Clear Sky Science · en
Structural transitions in the stepwise assembly of proteasome core particles
How Cells Take Out the Molecular Trash
Inside every one of our cells, used-up and damaged proteins must be broken down before they pile up like garbage. A giant molecular machine called the proteasome does much of this cleanup work, and it is already a target for anti-cancer drugs. This study peeks behind the scenes at how the proteasome’s catalytic core is built step by step in yeast cells, revealing unexpected shortcuts and safety checks that help ensure the final machine works precisely and only when it is ready.
Building a Protein-Shredding Barrel
The business end of the proteasome is a barrel-shaped core made of stacked rings of protein subunits. This barrel houses the hidden cutting sites that chop other proteins into small pieces. Because turning these cutters on too early would be dangerous, cells assemble the barrel in a series of partially built intermediates. The authors focused on early versions of the core, known as precursor complexes, which are usually rare and fleeting. By slightly slowing one of the last steps of assembly, they were able to capture and purify these early forms from yeast and image them using high-resolution cryo–electron microscopy, which freezes molecules in action and reveals their three-dimensional shapes.
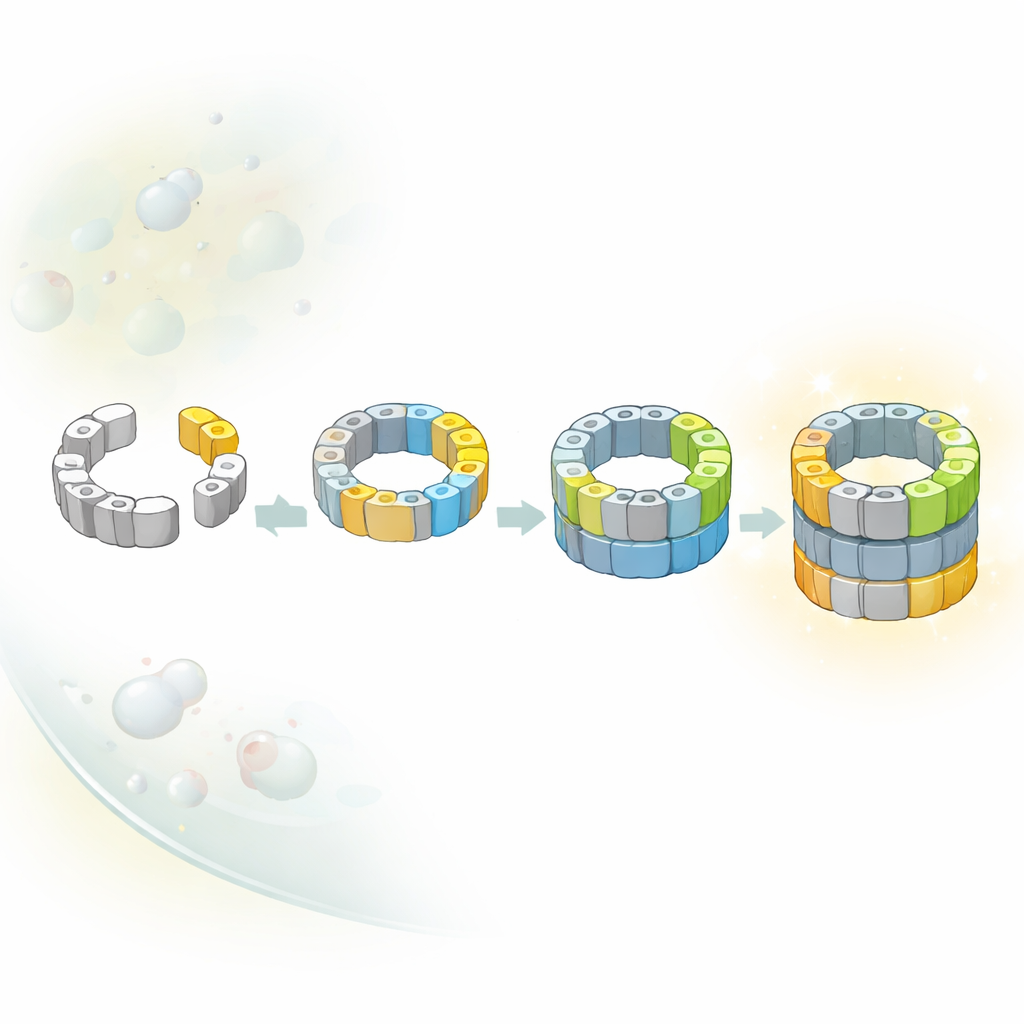
Multiple Roads to the Same Core
Classical textbook-style diagrams often suggest that complex machines inside cells are built along a single, fixed route. Here, the researchers discovered that this is not the case for the proteasome core. They identified several distinct intermediates that differ in which catalytic subunits have joined the half-finished barrel. By combining structural snapshots with genetic tricks that switch individual subunits on and off, they showed that at least two alternative paths lead from an early half-barrel to a nearly complete one. In one path, a particular subunit called β5 arrives first; in another, a different subunit, β1, can join earlier. The balance between these routes likely depends on which building blocks are most available in the crowded environment of the cell.
From Loose Parts to a Locked-In Cutting Chamber
The structures also reveal how the proteasome’s cutting sites are carefully primed as assembly proceeds. In early intermediates, key loop regions surrounding the buried cutting residues are floppy and poorly ordered, and the catalytic centers are still blocked by short “propeptide” segments. As additional subunits click into place around the ring, these loops gradually adopt more defined shapes that line up crucial amino acids needed for self-activation. Only when two half-barrels finally come together, completing the double-ring barrel, do these loops fully lock into their active arrangement and trigger removal of the blocking segments. This coupling of geometry and chemistry helps ensure that powerful cutting activity only appears inside a sealed chamber, protecting the rest of the cell.
Chaperone Helpers Guide and Then Let Go
Along the way, specialized helper proteins called chaperones shepherd the assembling barrel and prevent incorrect combinations. One chaperone, Ump1, starts out largely unstructured but folds progressively as more subunits surround it, eventually being trapped in the central cavity and then destroyed once the barrel is active. Another pair of chaperones, Pba1–Pba2, grips the outer ring surface in two clever ways. A flexible loop from Pba1 wedges between two outer-ring subunits like a spacer, holding them slightly apart and preventing premature closing. At the same time, the tail end of one outer-ring subunit (α1) helps position neighboring segments so that, at the final stage, the chaperones can be released and the ring can close properly. When the researchers deleted the α1 tail, the barrel stalled in a late, almost finished state that stubbornly retained its chaperones, confirming that this small region acts as part of a release switch.
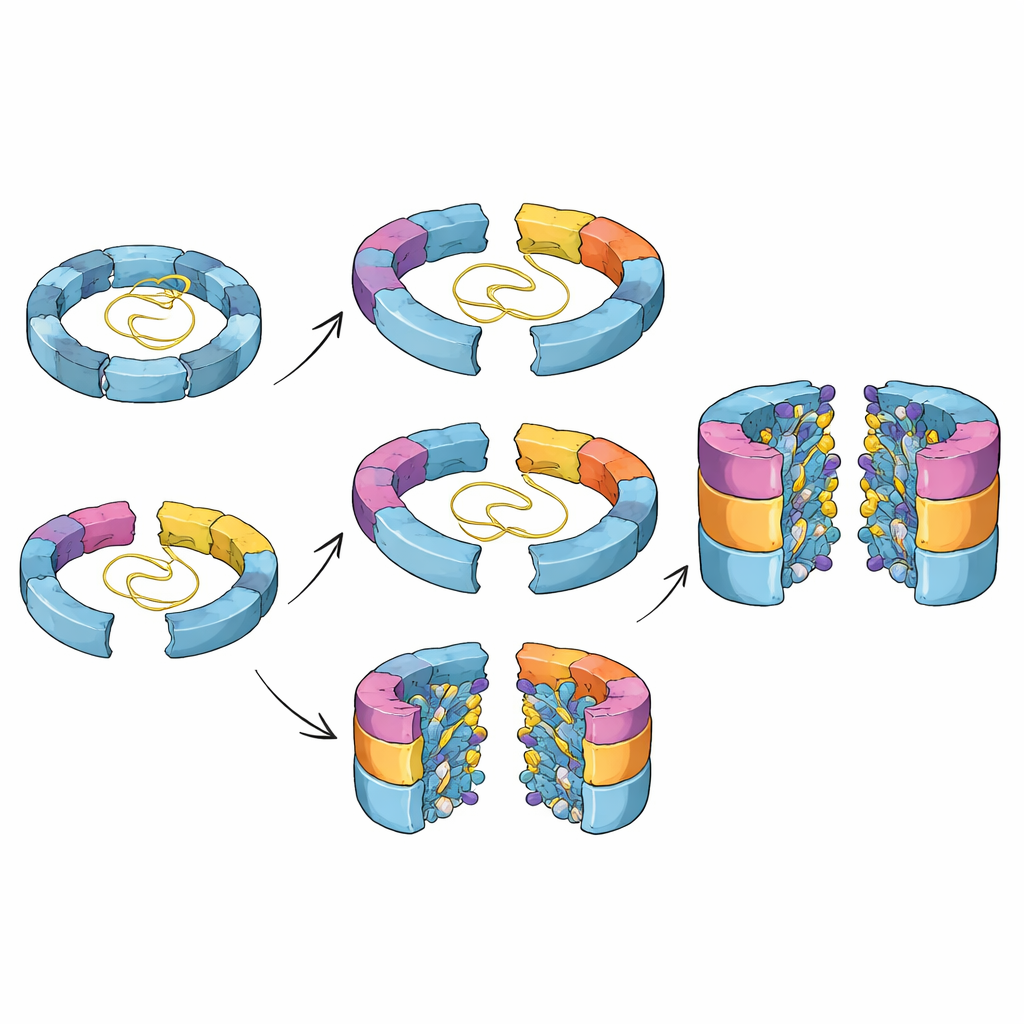
Why These Hidden Steps Matter
Together, these findings paint a detailed picture of how cells choreograph the assembly of a dangerous yet essential protein shredder. The work shows that the proteasome core can be built by more than one route, that its active sites are gradually sculpted into place, and that helper proteins not only stabilize early intermediates but are also wired into a timing circuit that ensures they depart at just the right moment. Understanding these hidden steps helps explain how small genetic changes in assembly factors can contribute to disease and may aid the design of drugs that fine-tune proteasome activity in cancer, immune disorders, or neurodegeneration by targeting its construction rather than its final form.
Citation: Mark, E., Ramos, P.C., Nunes, M.M. et al. Structural transitions in the stepwise assembly of proteasome core particles. Nat Commun 17, 2582 (2026). https://doi.org/10.1038/s41467-026-70525-w
Keywords: proteasome assembly, protein degradation, molecular chaperones, cryo electron microscopy, cellular quality control