Clear Sky Science · en
StrucGAP: a modular, streamlined and traceable data mining platform for structural and site-specific glycoproteomics
Making Sense of Sugar Coats on Proteins
Every cell in our body is covered with a forest of sugary structures attached to proteins. These “sugar coats,” known as glycans, quietly help control how cells stick together, communicate, and respond to their environment. Modern instruments can now catalog these sugar patterns in astonishing detail, but researchers often drown in the sheer volume and complexity of the data. This study introduces StrucGAP, a new computational platform designed to turn those dense measurements into clear, biologically meaningful stories, using aging of the mouse uterus as a test case.
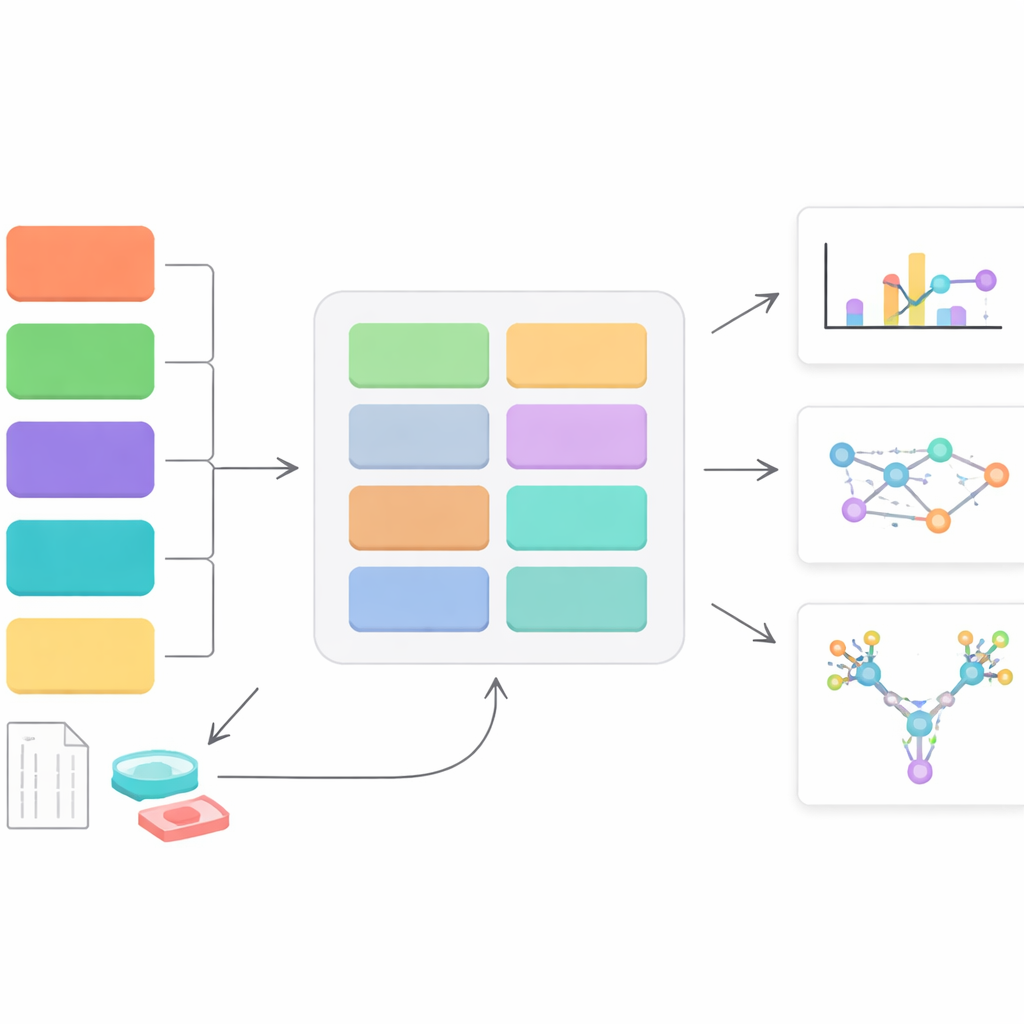
A New Control Center for Sugar-Coat Data
StrucGAP is a software platform built to analyze one specific kind of sugar decoration: N-linked glycans, which attach to particular spots on proteins. Instead of trying to be a general-purpose tool, StrucGAP is crafted from the ground up for this problem. It accepts results from several popular mass-spectrometry “search engines” that identify which glycans sit on which protein sites. Once those identifications are in, StrucGAP runs them through a series of modules that check data quality, summarize overall glycan patterns, track how specific sites change between conditions, and link those changes to known biological functions and pathways.
Breaking Complex Sugars into Meaningful Parts
Most existing tools treat each glycan as a single, indivisible object. StrucGAP takes a different approach: it breaks every glycan into smaller, biologically meaningful building blocks, such as common cores, branching patterns, and well-known motifs that include fucose or sialic acid sugars. It then asks not just which whole glycans go up or down, but which motifs appear more often, less often, or in new combinations. This “substructure” view makes the analysis more robust to uncertain assignments and helps uncover patterns that might otherwise be hidden, especially when rare but important motifs become enriched in a specific condition.
Following Sugar Changes During Uterine Aging
To show what StrucGAP can do, the authors applied it to a detailed dataset from the uterine tissue of young and middle-aged female mice. The raw experiment identified more than twenty thousand unique glycopeptides, each representing a particular protein site carrying a specific glycan. StrucGAP first cleaned and standardized the data, then charted how glycans were distributed across protein sites and how many structural variations appeared at each position. The uterus proved to be rich in both simple high-mannose glycans and more elaborate complex types, with many glycan compositions existing as multiple structural isomers. By zooming down to substructures, the platform cataloged how often different cores, branch counts, and motifs such as Lewis epitopes or particular forms of sialic acid occurred and co-occurred.
From Patterns to Function: Adhesion and Remodeling
StrucGAP’s quantitative module then compared young and aged uteri, finding over a thousand glycopeptides that increased and a few hundred that decreased with age. A recurring theme was “core fucosylation” – a specific way a fucose sugar attaches to the glycan core – which showed changes in both directions, suggesting finely tuned regulation rather than a simple on–off switch. Other patterns emerged as the statistical thresholds were tightened: glycans with more branches, specific Lewis-type motifs, and Neu5Ac-containing sialic acids became progressively enriched. By linking these structural features to databases of gene functions and pathways, StrucGAP revealed that the changing glycans were concentrated on proteins involved in cell adhesion, interactions with the surrounding matrix, and remodeling of tissue architecture. The platform also connected these patterns to shifts in the enzymes that build and trim glycans, as well as to glycan-binding proteins, sketching out coordinated networks that may drive uterine aging.
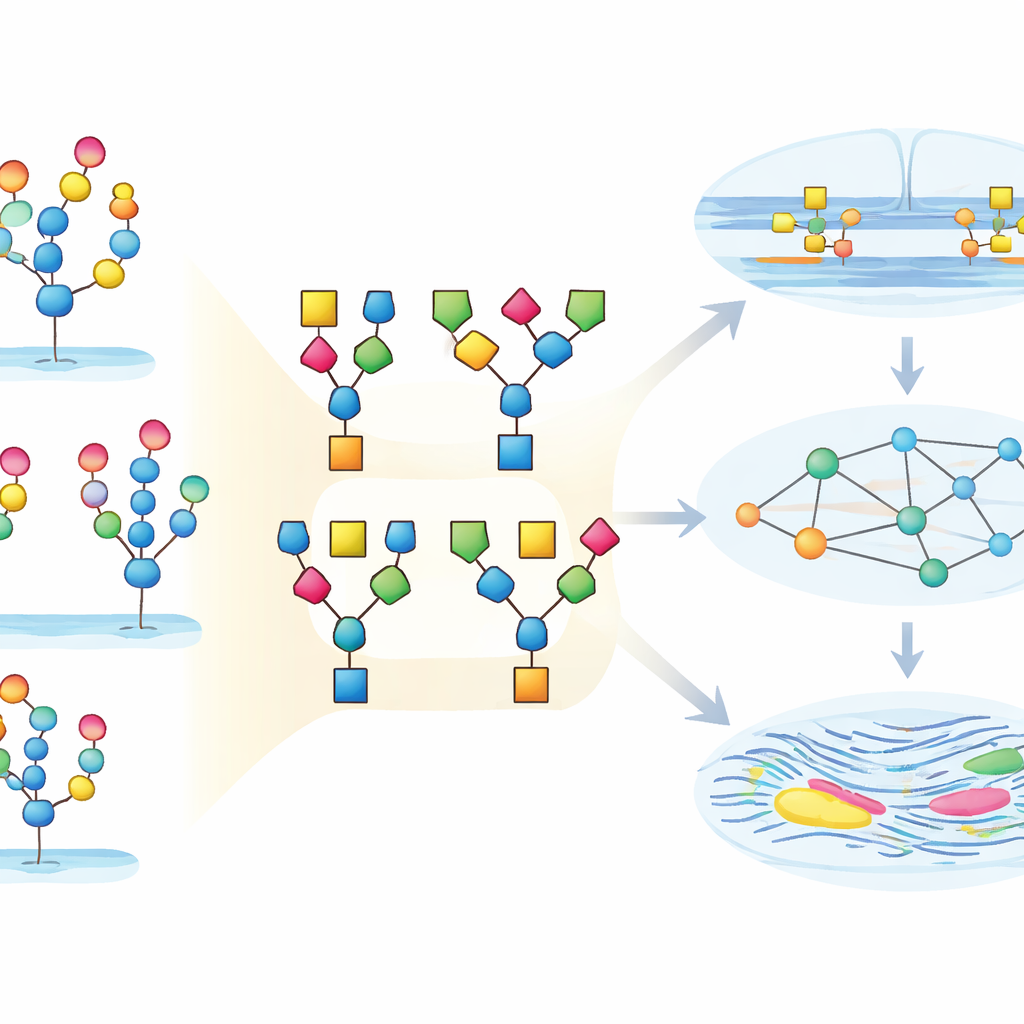
Turning Data Deluge into Biological Insight
In everyday terms, this work shows how a highly technical mass of “sugar-on-protein” measurements can be turned into a readable map of how tissues change over time. StrucGAP acts as both a quality guardian and a storytelling engine: it cleans the data, summarizes key sugar motifs, links them to the enzymes that shape them and to the pathways they influence, and automatically generates plots and reports that highlight the most important findings. In the mouse uterus, this reveals a coordinated shift toward more heavily decorated, Neu5Ac- and fucose-rich glycans tied to adhesion and tissue remodeling. More broadly, StrucGAP offers researchers a practical way to move from raw glycoproteomics data to testable ideas about how sugar coats help control health, disease, and aging.
Citation: Yang, M., Wu, Y., Zhang, Z. et al. StrucGAP: a modular, streamlined and traceable data mining platform for structural and site-specific glycoproteomics. Nat Commun 17, 2579 (2026). https://doi.org/10.1038/s41467-026-70560-7
Keywords: glycoproteomics, N-glycosylation, bioinformatics platform, uterine aging, protein glycosylation