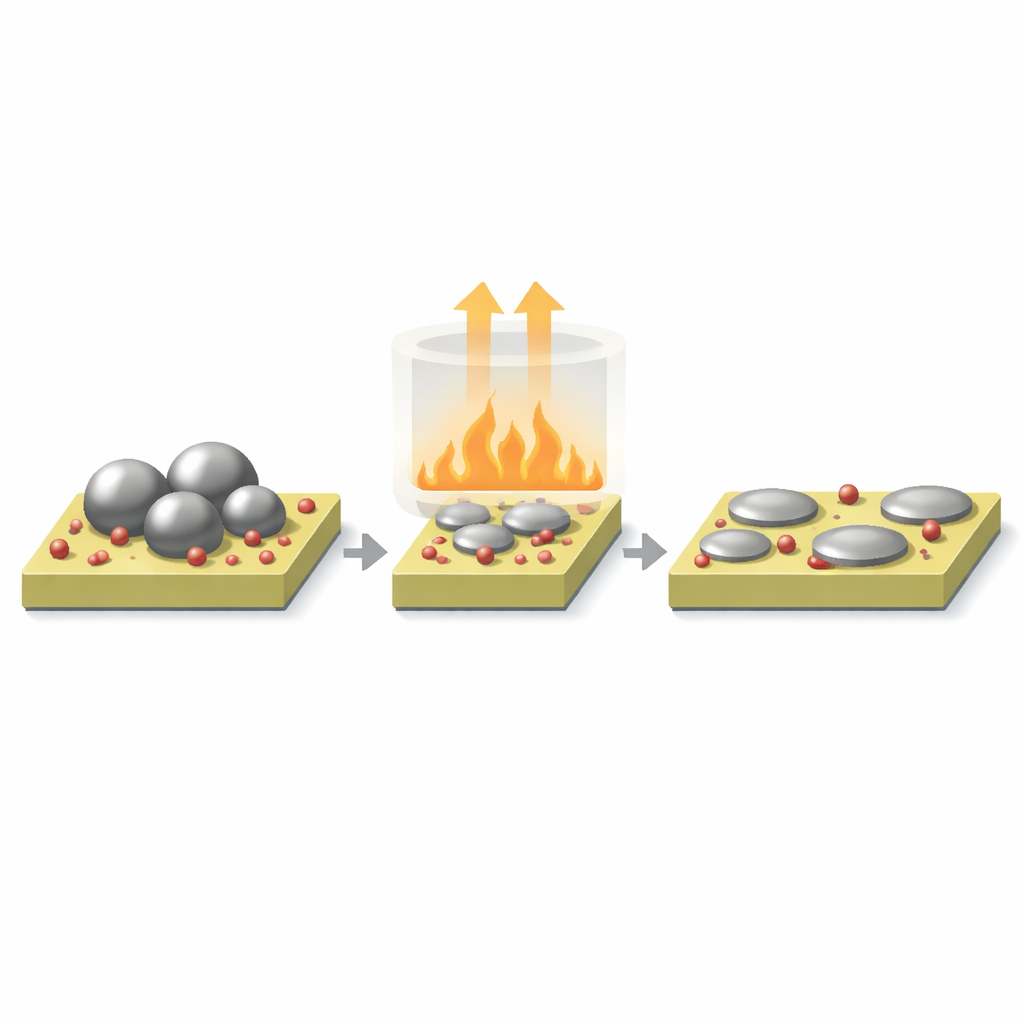
Using Heat to Reshape Palladium on Ceria
The team focused on palladium particles supported on cerium oxide, a so‑called reducible oxide that can exchange oxygen with metals on its surface. By heating these materials in air at controlled temperatures, they triggered a process called surface reconstruction. Larger, roughly spherical palladium nanoparticles broke up and spread out into much flatter, “puddle‑like” islands that made more intimate contact with the support. At the same time, electrons flowed from palladium into the cerium oxide, leaving many surface palladium atoms slightly electron‑poor. Microscopy, gas adsorption, and X‑ray measurements all confirmed that heating created highly dispersed, flattened palladium structures with strong metal–support interactions.
Linking Shape to Speed and Strength of Binding
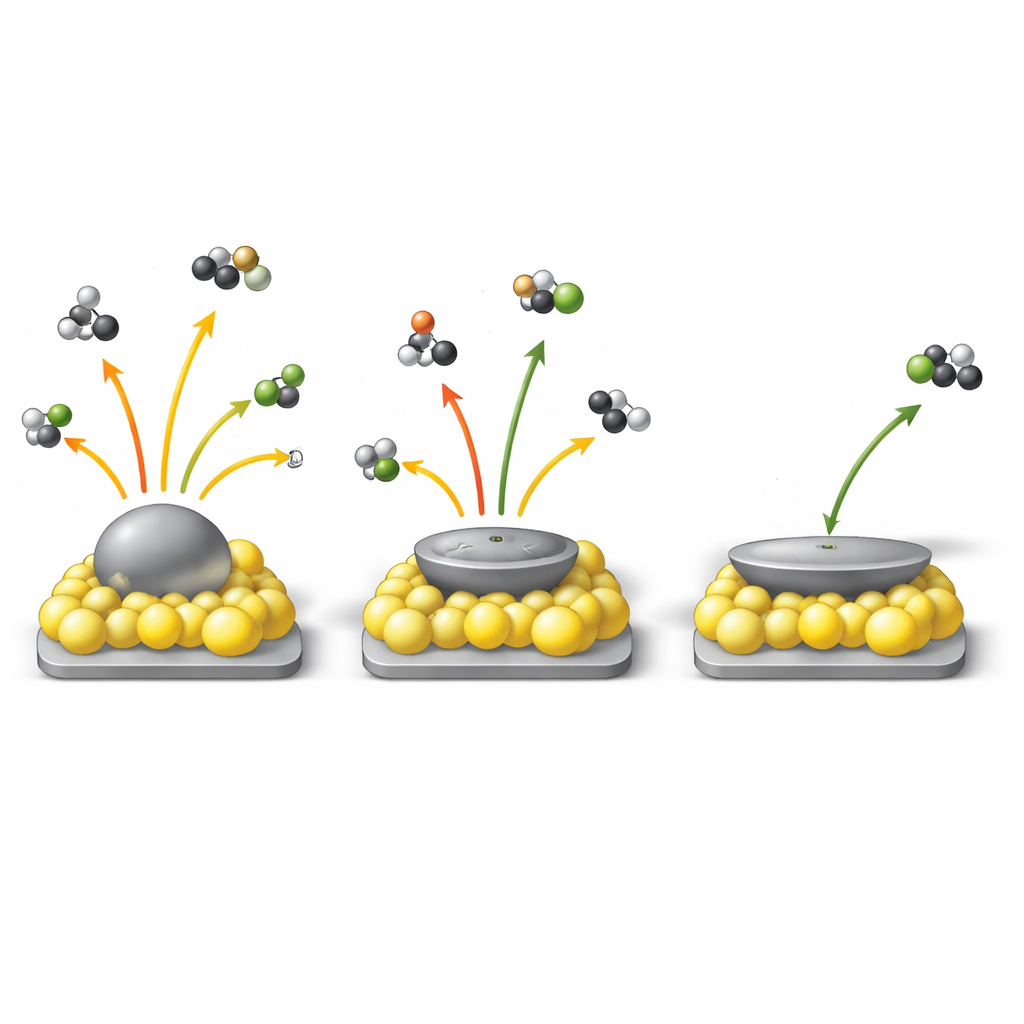
To test what this restructuring meant in practice, the researchers chose the semi‑hydrogenation of an alkyne called 2‑methyl‑3‑butyn‑2‑ol, a demanding reaction where the goal is to stop at a desired alkene instead of over‑reducing it. They quantified particle “flatness” with a simple geometric measure called W, the ratio of a particle’s short to long diameter in images: the smaller W is, the flatter the particle. Across many samples and preparation conditions, the reaction turnover frequency—the number of molecules each surface palladium atom converts per hour—scaled linearly with W: flatter particles were consistently more active. Kinetic studies and computer calculations showed why: as reconstruction increased the fraction of electron‑poor palladium sites, the alkyne no longer clung so strongly to the surface, reducing “self‑poisoning” and freeing sites for reaction.
How Selectivity Switches from Charge‑Controlled to Shape‑Controlled
The story for selectivity—the ability to stop at the desired alkene—was more subtle. When the particles were sufficiently flat (W below about 0.85), selectivity stayed on a high plateau, above roughly 96%, even though the detailed geometry continued to change. In this regime, the electron‑poor palladium created by strong bonding to cerium oxide dominated behavior, weakening the tendency of reactive sites to over‑hydrogenate the alkene. Once W rose above the threshold, however, palladium surfaces became more electron‑rich and the proportion of low‑coordination edges and corners grew. There, geometry took over: these sites strongly bind the alkene product and promote unwanted extra hydrogenation, so selectivity dropped sharply. By systematically varying particle size, support, and heat‑treatment conditions, the authors mapped these trends into contour diagrams that show where in “shape–charge” space over‑hydrogenation can occur and where it is effectively shut down.
A Simple Recipe for Better Hydrogenation Catalysts
In practical terms, the optimized thermally reconstructed palladium on cerium oxide outperformed a classic industrial Lindlar catalyst by more than an order of magnitude in activity while delivering around 97.5% selectivity, and it did so without toxic additives. Perhaps more importantly, the work offers a quantitative framework: a single geometric descriptor (W) and a measure of electron‑poor palladium content together predict not just how fast the catalyst will run, but whether it will favor the desired product. This blueprint should be transferable to other metals and supports where geometry and electronic structure are intertwined, helping chemists deliberately design catalysts that are both efficient and clean.
Citation: Li, M., Fu, Z., Luo, Q. et al. Decoupling electronic and geometric effects in Pd catalysts via thermal surface reconstruction for selective hydrogenation. Nat Commun 17, 2500 (2026). https://doi.org/10.1038/s41467-026-70568-z
Keywords: palladium catalysis, selective hydrogenation, metal–support interaction, ceria-supported catalysts, surface reconstruction